Mixtures. Matter SubstancesMixtures Elements Compounds Heterogeneous Mixtures Homogeneous Mixtures...
-
Upload
mark-oliver -
Category
Documents
-
view
267 -
download
1
Transcript of Mixtures. Matter SubstancesMixtures Elements Compounds Heterogeneous Mixtures Homogeneous Mixtures...

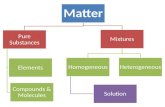
MixturesMixtures

Matter
Substances Mixtures
Elements CompoundsHeterogeneousMixtures
HomogeneousMixtures
Mixtures Substances separated by physical methodsCompounds Substances separated by chemical methods

MixturesMixtures
•Combo of 2 or more pure substances
•Physically combined NOT chemically combined
•Each substance retains its own identity and properties

MixturesMixtures• Variable composition
• No unique properties
• Ex: sugar and sand mixed together
• Separated by physical methodsphysical methods
• May be homogeneous or heterogeneous

TypesTypes of Mixtures of Mixtures• Heterogeneous: See boundary or
regions that look different
Ex:
•colloids •suspensions•ice water•granite•cereals

Colloid: milk
Suspension: smoke/smog
Heterogeneous mixture: ice water
Heterogeneous mixtures: cereal/granite

• Homogeneous: constant composition throughout, single phase
Ex:
•Solutions (all 3 phases)•Air•Windex•kool-aid

Air
Homogeneous Solutions:
Drink mixWindow cleaner

Hints for MixturesHints for Mixtures• Solutions (gas & liquid phases) transmit
light • No particles big enough to scatter light • Look translucent (see-through)
• Suspensions look cloudy – scatter light• Particles big enough to scatter light• Settle upon standing

Where does this liquid fit?
Homogeneous?
Heterogeneous?

Particle Diagrams
Atoms only: monatomic element
Molecules:diatomic element

Particle Diagrams
Molecules: •triatomic compound
Mixture: •monatomic element•diatomic element•triatomic compound

Conservation of MassConservation of Mass
•Mass before = Mass after•# atoms before = # atoms
after

Separating MixturesSeparating Mixtures• Physically combined• Separation based on physical properties
– Sorting: Appearance– Filtration: Size– Distillation: Boiling Point– Crystallization: Solubility– Magnet: Magnetization– Chromatography: “Travel” ability

Filter:
Solid particles can be separated from liquid

DistillationUsed to separate liquids with different boiling points

Paper Chromatograhy
Can separate liquids with different solubilities

Crystallization
Can separate solids dissolved in liquids