Solutions-homogeneous mixtures of two or more pure ... · Solutions-homogeneous mixtures of two or...
Transcript of Solutions-homogeneous mixtures of two or more pure ... · Solutions-homogeneous mixtures of two or...

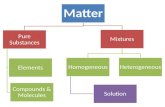
Solutions-homogeneous mixtures of two or more pure substances
2 Parts of a Solution
Solute- is dissolved (usually the lesser amount)
Solvent- dissolves the solute (usually the greater amount)
Solute Solvent Example
solid solid Alloys (brass, steel)
solid liquid Salt water
gas solid Air bubbles in ice cubes
liquid liquid “suicides” (mixed drinks)
gas liquid Soft drinks
gas gas Air
The solvent pulls solute particles apart and surrounds or solvates
them

Solubility- if a compound is soluble it must overcome attraction
forces between:
1. Solvent molecules
2. Solute molecules
3. New interactions between solute and solvent
Lattice energy- the energy
required to completely
separate a mole of a solid
ionic compound into its
gaseous ions.
If an ionic salt is soluble in water, it is because the ion-dipole
interactions are strong enough to overcome the lattice energy of
the salt crystal.
The energy change of the overall process depends on H for each
of these steps.
NaCH3CO2 (s) + heat → Na+(aq) + CH3CO2-(aq)
NaCH3CO2 (s) + heat → Na+(aq) + CH3CO2-(aq)

WARNING! Just because a substance disappears when it
comes in contact with a solvent, it doesn’t mean the substance
dissolved.
• Dissolution is a physical change—you can get back the original
solute by evaporating the solvent.
• If you can’t, the substance didn’t dissolve, it reacted.
Factors Affecting Solubility
“like dissolves like”
• In general, the solubility of gases in water increases with
increasing mass.
• Larger molecules have stronger dispersion forces.

• The solubility of liquids and solids does not change appreciably
with pressure.
• The solubility of a gas in a liquid is directly proportional to its
pressure.
Sg = kPg
Sg = the solubility of the gas;
k = Henry’s law constant for that gas in that solvent;
Pg = the partial pressure of the gas above the liquid.
Generally, the solubility
of solid solutes in liquid
solvents increases with
increasing temperature.
Generally, the solubility
of solid solutes in liquid
solvents increases with
increasing temperature.
Warm tea vs ice tea
Generally, the solubility
of gas solutes in liquid
solvents decreases with
decreasing temperature.
Ex. Carbonated soft drinks are more
“bubbly” if stored in the refrigerator.
Warm lakes have less O2 dissolved in
them than cool lakes.

Types of Solutions
Saturated
Solvent holds as much solute as is possible at that
temperature.
Dissolved solute is in dynamic equilibrium with solid
solute particles.
Unsaturated
Less than the maximum amount of solute for that
temperature is dissolved in the solvent.
Supersaturated
Solvent holds more solute than is normally possible at that
temperature.
These solutions are unstable; crystallization can usually be
stimulated by adding a “seed crystal” or scratching the side of
the flask.

An IDEAL SOLUTION is one where the properties depend only on
the concentration of solute.
Need conc. units to tell us the number of solute particles per
solvent particle.
The unit “molarity” does not do this! 𝑀 = 𝑚𝑜𝑙 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑒
𝐿 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑖𝑜𝑛
Mole Fraction, X 𝑋𝐴 =𝑚𝑜𝑙 𝐴
𝑚𝑜𝑙 𝐴 + 𝑚𝑜𝑙 𝐵
Molality, m 𝑚 𝑜𝑓 𝑠𝑜𝑙𝑢𝑡𝑒 =𝑚𝑜𝑙 𝑠𝑜𝑙𝑢𝑡𝑒
𝑘𝑖𝑙𝑜𝑔𝑟𝑎𝑚𝑠 𝑠𝑜𝑙𝑣𝑒𝑛𝑡
Weight % 𝑚𝑎𝑠𝑠 % 𝑜𝑓 𝐴 = 𝑚𝑎𝑠𝑠 𝑜𝑓 𝐴
𝑡𝑜𝑡𝑎𝑙 𝑚𝑎𝑠𝑠 𝑥 100
Colligative Properties-depend only on the NUMBER of solute
particles relative to solvent particles, not on the KIND of solute
particles.