Cell Membranes. Cells are surrounded by a membrane: the cell surface membrane or plasma membrane On...
-
Upload
joanna-fleming -
Category
Documents
-
view
223 -
download
1
Transcript of Cell Membranes. Cells are surrounded by a membrane: the cell surface membrane or plasma membrane On...

Cell Membranes

Cell Membranes
• Cells are surrounded by a membrane: the cell surface membrane or plasma membrane
• On an EM it appears as a double line• The distance
across the
membrane is
7-8nm

WHAT DOES A MEMBRANE LOOK LIKE?
The Fluid Mosaic Model


TRANSPORT ACROSS MEMBRANES

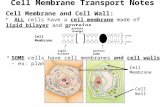
There are four main methods by which substances can move across a cell membrane:
• 1. Diffusion
Osmosis• 2. Active Transport• 3. Endocytosis and Exocytosis
Passive Transport

1: Diffusion
• Diffusion is the movement of molecules from a region where they are in high concentration to a region of low concentration.
• The difference in concentration is the concentration gradient
molecule
membrane

• Diffusion is a PASSIVE process
• There is a net movement of molecules down the concentration gradient until equilibrium is reached
• Ions move by diffusion along electrochemical gradients

Rate of diffusion
• The rate of diffusion depends on:– Concentration gradient– Temperature– Size of molecule– Lipid solubility
Extension: Look up Fick’s Law…

LipidBilayer
concentrationgrad ient
concentrationgradient
Intrinsic globular proteinswithin the membrane function
as carriers for the transportof certain molecules
Carriers are specific for the molecules that they transport them across the
membrane in the direction of the concentration gradient at a faster rate
than occurs for simple passive diffusion
Glucose molecules enter body cells from thebloodstream by facilitated diffusion
Facilitated Diffusion

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

LipidBilayer
concentrationgrad ient
concentrationgradient

Increasing external concentration of glucose
Rate ofuptake
A
Rate of uptakeincreases as the
glucose concentration
increases(i.e. the steepness ofthe concentration gradient increases)
Rate of uptake reachesa maximum at glucose
concentration A
No further increase inthe uptake rate despite the
increasing glucose concentration
When glucose concentrations exceed a certain value, there are
insufficient glucose carriers within the membrane for
the rate of uptake to increase any further
LIMITING FACTORS AND FACILITATED DIFFUSION
The rate at which molecules like glucose enter cells by facilitated diffusion is affected by factors other than the concentration gradient
The availability ofcarriers in the membrane
is the limiting factor

Summary of Passive Transport

2: Osmosis• Osmosis is the diffusion of water molecules from an area
of high concentration (of water) to an area of lower concentration across a semi-permeable membrane
watermolecules solute
molecules
low concentration of solute
membrane
high concentration of solute
high concentration of water low concentration of water
high water potential ( ) low water potential ( )
low osmotic pressure (OP) high osmotic pressure (OP)
dilute solution concentrated solution
net movement of water

Water Potential
Osmosis can be quantified using water potential, so we can calculate which way water will move, and how fast. Water potential (ψ, the Greek letter psi, pronounced "sy") is simply the effective concentration of water. It is measured in units of pressure (Pa, or usually kPa)
water always "falls" from a high to a low water potential
100% pure water has ψ = 0, which is the highest possible water potential, so all solutions have ψ < 0
you cannot get ψ > 0.

pure water = 0 kPa
water diffusesfrom 0 to -200 kPa
dilute solution = -200 kPa
concentrated solution = -500 kPa
water diffusesfrom -200 to -500 kPa
ψ ψψ

Cells and Osmosis.
The concentration of the solution that surrounds a cell will affect the state of the cell, due to osmosis. There are three possible concentrations of solution to consider:
• Isotonic solution a solution of equal concentration to a cell
• Hypertonic solution a solution of higher (salt) concentration than a cell
• Hypotonic solution a solution of lower (salt) concentration than a cell

Hypotonic solution Isotonic solution Hypertonic solution
AnimalCell
PlantCell
water enters, cell swellsand may burst (lysis).
no net movement ofwater, cell normal size
water leaves, cellshrinks and crenates
water enters, cell swellsa bit and becomes turgid.
no net movement ofwater, cell normal size
water leaves, cytoplasmshrinks and plasmolyses


3: Active Transport
• Pumping of molecules across cell membrane using a protein pump
UP a concentration Gradient
protein pump
ATP ADP + Pi
active site

• Most Active Transport systems are driven by metabolic energy derived from ATP
• Active Transport allows cells to uptake necessary ions and molecules and remove waste products
• These Mechanisms often referred to as Pumps
• Most common is Sodium Pump

Na+ / K+ Pump
• Present in all animal cells, most abundant and important of membrane pumps
outside cell3 Na+
2 K+
inside cell
-
+
ATP ADP + Pi
Na+K+
pump

• Sodium-Potassium Pump important in controlling cell volume, reducing Na thereby reducing water uptake by osmosis
• The accumulation of K ions are used in cell metabolic processes eg protein synthesis
• Na-K pump can be linked to active uptake of organic molecules such as glucose and amino acids

4: Endocytosis & Exocytosis
• Endocytosis

• Phagocytosis
Eg. Amoeba
White Blood Cells (neutrophils & monocytes)
• Pinocytosis
Eg. Protozoans, white blood cells, cells in embryos, liver and kidneys.