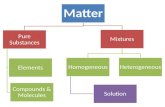
Solutions Chapter 12. Vocabulary Solution: a homogeneous mixture of two or more substances in a...
-
Upload
lenard-wells -
Category
Documents
-
view
221 -
download
1
Transcript of Solutions Chapter 12. Vocabulary Solution: a homogeneous mixture of two or more substances in a...

Solutions
Chapter 12

Vocabulary
• Solution: a homogeneous mixture of two or more substances in a single phase
• Solvent: the dissolving medium in a solution
• Solute: the substance dissolved in a solution
• Solution can be liquid or solid or gaseous

liquid-liquid
solid-liquid
solid-solid

gas-liquid

Factors Affecting the Rate of Dissolution
• Surface area: greater surface area speeds up the rate
• Agitation: shaking or stirring speeds up the rate
• Temperature: higher temperature usually favor a higher rate, except in the case of most gases, then a lower temperature speeds up the rate


Like Dissolves Like
• Polar solutes dissolve in polar solvents• Nonpolar solutes dissolve in nonpolar
solvents• Molecules can have a polar end and
nonpolar end (especially if they are large)• This is why soap will dissolve grease and
then be washed away in water

Concentration of Solutions
• Concentration: a measure of the amount of solute in a given amount of solvent or solution
• Molarity: the number of moles of solute in one liter of solution
• Molarity (M) = amount of solute (mol) volume of solution (L)

Molarity
• One mole of NaOH has a mass of 39.998 g.• If this mass is dissolved in enough water to
make exactly 1.00 L of solution, the solution is a 1 M solution.
• If 20.0 g of NaOH is dissolved in enough water to make 1.00 L of solution, what is the molarity? 0.500 M NaOH

• Notice that 1 mol of solute added to 1 L of solvent does not usually make 1 M solution
• Adding the solute to 1 L will cause the volume to increase

The preparation of a 0.0128 M solution of CuSO4 5H2O
Start by calculating the mass needed. Convert the mol to mass by multiplying by the molar mass. This mass is 3.20 g.


The resulting solution has. 0.0128 mol of solute dissolved in 1.000 L of solution, which is a 0.0128 M concentration

Problem
• You have 3.50 L of solution that contains 90.0 g of NaCl. What is the molarity of the solution?
• Given : solute mass of 90.0 g NaCl and solution volume of 3.50 L
• First change g to mol• 90.0 g NaCl = 1.54 mol NaCl

Continued
• Divide mol of solute by liters of solution• 1.54 mol NaCl 3.50 L solution• 0.440 M NaCl

Problem
• You have 0.8 L of a 0.5 M HCl solution. How many moles of HCl does this solution contain?
• M = mol/L so mol = M L• 0.5 M 0.8 L = 0.4 mol HCl• How many grams is this? (use 3 sig figs)• 14.6 g HCl

Problem
• If you have 61.9 mL of a 2M solution of HCl, how many moles do you have?
• Convert 61.9 mL to liters by dividing by 1000.
• 0.0619 L• mol = M L• mol = 0.1238 = 0.124 mol HCl

Dilutions
• Concentrated solutions are purchased in standard molarities called stock solutions
• You can prepare a less concentrated solution by diluting the stock solution with solvent (usually water)
• The number of moles of solute does not change when you dilute

Making Dilutions
• The molarity times the volume of the stock solution equals the molarity times the volume of the dilution
• M1V1 = M2V2
• Where M1V1 is the molarity and volume of the stock solution and M2V2 is the molarity and volume of the dilution

Problem
• What volume, in mL of 2.00 M calcium chloride stock solution would you use to make 0.50 L of 0.300 M calcium chloride solution?
• Write down what you are given• M1 = 2.00 M CaCl2 V1 = ?
• M2 = 0.300 M V2 = 0.50 L

Continued• (2.00 M)(x) = (0.300)(0.50)• 2.00x = 0.15• x = 0.075 L• Since the answer has to be in mL:• (0.075)(1000)• 75 mL of the stock solution must be
measured and then water added to make 0.50 L

Problems• What volume of a 3.00 M KI stock
solution would you use to make 0.300 L of a 1.25 M KI solution?
• How many mL of a 5.0 M H2SO4 stock solution would you need to prepare 100.0 mL of a 0.25 M H2SO4?
• If you dilute 20.0 mL of a 3.5 M solution to make 100.0 mL of solution, what is the molarity of the dilute solution?
0.125 L or 125 mL
5.0 mL
0.70 M