Substances, Compounds and Mixtures. Main Idea Supporting Facts The main concepts for this lecture...
-
Upload
dwain-dean -
Category
Documents
-
view
218 -
download
1
Transcript of Substances, Compounds and Mixtures. Main Idea Supporting Facts The main concepts for this lecture...

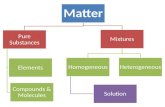
Substances, Compounds and Mixtures

Main Idea Supporting Facts The main concepts for this lecture are:
• Elements• Compounds• Substances• Mixtures

Main Idea Supporting Facts • Element • A chemical element is a pure
chemical substance that cannot be broken down into a simpler substance. It is made of only one type of atom (we’ll discuss atoms more in the next unit).

Examples of Elements (SPONCH)
Sulfur
Nitrogen
Carbon
Hydrogen

Main Idea Supporting Facts
• Compounds • A combination of two or more different elements (they must chemically combine to form a formula)

Examples of Compounds
Water Sodium hypochlorite - Bleach
sugar
NaClO- H2O
C12H22O11

Question Time!
Which of the following is a compound?
A.Copper wire
B.Frozen water
C.Pure gold coin
D.Aluminum Foil

Main Idea Supporting Facts • Substance • Matter that has a defined
composition. • Also called a pure
substance.• Examples are distilled
water, table salt, fructose (fruit sugar)

Main Idea Supporting Facts • Mixtures • A mix of two or more substances.
• If the substances experience a physical change, they can be separated by physical means, such as dissolution (to fall out of a solution), separation, etc…
• If the substances experience a chemical change, then they cannot be separated via physical means.
• There are two types:• Homogeneous• Heterogeneous

Example of a Mixture being made via a CHEMICAL change
Mentos and Coca-Cola

Main Idea Supporting Facts • Homogeneous
Mixture
• The mixture is the same throughout. It is also called a solution.
• With food, this means each bite is exactly the same as the next bite.

Main Idea Supporting Facts • Heterogeneous Mixture • A mixture that is NOT the same
throughout.• In food, this would be items
where one bite is different from the next.

Question Time!
Which of the following is a heterogeneous mixture?
A.Blood
B.Chocolate Ice Cream
C.Pasteurized Milk
D.Pulp-free Orange juice

Main Idea Supporting Facts • Vocabulary for Liquid
Mixtures
• Mixtures in chemistry are often in the form of liquids. A solute is an object that is mixed into a larger amount of chemical, called a solvent. It will dissolve into the solvent.
• A colloid and suspension are two types of liquid mixtures. Colloids are homogeneous mixtures that do not settle out of solution on their own. Milk an example of one
• A suspension is a homogeneous mixture that will have its particles settle out when left alone. Iced Tea and Lemonade would be an example of this

Main Idea Supporting Facts • Some ways to separate
mixtures?• Hand separation – If mixture is dry (like
sand and seashells), you can pick out the unlike parts by hand.
• Filtration – Using a filter system to separate smaller particles from larger ones (like washing of archaeological artifacts to rid them of dirt, silt, clay and sand)
• Distillation – boiling a liquid substance so the particles separate out their boiling parts (i.e., how we get gasoline from crude oil)
• Centrifugation – spinning a liquid in a small chamber until its components separate out via their densities (i.e., blood centrifugation)