Training of A3-A3 side-to-side anastomosis in a deep corridor using a box with 6.5-cm depth:...
-
Upload
tomohiro-inoue -
Category
Documents
-
view
234 -
download
3
Transcript of Training of A3-A3 side-to-side anastomosis in a deep corridor using a box with 6.5-cm depth:...

Surgical Neurolog
Technique
Training of A3-A3 side-to-side anastomosis in a deep corridor using
a box with 6.5-cm depth: technical note
Tomohiro Inoue, MD, Kazuo Tsutsumi, MD4, Kuniaki Saito, MD, Shinobu Adachi, MD,
Shota Tanaka, MD, Naoto Kunii, MDDepartment of Neurosurgery, Showa General Hospital, Tokyo 187-8510, Japan
Received 23 February 2006; accepted 4 March 2006
Abstract Background: Cerebral revascularization in the deep surgical field is technically challenging.
www.surgicalneurology-online.com
0090-3019/$ – see fro
doi:10.1016/j.surneu.2
Abbreviations: OA
artery; SCA, superior
4 Corresponding a
E-mail address: k
Especially, side-to-side anastomosis like A3-A3 could be technically more difficult compared with
end-to-side anastomosis. To improve surgeon’s dexterity and maneuverability in the deep surgical
field, the authors developed an easily accessible and well-simulating training system using prosthetic
tubes and a box.
Methods: Two prosthetic tubes (silicon tube, 1.2 mm in diameter) are mounted in parallel on the
bottom of 6.5-cm-deep emptied dtissue paper box.T The orifice of the box is restricted to 2 � 2 cm to
simulate a deep and narrow surgical corridor. Using bayonet-shaped micro needle holder and forceps,
the side-to-side anastomosis of the tubes is performed with 10-0 nylon under operative microscope.
Results: Prosthetic tubes well simulated real A3-A3 anastomosis. From the standpoint of technical
difficulty, this training system needed slightly higher level of dexterity compared with real A3-A3
anastomosis because of narrower and deeper surgical corridor, and the wall of prosthetic tube was
slightly thicker and more inflexible. After this training, the surgical technique in real A3-A3
anastomosis was improved.
Conclusions: This training system worked well to ease the transition from anastomosis in shallow
surgical field to deep and narrow surgical field. The prosthetic tube we used approximates real A3
relatively well, and the ease in setting up this system enabled repeated practice, which resulted in
steep learning curve of the technique.
D 2006 Elsevier Inc. All rights reserved.
Keywords: Cerebral revascularization; Training; Side-to-side anastomosis
1. Introduction
Cerebral revascularization in deep surgical field remains
a critical part of the neurosurgical skill for treating complex
aneurysm and very rarely atherosclerotic occlusive cerebro-
vascular disease. Because of the inherent difficulty of this
technique, as well as the limited surgical case volumes, only
some experienced neurosurgeons perform this technique.
Here, we present a unique microsurgical training system in
deep surgical field that is easy to set up and works as an
nt matter D 2006 Elsevier Inc. All rights reserved.
006.03.035
, occipital artery; PICA, posterior inferior cerebral
cerebellar artery; STA, superficial temporal artery.
uthor. Tel.: +81 424 61 0052; fax: +81 424 64 7912.
[email protected] (K. Tsutsumi).
adjunct to transit from anastomosis in shallow surgical field
to deep and narrow surgical field.
2. Materials and methods
An emptied dtissue paper boxT with depth of 6.5 cm is
used. Because the longest bayonet-shaped (bent-knee–
shaped) micro forceps available that can handle the needle
and thread of a 10-0 nylon is 7 cm long from the tip to
the angle of knee, we choose this size of box. In addition,
we expect that this depth probably covers most of the
neurosurgical deep anastomosis. The orifice of the tissue
paper box is restricted to 2 � 2 cm, which is as narrow as
y 66 (2006) 638–641

T. Inoue et al. / Surgical Neurology 66 (2006) 638–641 639
a 1-yen Japanese coin, with rigid tape (Fig. 1). In this
condition, if we use straight instruments, the hands or
fingers will shadow the surgical field under the operative
microscope. Only when the bayonet-shaped instruments
are used bilaterally, will dshadowingT not happen in higher
magnification. Two prosthetic silicon tubes of 1.2-mm
diameter (Fujita, Tokyo, Japan) are mounted in parallel on
the gauze for side-to-side anastomosis at the bottom of
the box by just passing each end of the tube under the
fiber of gauze. The usual surgical skin marker can be used
to visualize well the orifice of the darteriotomy.T Slightly
internally curved incision is made with curved micro-
scissors on both tubes. Firstly, with a 10-0 nylon, the
running suture of the back wall is performed (Fig. 2), with
the first stitch passed from outside to the inside on the left
tube and the last stitch passed from inside to the outside
on the right tube (in the case of a right-handed surgeon).
Both ends of the thread of the running suture are pulled
gently to tighten once. Then, the stay suture is performed
on each end of arteriotomy, and the threads of stay
suture’s knots are left in some length. After tightening the
running suture by pulling both ends of the thread again,
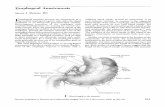
Fig. 1. The suturing training is performed at the bottom of 6.5-cm-deep box
through the orifice as narrow as a 1 yen Japanese coin (2 cm in diameter)
with bilateral bayonet-shaped micro instruments (top). The training
condition is simulating the real A3-A3 anastomosis well (bottom).
Fig. 2. During suturing the back wall in the training (top) and in the real
A3-A3 anastomosis (bottom).
these are tied to each side of thread of the stay suture’s
knot (Fig. 3). The anterior wall is then sutured with in-
terrupted stitches (Fig. 4).
3. Result
The bottom part of Figs. 1-4 shows real A3-A3
anastomosis operation of a 67-year-old woman with a right
A2 atherosclerotic occlusion with hemodynamic ischemia.
In this operation, the 9-0 nylon was used so that the running
suture of the back wall will not tear. In this patient, the site
suitable for anastomosis was just below the bridging vein,
and the depth and narrowness of the surgical corridor made
it slightly more difficult than the usual A3-A3 anastomosis.
The training system illustrated here well simulated this
condition (Fig. 1). In addition, this training needed a slightly
higher level of dexterity compared with real A3-A3
anastomosis because of the narrower and deeper surgical
corridor, and the wall of the prosthetic tube was slightly
thicker and more inflexible. After repeated training with this
setting, the occlusion time of the above-mentioned A3-A3
operation was 35 minutes, which is remarkably shortened
compared with the previous occlusion time of 55 minutes

T. Inoue et al. / Surgical Neurology 66 (2006) 638–641640
(in the operation of ruptured giant anterior communicating
artery aneurysm).
4. Discussion
Although the international cooperative study of external/
internal arterial anastomosis published in 1985 brought
about a decrease of microvascular cerebral revascularization
[2], the subset of aneurysm with complex anatomy, skull
base tumors and some very restricted atherosclerotic
cerebrovascular occlusive cases still require this technique
[5,11]. Various training methods have been developed to
overcome difficult circumstances with lowered surgical
volume and to improve revascularization technique
[1,3,4,6-10,12,13]. We also developed a basic anastomosis
training system that we are doing routinely [14]. However,
the training of anastomosis in the deep and narrow corridor
has not been well discussed despite the challenging nature
of this technique even after the mastery of anastomosis in
the shallow surgical field.
We developed this training system as an adjunct to ease the
transition from anastomosis in the shallow surgical field to
the deep and narrow surgical field. Firstly, we simply sutured
Fig. 3. Confirming the status of the back wall after completion of running
suture and stay suture in the training (top) and in the real A3-A3
anastomosis (bottom).
Fig. 4. The anterior wall is sutured with interrupted stitches in the training
(top) and in the real A3-A3 anastomosis (bottom).
the neighboring fibers of the gauze at the bottom of the
6.5-cm-deep box with a 10-0 nylon through the orifice
approximately 3 � 3 cm with the bayonet-shaped micro
needle holder in the dominant hand and the straight forceps in
the nondominant hand. This training was also very effective
as long as the superficial temporal artery–M2 anastomosis or
occipital artery–posterior inferior cerebellar artery anasto-
mosis was concerned. However, in some superficial temporal
artery–superior cerebellar artery anastomosis or in revascu-
larization of the P2 area, the situation wherein the surgical
corridor is so narrow and deep that maneuverability of the
nondominant hand with the straight forceps was restricted
and that the straight forceps shadowed the surgical field
presented. In addition, in the A3-A3 anastomosis, we
encountered the situation in which the corridor was narrower
and deeper compared with the usual A3-A3 anastomosis at
the corner of genu because of the distribution of bridging
vein. After experiencing those difficulties, we restricted the
orifice of the box to 2� 2 cm, in which the bilateral bayonet-
shaped micro instruments could avoid shadowing. During the
training course, special attention was paid to improve the
maneuverability of the nondominant hand with the bayonet-
shaped forceps that are apparently more difficult to deal with
than straight forceps. The reason why we used side-to-side

T. Inoue et al. / Surgical Neurology 66 (2006) 638–641 641
anastomosis in the training system was that this technique
needed a higher level of dexterity from the standpoint of
anastomosis itself, such as bilateral curved arteriotomy and
running suture of the back wall, compared with end-to-side
anastomosis. The prosthetic tube was used simply because
the animal model was not available in our institution.
However, the prosthetic tube we used simulated the real A3
well as for the caliber and the thickness of the wall. In
addition, this tubing is somewhat more inflexible than the real
A3 wall, which resulted in a slightly higher demand of
dexterity during suturing training. This inherent nature of the
prosthetic wall provided the good occasion to master a
precise controlled stabilization of the vessel wall by the
forceps held in the nondominant hand.
The balance of the ease to set up and the adequacy in
simulating clinical setting are the important aspects of the
training system. Our system apparently has a disadvantage
in simulating live anastomosis compared with the animal
model. However, once the definite anastomosis technique
with high patency rate is accomplished in the shallow
surgical field, the higher level of dexterity that is inherent to
the deep and narrow corridor itself would be the most
essential element of the training system. As the technical
difficulty increases, the repeated practice would be neces-
sary as well. From those standpoints, we believe that our
training system, which simulates the most difficult condition
and is easy to set up, would contribute to the acquisition and
refinement of skills in deep anastomosis.
References
[1] Crosby NL, Clapson JB, Buncke HJ, Newlin L. Advanced non-animal
microsurgical exercises. Microsurgery 1995;16:655 -8.
[2] EC/IC bypass study group: failure of extracranial-intracranial arterial
bypass to reduce the risk of ischemic stroke: results of a international
randomized trial. N Engl J Med 1985;7:1191-200.
[3] Hino A. Training in microvascular surgery using a chicken wing
artery. Neurosurgery 2003;52:1495-8.
[4] Hui KCW, Zhang F, Shaw WW, Kryger Z, Piccolo NS, Harper A,
Lineaweaver WC. Learning curve of microvascular venous anasto-
mosis: a never ending struggle? Microsurgery 2000;20:22-4.
[5] Inoue T, Tsutsumi K, Ohno H, Shinozaki M. Revascularization of the
anterior cerebral artery with an A3-A3 anastomosis and a superficial
temporal artery bypass using an A3-radial artery graft to trap a giant
anterior communicating artery aneurysm: technical case report.
Neurosurgery 2005;57(ONS Suppl 1):ONS-207.
[6] Kim DC, Niazi ZBM, Hayward PG, Morrison WA. Training model for
microvessel anastomosis. Microsurgery 1994;15:820 -1.
[7] Klein I, Steger U, Timmermann W, Thiede A, Gassel HJ. Microsur-
gical training course for clinicians and scientists at German university
hospital: a 10-year experience. Microsurgery 2003;23:461-5.
[8] Korber KE, Kraemer BA. Use of small-caliber polytetrafluoroethylene
(Gore-Tex) grafts in microsurgical training. Microsurgery 1989;10:
113-5.
[9] Lahiri A, Lim AYT, Qifen Z, Lim BH. Microsurgical skills training: a
new concept for simulation of vessel-wall suturing.Microsurgery 2005;
25:21 -4.
[10] Matsumura N, Hamada H, Yamatani K, Hayashi N, Hirashima Y,
Endo S. Side to side arterial anastomosis model in the rat internal and
external carotid arteries. J Reconstr Microsurg 2001;17:263-6.
[11] Quinones-Hinojosa A, Lawton MT. In situ bypass in the management
of complex intracranial aneurysms: technique application in
13 patients. Neurosurgery 2005;57(ONS Suppl 1):ONS-140-5.
[12] Senior MA, Southern SJ, Majumder S. Microvascular simulator—a
device for microanastomosis training. Ann R Coll Surg Engl 2001;
83:358-60.
[13] Yoleri L, Songqr E. Different venous end-to-side microanastomosic
techniques: comparative study in a new rat model. Ann Plast Surg
2002;48:410 -4.
[14] Inoue T, et al: Effectiveness of suturing training with 10-0 nylon under
fixed and maximum magnification (�20) using desk type microscope.
Surgical Neurology [in press].