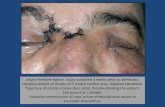
USE OF INFLIXIMABE IN 25 LIBYAN PSORIASIS VULGARIS...
-
Upload
truongphuc -
Category
Documents
-
view
213 -
download
0
Transcript of USE OF INFLIXIMABE IN 25 LIBYAN PSORIASIS VULGARIS...
1
USE OF INFLIXIMABE IN 25 LIBYAN PSORIASIS VULGARIS PATIENS
A Altrabulsy1, A.H.Griew2, E .M Elmangoush1, G.A.Duweb1
1 Dermatology Department, 2 Family& Community Department . Faculty of medicine,
Benghazi University, Benghazi-Libya
INTRODUCTION:
Psoriasis is a distressing, chronic immune mediated, inflammatory disease that affects skin and
joints. It is characterized by infiltration of the skin by activated T cells, abnormal keratinocyte
proliferation, cytokines, chemokines, and excessive production of tumor necrosis factor (TNF) in
the psoriatic lesion(1).
Estimates of the prevalence of psoriasis vary from 2% to 3%, with rates varying between
countries and races (2). Burd 2006 has been found that, a quarter of patients develop the disease
before the age of 20 years. A further peak in incidence is recorded in the fifth and sixth decades.
The age of onset seems to be genetically determined ( 3,4 ).
Clinically have been found that, around 80% of individuals present with chronic plaque psoriasis
characterized by well demarcated, red, thickened patches of skin which become elevated and
covered with an adherent, silvery scales ( 5,6).
Erythrodermic psoriasis denotes the involvement of the entire skin surface which is rare and it
can be life threatening with a risk of sepsis and thermoregulatory disruption. Pustular psoriasis
may be localized or generalized. In the generalized variant sheets of sterile pustules are found on
a background of red skin. Fever, arthralgia and malaise are common accompaniments(7).
Psoriatic arthritis affects between 5% and 30% of psoriasis patients and can be aggressive and
deforming. Both the axial and the peripheral joints may be involved with several overlapping
patterns: asymmetric, oligoarticular disease, predominant distal arthritis characterized by
involvement of the distal interphalangeal joints, arthritis mutilans, symmetric polyarthritis
(closely resembling rheumatoid arthritis) and axial involvement/spondolarthropathy
Enthesopathy and dactylitis are characteristic features.(8, 9)
2
Infliximab belongs to the class of biological response modifiers called Tumor Necrosis Factor
(TNF-α) blockers, with human constant regions and murine variable regions. It engineered from
human and mouse antibody molecules. It works by directly binding to both trans-membrane and
soluble TNF-α molecules in the blood and diseased tissue. with high affinity, specificity, and
avidity.(10 )
Infliximab helps to control angiogenesis, a key factor in the pro-inflammatory state of psoriasis,
by down-regulating angiopoietin, a growth factor critical for new blood vessel growth, and its
receptor. Complementary to its anti-inflammatory effect that is mediated through the
neutralization of TNF-α, infliximab normalizes keratinocyte differentiation (8, 9,10).
AIM OF THE STUDY
To assess the efficacy and tolerability of infliximab monotherapy in Libyan patients with severe
psoriasis vulgaris..
SUBJECTS AND METHODS
The study group included twenty five adult patients who had sever psoriasis vulgaris involving
more than 10% of the body surface area, over one year ( December 2010 to November 2011) at
dermatology department Al-Jamhoria hospital, Benghazi-Libya. All patients had signed written,
informed consent.
Each patient was exposed to detailed medical history, clinical and laboratory assessments
including complete blood count, chemistries, urine analysis, serum for human chorionic
gonadotrophin concentration, chest x- ray, tuberculin test to exclude tuberculosis, serological
examination including HIV, HBV and HCV.
INCLUSION CRITERIA
Adult, healthy patients by investigation and clinical examination has no evidence of any chronic
illness, or infections.
Patients had severe psoriasis vulgaris more than 10% of body surface area (BSA).
EXCLUSION CRITERIA
Moderate to sever CHD.
Pre-existing CNS disorder.
3
Untreated chronic infection, such as TB or hepatitis.
Elderly patients and patients with kidney or liver failure.
Pregnancy and breast feeding.
Measures of psoriasis activity (Psoriasis Area Severity Index) (PASI) , as screening baseline and
before each dose. Each patient prepared by measuring the BP and pulse and given hyrocortisone
IV, antihistamine.
Each patient received infliximab IV infusion 5mg/kg in 250 ml normal saline, slowly over hours.
The treatment doses given at 0, 2, 6 Weeks and then every 8 weeks. Patient assessment was
based on measuring PASI score and observing any side effects and repeating the investigations at
each visit.
RESULTS
All patients were assigned infliximab 5 mg/kg. Two patients withdrew during the course of the
study. One patient was withdrawn at week 2, secondary to an infusion reaction and another
patient was withdrawn 7 days after her first infusion, because of hypotension. Figure 1
We determined the disease severity by PSORIASIS AREA SEVERITY INDEX (PASI) Score.
Most patients showed good improvement at 3rd dose, total improvement occurred mainly at 5th
and 6th dose. Mean total score of base line PASI for 25 patients was (32.88±10.871.
Among 25 patients were included in this study 15 (60%) were males and 10 (40%) were females
(Fig.2). The patient´s age was ranging from 25 to 51 years, patients aged (40-44) years had the
highest frequency 35%, followed by those who aged (30-34) years representing 30% of patients
(Fig. 3). Figure 4 showed that, patients who had the disease for a duration ranged between 10- 14
years represented the highest frequency ( 30 % )
4
Efficacy
Therapeutic response was evaluated in accordance with PASI. About 17 of 23 (72.2%) patients
treated with the infliximab 5 mg/kg had at least 50% improvement in the PASI score, while (16)
of (23 ) (67% ) had at least 75% improvement in PASI score and 23 patients (100%) had at least
90% improvement in the PASI score (Fig.4). The mean PASI scores at baseline for infliximab
5mg/kg was (32.8 ). At week 2 and 6, the mean PASI scores were (16.2 ), (2.2 ) respectively,
and (0.5 ), (0.1) at week 16, 24 respectively(Fig.5).
The mean percentage improvements in PASI scores were higher 92% as early as 6th week of
therapy and reached maximum improvement 100% at 32 week as compared to initial time of
treatment. (Fig.6). Photographs illustrating shows the typical degree of improvement observed
from baseline PASI (Fig.7), at week 6th (Fig.8), at week 32th (Fig.9). Improvement in nail was
observed early at 6th week ,as shown in (Fig. 10,11)
Adverse events
There were no serious adverse events. UTI was the adverse event that occurred in a higher
proportion (10%). The acute adverse effect was infusion reactions were reported in one patient
during the initial first week, and hypotension for another one which occurred at week 2 this led
to discontinuation of the drug. Infections were occurred in 2 patients (8 %) developed UTI, two
at 2nd week and week 16th also another patient at week 6th developed URTI & UTI from week
0. All delayed adverse effects were followed up and treated without the drug having to be
interrupted. ( Fig.12 ).
5
Figure.1: Summary of patients in the study.
25 patients with severe
psoriasis vulgaris were
screened.
Full medical history
and laboratory
assessments
25 patients assigned infliximab
5mg/kg
2 patients were dropped out
due to severe hypotension and
urticaria.
23 patients completed the course of
infliximab.
20 patients with out
complications
3 patients with
complications
8
Figure 4: Duration categories of psoriasis cases in years
15
5
30
15
25
10
0
5
10
15
20
25
30
35
1-4 5-9 10-14 15-19 20-24 25 and
above
%
9
Figure 5: Mean total score of PASI for each dose
32.8
16.2
0 0.1 0.5
2.2
0
5
10
15
20
25
30
35
First
dose
Second
dose
Third
dose
Fourth
dose
Fifth
dose
Sixth
dose
10
Figure 6: Mean percentage of PASI improvement
54.5
92.3
45.5
98.2 99.6 100
0
20
40
60
80
100
120
Base line Second week
Tenth week Sixteenth week
Twenty fourth week
Thirty two
week
Mean %
12
Figure 8 : Much improvement in psoriatic plaques at 6th week.
Figure 9: Total clearance at 32 week.
15
Figure 12: Adverse effect of Infliximab among
Psoriatic patients
76%
4%
4%
12%
4%
None
Infusion reaction
(IR)
Hypotension
Urinary tract
infection (UTI)
UTI & URTI
16
DISCUSSION
Psoriasis is a distressing, chronic immune mediated, inflammatory disease that affects skin and
joints. It is characterized by infiltration of the skin by activated T cells, abnormal keratinocyte
proliferation, cytokines, chemokines, and excessive production of tumor necrosis factor (TNFα)
in the psoriatic lesion( 1).
The mean age of onset was (37.7year ±6.815) ranged from 25-51year which in contrast to study
by Duarte A.A 2011 with mean age 49.2year (11). The peak age group in our study was
observed in the 40-44year age group (35%), which disagreement with Sinniah B 2010 where the
peak age group was 40-60year (17.2%) (12).
About the mean duration of the disease was (14.90year ±8.25years) ranging from 2-27year,
(30%) of patients were between 10-14year which is in contrast to study by Natarajan V 2010
where the mean duration of the disease was found to be 9.63 years ranged from 1 month -49 year
(13). Patients with psoriasis are at significant risk of developing psoriatic arthritis. The frequency
of psoriatic arthritis appears to be strongly related to the degree of skin severity. A population
based study indicated that the prevalence of psoriatic arthritis in patients with ≤ 1% body surface
area, 1-2% BSA, 3-10% BSA and 10+% BSA was 6%, 14%, 18% and 56% respectively(14). In
our study 6 patients (30%) out of 20 had joint involvement.
Regarding PASI is considered by many to be the gold standard severity assessment tool and
consists an average measure of 4 parameters; redness, thickness and scaling of lesions and extent
of disease. Each parameter is graded as 0–4 with 0 equating to no disease and 4 severe
involvement. The body is divided into 4 areas (head, arms, trunk to groin, legs to top of
buttocks). The scores are summed, a percentage of the area covered by psoriasis estimated and a
multiplier applied to each area generating a final PASI. PASI ranges from 0 to 72 with PASI >
10 recognized as indicative of severe disease (15).
A 75% reduction in PASI from baseline (PASI 75) is commonly used as an indicator of disease
response, and in most clinical trials this is the primary efficacy endpoint. For individual patients
a smaller improvement in PASI can be clinically meaningful (16). This is reflected in clinical
trials which frequently report PASI 50 rates as well as PASI 75. The mean PASI score at
17
baseline was 32.8 which show marked decrease at third dose (6th week) where it became 2.2 and
reached zero at sixth dose (32nd week), the mean percentage of PASI improvement at base line
was 45.5% and had shown marked improvement at third dose (6th week) and reached total
clearance at sixth dose (32nd week), as compared our study with finding by Chaudhari 2001, the
mean PASI at base line was 17.5 with marked decrease at (2nd week) (17).
50% improvement in PASI had been observed in 6 (33.3%) patients out of 18 after first dose
and 7 patients (38.8%) were show improvement in PASI after 2nd dose .75% improvement in
PASI was observed in one patient (5.5%) after first dose, 2 patients (11.1%) at second dose, 7
patients (38.8%) at third dose and 2 (11.1%) patients at fourth dose, as compared our study with
finding by Reich et al 2005 study of 378 patients with severe psoriasis vulgaris 75%
improvement in PASI occur after 10th week in 80% of patients(18).
90% improvement in PASI was observed at 2nd week by 2 patients (11.1%), 11 patients (61.1%)
at 6th week, 16 patients (88.8%) at week 16th and at 24th week all patients had 90%
improvement, as compared this finding with Reich et al 90% improvement occur in 57% of
patients (18).
Infliximab had been generally well tolerated and available for clinical use in adult. The acute
adverse effect was infusion reactions were reported in 1 (5%) of patients during the initial first
week of treatment and hypotension in one patient (5%), this lead to discontinuation of the drug
,as compared our study with Kipnis et al infusion reaction were reported in 20% of patients
receiving infliximab(19). UTI was the adverse event that occurred in a higher proportion (10%),
two at 2nd week and week 16th also another patient at week 6th developed URTI & UTI . All
delayed adverse effects were followed up and treated without the drug having to be interrupted.
CONCLUSION & RECOMMENDATION
Infliximab was found to be safe and effective for treatment of sever psoriasis vulgaris.
Infliximab has a rapid induction phase and 75% improvement was reported after 3rd dose (6
weeks ) in majority of the studied cases. Additional effect was observed, early improvement in
psoriatic arthritis and nail changes improvements including complete regrowth of destructed
18
nails and return to normal. Mild adverse effects including hypotension, urticarial rash and
urinary tract infection has been reported and no serious side effects.
REFERENCES
1-Winterfield L, Menter A. Psoriasis and it is treatment with infliximab tumor necrosis factor
alpha blockade. Dermatol Clin. 2004;22(4) :437-47 .
2- Joel M. Gelfand, Rachel Weinstein, Steven B. Porter. Prevalence and treatment of Psoriasis in
the United Kingdom .Arch. Dermatol. 2005: 141 (12): 1537-1541.
3- Burd RM. Psoriasis: a general overview. Br J Hosp Med. 2006;67:259–62.
4- Lebowohl M. Psoriasis. Lancet. 2003;361:1197–204.
5- Camp RDR, El-Rachkidy RG, Young HS,et al. Immunological evidence for increasing
reactivity to secreted Streptococcus pyogenes proteins in chronic plaque psoriasis: BrJ
Dermatol. 2006;154(Suppl 1):5
6- Elder JT .PSORS1: linking genetics and immunology. J Invest Dermatol. 2006;126:1205-6.
7- Lebowohl M, Callen JP. Obesity, smoking and psoriasis. JAMA. 2006;295:208–10.
8- Leman JA, Burden AD. Treatment of severe psoriasis with infliximab. Br Dermatol.
2008;4(6):1165-1176.
9- Bos JD, De Rie MA. The pathogenesis of psoriasis: immunological facts and speculations.
Immunol Today 1999; 1: 40–45.
10- Scallon BJ, Moore MA, Trinh H, etal. Chimeric anti-TNF-alpha monoclonal anti-body cA2
binds recombinant transmembrane TNF-alpha and activates immune effector functions.Cytokine
1995;7:251-9.
11-Duarte AA, Chehin FB. Moderate to severe psoriasis treated with infliximab- 53 patients
profile, efficacy and adverse effects. An Bras Dermatol 2011;86:257-63.
12- Sinniah B, Epidemiology of psoriasis in Malaysia: A Hospital Based Study. Med J Malaysia
2010;65:112-114.
13- Natarajan V, Nath AK, Thappa DM, et al. Coexistence of onychomycosis in psoriatic nail: A
descriptive study. Indian J Dermatol Venereol Leprol 2010;76:723-731.
14-Gelfand JMGD, Mease PJ, Smith N et al. Epidemiology of psoriatic arthritis in the population
of the United states. J Am Acad Dermatol 2005;53:573-577
19
15- Feldman SR, Krueger GG. Psoriasis assessment tools in clinical practice. Ann Rheum
Dis.2005;64(Suppl ii):65–8.
16- Carlin CS, Feldman SR, Krueger JG, et al. A 50% reduction in the Psoriasis Area and
Severity Index (PASI 50) is a clinically significant endpoint in the assessment of psoriasis. J Am
Acad Dermatol. 2004;50:859–66.
17- Chaudhari U,RomanoP, Mulcahy LD etal, Efficacy and safety of infliximab for plaque type
psoriasis: a randomised trial. 2001; 357: 1842-47.
18- Reich K, Nestle FO, Papp K, Ortonne JP, Evans R, Guzzo C, et al. Infliximab induction and
maintenance therapy for moderate-to-severe psoriasis: a phase III, multicentre, double-blind trial.
Lancet. 2005;366:1367-74.
19- Kipnis D, Wendy A, Opeola M et al. Biologic treatments for psoriasis. J Am Acad Dermatol
2005;52:671-82.