Section 4. Fuel oxidation, generation of ATP Fig.iv.1 Section 4. Overview of Fuel oxidation, ATP...
-
Upload
vivien-annabella-charles -
Category
Documents
-
view
219 -
download
0
Transcript of Section 4. Fuel oxidation, generation of ATP Fig.iv.1 Section 4. Overview of Fuel oxidation, ATP...

Section 4. Fuel oxidation, generation of ATP
Fig.iv.1
Section 4. Overview of Fuel oxidation, ATP generation:
Physiological processes require energy transfer from chemical bonds in food:
• Electrochemical gradient• Movement of muscle• Biosynthesis of complex molecules
3 phases:• Oxidation of fuels (carbs, fats, protein)• Conversion of energy to ~PO4 of ATP• Utilization of ATP to drive energy-requiring reactions

Fuel oxidation overview - respiration
Phase 1: energy (e-) from fuel transfer to NAD+ and FAD;Acetyl CoA, TCA intermediates are central compounds
Phase 2: electron transport chain convert e- to ATP;membrane proton gradient drives ATP synthase
Phase 3: ATP powers processes
Fig. iv.2

Respiration occurs in mitochondria
Fig. iv.3
Respiration occurs in mitochondria:
• Most enzymes in matrix
• Inner surface has• e- transport chain• ATP synthase
• ATP transported through inner membrane, diffuses through outer
• Some enzymes encoded by mitochondrion genome,
• most by nuclear genes

Glucose is universal fuel for every cell
Fig. iv.4
Glycolysis is universal fuel:1 glucose -> 2 pyruvate + 2 NADH + 2 ATP
• Aerobic path:• Continued oxidation• Acetyl CoA -> TCA, • NADH, FAD(2H) -> e- transport chain• Lots of ATP
• Anaerobic: fermentation:• ‘anaerobic glycolysis’• Oxidation of NADH to NAD+• Wasteful reduction of pyruvate
• to lactate in muscles• to ethanol, CO2 by yeast

Chapt. 19 Cellular bioenergetics of ATP, O2
Ch. 19 Cellular bioenergeticsStudent Learning Outcomes:
• Explain the ATP-ADP cycle• Describe how chemical bond energy of fuels can do
cellular work through ~PO4 bond of ATP• Explain how NADH, FAD(2H) coenzymes carry
electrons to electron transport chain
• Describe how ATP synthesis is endergonic (requires energy)
• Describe how ATP hydrolysis (exergonic) powers biosynthesis, movement, transport

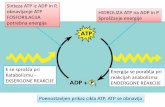
Fuel oxidation makes ATP
Fig. 19.1
Cellular Bioenergetics of ATP and O2:
• Chemical bond energy of fuels transforms to physiological responses necessary for life
• Fuel oxidation generates ATP• ATP hydrolysis provides energy for most work
• High energy bonds of ATP:• Energy currency of cell

ATP
High energy phosphate bond of ATP:
• Strained phosphoanhydride bond• G0’ -7.3 kcal/mol standard conditions
• Hydrolysis of ATP to ADP + Pi transfers PO4 to metabolic intermediate or protein, for next step
Fig. 19.2

Thermodynamics brief
Thermodynamics states what is possible:
G = change in Gibbs free energy of reaction: G = G0 + RT ln [P]/[S] (R = gas const; T = temp oK)
GG at standard conditions of1 M substrate & product and proceeding to equilibrium)
G0’ = G0 under standard conditions of [H2O] = 55.5 M, pH 7.0, and 25oC [37oC not much different]
Concentrations of substrate(s) and products(s):At equilibrium, G = 0, therefore
G0’ = -RT lnKeq’ = -RT ln[P]/[S]

Thermodynamics brief
Thermodynamics states what is possible:
• Exergonic reactions give off energy (G0’ < 0)• typically catabolic
• Endergonic reactions require energy (G0’ > 0)• typically anabolic
• Unfavorable reactions are coupled to favorable
reactions • Hydrolysis of ATP is very favorable• Additive G0’ values determine overall direction

C. Exogonic, endogonic reactions
Phosphoglucomutase converts G6P to/from G1P:• G6P to glycolysis• G1P to glycogen synthesis• Equilibrium favors G6P
Exergonic reactions give off energy (DG0’ < 0)Endergonic reactions require energy (DG0’ > 0)
Fig. 19.3

III. Energy transformation for mechanical work
ATP hydrolysis can power muscle movement:• Myosin ATPase hydrolyzes ATP, changes shape
• ADP form changes shape back, moves along• Actin was activated by Ca2+
Fig. 19.4

ATP powers transport
Active transport: ATP hydrolysis moves molecules:
• Na+, K+ ATPase sets up ion gradient; bring in items• Vesicle ATPases pump protons into lysosome• Ca2+-ATPases pump Ca2+ into ER, out of cell
Fig. 10.6

III. ATP powers biochemical work
ATP powers biochemical work, synthesis:
Anabolic paths require energy: Go’ additive• Couple synthesis to ATP hydrolysis:
• Phosphoryl transfer reactions• Activated intermediate
Ex. Table 19.3: glucose + Pi -> glucose 6-P + H2O + 3.3 kcal/mol ATP + H2O -> ADP + Pi - 7.3 kcal/molSum: glucose + ATP -> glucose 6-P + ADP -4.0 Also Glucose -> G-1-P will be -2.35 kcal/mol overall:
hydrolysis of ATP, through G-6-P to G-1-P

Activated intermediates in glycogen synthesis
Glycogen synthesis needs 3 ~P:
• Phosphoryl transfer to G6P
• Activated intermediate with UDP covalently linked
Fig. 19.5
Fig. 19.6

G depends on substrate, product concentrations
G depends on substrate, product concentrationsG = G0 + RT ln [P]/[S]
• Cells do not have 1M concentrations• High substrate can drive reactions with positive G0’• Low product (removal) can drive reactions with positive G0’
• Ex., even though equilibrium (G0’= +1.6 kcal/mol)favors G6P: G1P in a ratio 94/6,
• If G1P is being removed (as glycogen synthesis), then equilibrium shifts
ex. If ratio 94/3, then G = -0.41 favorable

Activated intermediates with ~bonds
Other compounds have high-energy bonds to aid biochemical work: (equivalent to ATP)
• UTP, CTP and GTP also (made from ATP + NDP):• UTP for sugar biosyn, GTP for protein, CTP for lipids
• Some other compounds:• Creatine PO4 energy reserve muscle, nerve, sperm• Glycolysis• Ac CoA TCA cycle
Fig. 19.7

V. Energy from fuel oxidation
Fig. 19.8
Energy transfer from fuels through oxidative phosphorylation in mitochondrion:
• NADH, FAD(2H) transfer e- to O2
• Stepwise process through protein carriers• Proton gradient created• e- to O2 -> H2O• ATP synthase makes ATP
• lets in H+

Oxidation/reduction
Fig. 19.9 NADH
Fig. 19.10FAD(2H)
Oxidation: reduction reactions:• Electron donor gets oxidized; recipient is reduced• LEO GER:
•Loss Electrons = oxidation; gain electrons is reduction• use coenzyme e- carriers

Redox potentials
Redox potentials indicate energetic possibility:
Energy tower; combine half reactions for overall:
Ex. Table 19.4:
½ O2 + 2H+ + 2e- -> H2O E0’ 0.816
NAD+ + 2H+ + 2e- -> NADH + H+ -0.320
Combine both reactions (turn NADH -> NAD+) = 0.320
Total 1.136 (very big) = -53 kcal/mol
FAD(2H) gives less, since its only +0.20 (FAD(2H) -> FAD

Calorie content of fuels reflects oxidation state
Calorie content of fuels reflects oxidation state:
• C-H and C-C bonds will be oxidized:
• Glucose has many C-OH already:• 4 kcal/g
• Fatty acids very reduced: 9 kcal/g
• Cholesterol no calories: not oxidized in reactions giving NADH

Anaerobic glycolysis” = fermentation
‘Anaerobic glycolysis’ = fermentationIn absence of O2, cell does wasteful recycling:
• NADH oxidized to NAD+ (lose potential ATP)• pyruvate reduced to lactate• glycolysis can continue with new NAD+
• yeast makes ethanol, CO2 from pyruvate
• bacteria make diverse acids, other products
Fig. 19.11

Oxidation not for ATP generation
Fig. 19.12
Most O2 used in electron transport chain.Some enzymes use O2 for substrate oxidation, not for ATP generation:
• Oxidases transfer e- to O2
• [Cytochrome oxidase in
electron transport chain] Peroxidases in peroxisome
• Oxygenases transfer e- and O2 to substrate
• Form H2O and S-OH• Hydroxylases
• (eg. Phe -> Tyr)

VII Energy balance
Fig. 19.14
Energy expenditure reflects oxygen consumption:
• Most O2 is used
by ATPases

Energy balance
Portion of food metabolized is related to energy use:
• Basal metabolic rate
• Thermogenesis
• Physical activity
• Storage of excess
“If you eat to much and don’t exercise, you will get fat” (summarizes ATP-ADP cycle)