Overview of dna replication (prokaryotic & eukaryotic)
-
Upload
magendiramani-vinayagam -
Category
Education
-
view
199 -
download
2
Transcript of Overview of dna replication (prokaryotic & eukaryotic)

DNA Replication Overview
Compiled by
V. Magendira ManiAssistant Professor, PG & Research Department of Biochemistry,Islamiah College (Autonomous),Vaniyambadi,Vellore District – 6357512,Tamilnadu, [email protected]
https://tvuni.academia.edu/mvinayagam

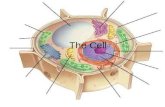
Structure of DNADNA, or deoxyribonucleic acid, is the hereditary
material in humans and almost all other
organisms. Nearly every cell in a person’s body
has the same DNA. Most DNA is located in the
cell nucleus (where it is called nuclear DNA), but
a small amount of DNA can also be found in the
mitochondria (where it is called mitochondrial
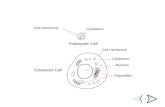
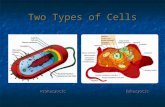
DNA or mtDNA). DNA is found in the nucleus of
eukaryotes and the cytoplasm or nucleoid of
prokaryotes and functions as the molecule of
heredity.
In DNA there are four bases: adenine
(abbreviated A), guanine (G), thymine (T) and
cytosine (C). Adenine and guanine are purines;
thymine and cytosine are pyrimidines.

A nucleoside is a pyrimidine or purine base
covalently bonded to a sugar. In DNA, the sugar is
deoxyribose and so this is a deoxynucleoside.
There are four types of deoxynucleoside in DNA;
deoxyadenosine, deoxyguanosine, deoxythymidine
and deoxycytidine.
A nucleotide is base + sugar + phosphate
covalently bonded together. In DNA, where the
sugar is deoxyribose, this unit is a
deoxynucleotide.
In DNA the nucleotides are covalently joined
together by 3’-- 5’ phosphodiester bonds to form a
repetitive sugar–phosphate chain which is the
backbone to which the bases are attached.

The DNA sequence is the sequence of A, C, G and T
along the DNA molecule which carries the genetic
information.
In a DNA double helix, the two strands of DNA are
wound round each other with the bases on the inside
and the sugar–phosphate backbones on the outside.
The two DNA chains are held together by hydrogen
bonds between pairs of bases; adenine (A) always
pairs with thymine (T) and guanine (G) always pairs
with cytosine (C).
Each nucleotide consists of three major parts: (1) a
five-carbon sugar (pentose); (2) a flat, heterocyclic,
nitrogen-containing organic base; and (3) a negatively
charged phosphate group, which gives the polymer its
acidic property. The nitrogenous base in each
nucleotide is covalently attached to the sugar by a
glycosidic bond. The phosphate group is also
covalently linked to the sugar.

Bases in the nucleotides spontaneously form hydrogen
bonds in a highly specific manner. Adenine normally
forms two hydrogen bonds with thymine in a
complementary strand of the DNA double helix like,
Guanine forms three hydrogen bonds with cytosine.
Human DNA consists of about 3 billion bases, and more
than 99 percent of those bases are the same in all
people.
Nucleotides are arranged in two long strands that form
a spiral called a double helix. The structure of the
double helix is somewhat like a ladder, with the base
pairs forming the ladder’s rungs and the sugar and
phosphate molecules forming the vertical sidepieces of
the ladder.

An important property of DNA is that it can
replicate, or make copies of itself. Each strand
of DNA in the double helix can serve as a
pattern for duplicating the sequence of bases.
This is critical when cells divide because each
new cell needs to have an exact copy of the
DNA present in the old cell.
Watson and Crick model of DNA
DNA is a double stranded helix, with the two
strands connected by hydrogen bonds. Adenine
bases are always paired with thymine, and
cytosine is always paired with guanine, which is
consistent with and accounts for Chargaff's
rule. This is called complementary base pairing.
Watson and Crick discovered that DNA had two
sides, or strands, and that these strands were
twisted together like a twisted ladder the
double helix.

Most DNA double helices are right handed, only
one type of DNA, called ZDNA, is left handed.
The G:C and A:T base pairing also maximizes the
number of effective hydrogen bonds that can form
between the bases; there are three hydrogen
bonds between each G:C base pair and two
hydrogen bonds between each A:T base pair. Thus
A:T and G:C base pairs form the most stable
conformation both from steric considerations and
from the point of view of maximizing hydrogen
bond formation.
The sides of the ladder comprise the sugar
phosphate portions of adjacent nucleotides bonded
together. The phosphate of one nucleotide is
covalently bound to the sugar of the next
nucleotide.

The hydrogen bonds between phosphates cause the DNA
strand to twist.

The DNA double helix is antiparallel, which means
that the 5' end of one strand is paired with the 3'
end of its complementary strand (and vice versa).
As shown in Figure, nucleotides are linked to each
other by their phosphate groups, which bind the 3'
end of one sugar to the 5' end of the next sugar.

Along the whole length of the DNA molecule,
there are two depressions—referred to as the
“minor groove” and the “major groove”—that lie
between the strands.
In a DNA molecule, the different nucleotides are
covalently joined to form a long polymer chain by
covalent bonding between the phosphates and
sugars.

For any one nucleotide, the phosphate attached
to the hydroxyl group at the 5’ position of the
sugar is in turn bonded to the hydroxyl group on
the 3’ carbon of the sugar of the next nucleotide.
Since each phosphate–hydroxyl bond is an ester
bond, the linkage between the two
deoxynucleotides is a 3’5’ phosphodiester bond.
Thus, in a DNA chain, all of the 3’ and 5’ hydroxyl
groups are involved in phosphodiester bonds
except for the first and the last nucleotide in the
chain. The first nucleotide has a 5’ phosphate not
bonded to any other nucleotide and the last
nucleotide has a free 3’ hydroxyl. Thus each DNA
chain has polarity; it has a 5’ end and a 3’ end.

Erwin Chargaff was one of a handful of
scientists who expanded on Levene's work by
uncovering additional details of the structure
of DNA, thus further paving the way for
Watson and Crick.

A, B and Z forms of DNA
The Watson-Crick structure is also referred to as
B form DNA, or B-DNA. The B form is the most
stable structure for a random-sequence DNA
molecule under physiological conditions and is
therefore the standard point of reference in any
study of the properties of DNA. Two structural
variants that have been well characterized in
crystal structures are the A and Z forms. These
three DNA conformations are shown in Figure,
with a summary of their properties.

The A form is favored in many solutions that are
relatively devoid of water. The DNA is still
arranged in a right-handed double helix, but the
helix is wider and the number of base pairs per
helical turn is 11, rather than 10.5 as in B-DNA.
The plane of the base pairs in A-DNA is tilted
about 20with respect to the helix axis. These
structural changes deepen the major groove while
making the minor groove shallower. The reagents
used to promote crystallization of DNA tend to
dehydrate it, and thus most short DNA molecules
tend to crystallize in the A form.

Z-form DNA is a more radical departure from the B
structure; the most obvious distinction is the left handed
helical rotation. There are 12 base pairs per helical turn,
and the structure appears more slender and elongated.
The DNA backbone takes on a zigzag appearance.
Certain nucleotide sequences fold into left handed Z
helices much more readily than others. Prominent
examples are sequences in which pyrimidines alternate
with purines, especially alternating C and G or 5-methyl-
C and G residues. To form the left-handed helix in Z-
DNA, the purine residues flip to the syn conformation,
alternating with pyrimidines in the anti-conformation.
The major groove is barely apparent inZ-DNA, and the
minor groove is narrow and deep. Whether A-DNA
occurs in cells is uncertain, but there is evidence for
some short stretches (tracts) of Z-DNA in both
prokaryotes and eukaryotes. These Z-DNA tracts may
play a role (as yet undefined) in regulating the
expression of some genes or in genetic recombination.

Chargaff Rules The nucleotide composition of DNA varies
among species
The amount of adenine (A) is usually
similar to the amount of thymine (T), and
the amount of guanine (G) usually
approximates the amount of cytosine (C).
The total amount of purines (A + G) and
the total amount of pyrimidines (C + T) are
usually nearly equal.

DNA “semi-conservative”
Replication
DNA replication is the process by which
the genetic material is copied prior to
distrubution into daughter cells
The original DNA strands are used as
templates for the synthesis of new strands
It occurs very quickly, very accurately and at
the appropriate time in the life cycle of the
cell
DNA replication relies on the
complementarity of DNA strands
The AT/GC rule or Chargaff’s rule

In the semi-conservative model, the two
parental strands separate and each makes a copy
of itself. After one round of replication, the two
daughter molecules each comprise one old and
one new strand. Note that after two rounds, two
of the DNA molecules consist only of new
material, while the other two contain one old and
one new strand.
The semi-conservative model is the spontaneously
appealing model, because separation of the two
strands provides two templates, each of which
carries all the information of the original
molecule. It also turns out to be the correct one
(Meselson & Stahl, 1958).

The process can be summarized as follows:
The two DNA strands in the parent DNA
molecule come apart
Each “parent strand” then serves as a
template for the synthesis of a new
complementary strand
The two newly-made strands =
daughter strands
The two original ones = parental
strands

This process is called semi-conservative because it
conserves only half of the original (parent) DNA
molecule in the two daughter DNA molecules. One
strand in each daughter molecule is completely
new.
In the late 1950s, three different mechanisms were
proposed for the replication of DNA
Conservative model
• Both parental strands stay together after
DNA replication and one of the daughter
molecules contains all new nucleotides
Semiconservative model
• The double-stranded DNA contains one
parental and one daughter strand following
replication
Dispersive model
• Parental and daughter DNA are interspersed
in both strands following replication

In 1958, Matthew Meselson and Franklin
Stahl devised a method to investigate these
models.
Their experiment can be summarized as follows:
Grow E. coli in the presence of 15N (a heavy
isotope of nitrogen) for many generations
The population of cells now has heavy-labeled
DNA because the DNA bases are rich in
nitrogen
Switch E. coli to medium containing only 14N
(a light isotope of nitrogen)
Collect sample of cells after various times
Analyze the density of the DNA by
centrifugation using a CsCl gradient

The actual data from the Mesleson-Stahl
experiment is shown below.
After one generation, DNA is “half-heavy”
After ~ two generations, DNA is of two types: “light” and “half-heavy”
This is consistent with only the semi-conservative model

The Meselson–Stahl experiment was an experiment
by Matthew Meselson and Franklin Stahl in
1958 which supported the hypothesis that DNA
replication was semiconservative. Meselson and
Stahl decided the best way to tag the parent DNA
would be to change one of the atoms in the parent
DNA molecule. Remember that nitrogen is found in
the nitrogenous bases of each nucleotide. So they
decided to use an isotope of nitrogen to distinguish
between parent and newly-copied DNA. The isotope
of nitrogen had an extra neutron in the nucleus,
which made it heavier.
Three hypotheses had been previously proposed for
the method of replication of DNA.
In the semiconservative hypothesis, proposed by
Watson and Crick, the two strands of a DNA
molecule separate during replication. Each strand
then acts as a template for synthesis of a new
strand.

The conservative hypothesis proposed that the
entire DNA molecule acted as a template for the
synthesis of an entirely new one. In the
conservative model, the parental molecule directs
synthesis of an entirely new double-stranded
molecule, such that after one round of
replication, one molecule is conserved as two old
strands. This is repeated in the second round.
The dispersive hypothesis is exemplified by a
model proposed by Max Delbrück, which
attempts to solve the problem of unwinding the
two strands of the double helix by a mechanism
that breaks the DNA backbone every 10
nucleotides or so, untwists the molecule, and
attaches the old strand to the end of the newly
synthesized one. This would synthesize the DNA
in short pieces alternating from one strand to the
other.

Each of these three models makes a different
prediction about the distribution of the "old"
DNA in molecules formed after replication. In
the conservative hypothesis, after replication,
one molecule is the entirely conserved "old"
molecule, and the other is all newly
synthesized DNA. The semiconservative
hypothesis predicts that each molecule after
replication will contain one old and one new
strand. The dispersive model predicts that
each strand of each new molecule will contain
a mixture of old and new DNA.

Experimental procedure and
results Matthew Meselson and Franklin Stahl were well
acquainted with these three predictions, and
they reasoned that if there were a way to
distinguish old versus new DNA, it should be
possible to test each prediction. Aware of
previous studies that had relied on isotope labels
as a way to differentiate between parental and
progeny molecules, the scientists decided to see
whether the same technique could be used to
differentiate between parental and progeny
DNA. If it could, Meselson and Stahl were
hopeful that they would be able to determine
which prediction and replication model was
correct.

Meselson & Stahl began their experiment by
choosing two isotopes of nitrogen -the common
and lighter 14N, and the rare and heavier 15N (so-
called "heavy" nitrogen) - as their labels and a
technique known as cesium chloride (CsCl)
equilibrium density gradient centrifugation as
their sedimentation method. Meselson and Stahl
opted for nitrogen because it is an essential
chemical component of DNA; therefore, every time
a cell divides and its DNA replicates, it
incorporates new N atoms into the DNA of either
one or both of its two daughter cells, depending on
which model was correct. "If several different
density species of DNA are present," they
predicted, "each will form a band at the position
where the density of the CsCl solution is equal to
the buoyant density of that species. In this way,
DNA labeled with heavy nitrogen (15N) may be
resolved from unlabeled DNA" (Meselson & Stahl,
1958).


The scientists then continued their experiment
by growing a culture of E. coli bacteria in a
medium that had the heavier 15N (in the form
of 15N-labeled ammonium chloride) as its only
source of nitrogen. In fact, they did this for 14
bacterial generations, which was long enough
to create a population of bacterial cells that
contained only the heavier isotope (all the
original 14N-containing cells had died by then).
Next, they changed the medium to one
containing only 14N-labeled ammonium salts as
the sole nitrogen source. So, from that point
onward, every new strand of DNA would be
built with 14N rather than 15N.

Just prior to the addition of 14N and periodically
thereafter, as the bacterial cells grew and
replicated, Meselson and Stahl sampled DNA for
use in equilibrium density gradient
centrifugation to determine how much 15N (from
the original or old DNA) versus 14N (from the
new DNA) was present. For the centrifugation
procedure, they mixed the DNA samples with a
solution of cesium chloride and then centrifuged
the samples for enough time to allow the
heavier 15N and lighter 14N DNA to migrate to
different positions in the centrifuge tube.

By way of centrifugation, the scientists found that
DNA composed entirely of 15N -labeled DNA (i.e.,
DNA collected just prior to changing the culture
from one containing only 15N to one containing
only 14N) formed a single distinct band, because
both of its strands were made entirely in the
"heavy" nitrogen medium. Following a single
round of replication, the DNA again formed a
single distinct band, but the band was located in a
different position along the centrifugation
gradient. Specifically, it was found midway
between where all the 15N and the entire 14N DNA
would have migrated-in other words, halfway
between "heavy" and "light".

Based on these findings, the scientists were
immediately able to exclude the
conservative model of replication as a
possibility. After all, if DNA replicated
conservatively, there should have been two
distinct bands after a single round of
replication; half of the new DNA would have
migrated to the same position as it did
before the culture was transferred to the
14N-containing medium (i.e., to the "heavy"
position), and only the other half would have
migrated to the new position (i.e., to the
"light" position). That left the scientists with
only two options: either DNA replicated
semi-conservatively, as Watson and Crick
had predicted, or it replicated dispersively.

To differentiate between the two, Meselson and Stahl
had to let the cells divide again and then sample the
DNA after a second round of replication. After that
second round of replication, the scientists found that
the DNA separated into two distinct bands: one in a
position where DNA containing only 14N would be
expected to migrate, and the other in a position
where hybrid DNA (containing half 14N and half 15N)
would be expected to migrate. The scientists
continued to observe the same two bands after
several subsequent rounds of replication. These
results were consistent with the semiconservative
model of replication and the reality that, when DNA
replicated, each new double helix was built with one
old strand and one new strand. If the dispersive
model were the correct model, the scientists would
have continued to observe only a single band after
every round of replication.

Rolling circle replication Rolling circle replication describes a process of
unidirectional nucleic acid replication that can
rapidly synthesize multiple copies of circular
molecules of DNA or RNA, such as plasmids,
the genomes of bacteriophages, and
the circular RNA genome of viroids. Some
eukaryotic viruses also replicate their DNA via
a rolling circle mechanism.

Rolling circle DNA replication is initiated by an
initiator protein encoded by the plasmid or
bacteriophage DNA, which nicks one strand of
the double-stranded, circular DNA molecule at a
site called the double-strand origin, or DSO. The
initiator protein remains bound to the 5'
phosphate end of the nicked strand, and the free
3' hydroxyl end is released to serve as a primer
for DNA synthesis by DNA polymerase III. Using
the un-nicked strand as a template, replication
proceeds around the circular DNA molecule,
displacing the nicked strand as single-stranded
DNA. Displacement of the nicked strand is
carried out by a host-encoded helicase called
PcrA (the abbreviation standing for plasmid copy
reduced) in the presence of the plasmid
replication initiation protein.

Continued DNA synthesis can produce multiple
single-stranded linear copies of the original DNA in
a continuous head-to-tail series called a concatemer.
These linear copies can be converted to double-
stranded circular molecules through the following
process:
First, the initiator protein makes another nick to
terminate synthesis of the first (leading) strand.
RNA polymerase and DNA polymerase III then
replicate the single-stranded origin (SSO) DNA to
make another double-stranded circle. DNA
polymerase I removes the primer, replacing it with
DNA, and DNA ligase joins the ends to make
another molecule of double-stranded circular DNA.

Rolling circle replication has found wide uses in
academic research and biotechnology, and has
been successfully used for amplification of DNA
from very small amounts of starting material.
Some viruses replicate their DNA in host cells
via rolling circle replication. For instance,
human herpesvirus-6 (HHV-6) (hibv) expresses a
set of “early genes” that are believed to be
involved in this process. The long concatemers
that result are subsequently cleaved between
the pac-1 and pac-2 regions of HHV-6's genome
by ribozymes when it is packaged into individual
virions.

John Cairns Experiment
Cairns grew E.Coli bacteria in a medium
containing radioactive thymine, a component of
one of the DNA nucleotides. The radioactivity
was in tritium (31H). The DNA was then carefully
extracted from the bacteria and placed on
photographic emulsion for a period of time. The
emulsion was then developed to produce
autoradiograph that was examined under the
electron microscope. Each grain of silver
represents a radioactive decay. Interpretation of
this autoradiograph reveals several points.

The first, known at the time, is that the E.Coli DNA is a circle. The Second point is that DNA is replicated while maintaining the integrity of the circle i.e., the circle does not appear to be broken in the process of DNA replication; an intermediate theta structure is formed which is due to the formation of replication eye. Third, replication of the DNA seems to be occurring at one or two moving Y-junctions in the circle Replication forks, which further supports the Semiconservative replication.

DNA
REPLICATION
IN
PROKARYOTES

DNA Replication in Bacteria
(E.Coli) DNA synthesis begins at a site termed the origin
of replication (“Ori -C”) Each bacterial chromosome has only one ori- C Synthesis of DNA proceeds bi-directionally
around the bacterial chromosome The “replication forks” eventually meet at the
opposite side of the bacterial chromosome This ends replication
Bacterial DNA replication has been studied most
extensively in E. coli, the favorite bacterial
“model organism” of molecular geneticists.
The ORI in E. coli is called “oriC” Three types of DNA sequences in oriC are
functionally significant AT-rich region DnaA boxes GATC methylation sites


DNA replication is initiated by the binding of
DnaA proteins to the DnaA box sequences
• This binding stimulates the cooperative
binding of an additional 20 to 40 DnaA
proteins to form a large complex.
• This causes the DNA to twist and the puts
torque on the nearby AT-rich region to
denature and form a replication bubble
AT base pairs are held together by only 2
H bonds
CG base pairs are held together by 3 H
bonds
Therefore, AT-rich regions of DNA
denature more easily than CG-rich regions
of DNA

In the next step, DnaB (also called helicase)
binds to each strand of the separated double
helix. It’s job is to move along the DNA,
progressively expand the replication bubble in
both directions.Travels along the DNA
strand in the 5’ to 3’
direction, using energy
from ATP
As the helicases move on each strand in opposite
directions, two replication forks are created. These
forks move progressively farther and farther in each
direction as the bubble widens.


DNA helicase separates the two DNA strands by
breaking the hydrogen bonds between them
This generates positive supercoiling ahead of
each replication fork so another enzyme,
topoisomerase, travels ahead of the helicase
and alleviates these supercoils
Single-strand binding proteins (SSBPs) are also
needed to bind to the separated DNA strands and
keep them apart
Otherwise, the strands would simply reanneal
After the helicase, gyrase, and SSBPs are in place,
short (10 to 12 nucleotides) RNA primers are
synthesized by DNA primase
These short RNA strands start, or prime, DNA
synthesis because DNA polymerase, the enzyme
that copies DNA, cannot start a new strand on its
own
The RNA primers are later removed and replaced
with DNA

Keep the parental strands apart
Breaks the hydrogen bonds between the two strands
Alleviates supercoiling
Synthesizes an RNA primer
DNA Polymerases
DNA polymerases are the enzymes that catalyze the
attachment of nucleotides to make new DNA
In E. coli there are five proteins with polymerase
activity
DNA pol I
• Composed of a single polypeptide
• Removes the RNA primers and replaces
them with DNA during DNA replication

DNA pol III
Composed of 10 different subunits
The a subunit synthesizes DNA
The other 9 fulfill other functions
The complex of all 10 is referred to as
the “DNA pol III holoenzyme”
Is responsible for most of the DNA
replication process
DNA pol II, IV and V
Specialized DNA polymerases that
replicate short areas of DNA for the
purposes of genome repair
The numbering of these polymerases was done in
the order they were discovered
Bacterial DNA polymerases may vary in their
subunit composition. However, they have the same
type of catalytic subunit.

Structure resembles a human
right hand:
Thumb and fingers wrapped around the
DNA
Template DNA thread through the palm;
All DNA polymerases, whether bacterial or
eukaryotic, share 2 very important limitations:
1. They cannot initiate DNA synthesis on their
own. They require that an RNA primer be laid
down on the DNA first by DNA primase.

They can only “grow” a new DNA chain in the 5’ to 3’ direction.
It is not fully understood why all DNA
polymerases have these limitations. As will be
demonstrated below, DNA replication would
be much simpler if they did not!
Because DNA polymerase can only synthesize
a new strand 5’ to 3’, the two new daughter
strands are synthesized in different ways:

Leading strand
One RNA primer is made at the origin
DNA pol III attaches nucleotides in a 5’ to 3’
direction as it slides toward the replication fork
Lagging strand
Synthesis is also in the 5’ to 3’ direction
However it occurs away from the replication fork
Many RNA primers are required
DNA pol III uses the RNA primers to synthesize small
DNA fragments (1000 to 2000 nucleotides each)
These are termed Okazaki fragments after their
discoverers
DNA pol I removes the RNA primers and fills the
resulting gap with DNA
After the gap is filled, a covalent bond is still
missing so
DNA ligase must create this bond

Can be synthesized continuously in the 5’ to 3’ direction
Must be synthesized discontinuously to
maintain 5’ to 3’ synthesis
Note that if DNA polymerase was able to
synthesize a new strand in either direction (5’
to 3’ or 3’ to 5’), lagging strand synthesis and
Okasaki fragments would not be needed.
The process can also be visualized in 3-D as
follows:

The Synthesis Reaction
DNA polymerases catalyze a phosphodiester
bond between the innermost phosphate group of
the incoming deoxynucleoside triphosphate and
the
3’-OH of the sugar of the previous
deoxynucleotide.
In the process, the last two phosphates
of the incoming nucleotide are released in the
form of pyrophosphate (PPi)
In E. coli, DNA pol III stays on the DNA
template long enough to polymerize up to 50,000
nucleotides at a rate of ~ 750 nucleotides per
second!

Proofreading
DNA replication exhibits a high degree of
fidelity.
Mistakes during the process are extremely rare
In E. coli, DNA pol III makes only one
mistake per 108 bases
There are several reasons why fidelity is high:
1. Instability of mismatched pairs
Complementary base pairs have much higher
stability than mismatched pairs
This feature only accounts for part of the
fidelity
It has an error rate of 1 per 1,000
nucleotides
2. Configuration of the DNA polymerase active
site
DNA polymerase is unlikely to catalyze bond
formation between mismatched pairs
This induced-fit phenomenon decreases the
error rate to a range of 1 in 100,000 to 1
million

3. Proofreading function of DNA polymeraseDNA polymerases can identify a mismatched nucleotide and remove it from the daughter strandThe enzyme uses its 3’ to 5’ exonuclease activity to remove the incorrect nucleotideIt then changes direction and resumes DNA synthesis in the 5’ to 3’ direction
Termination of Replication in BacteriaDNA replication ends when oppositely advancing forks meet (remember that the chromosome is circular).• DNA replication often results in two intertwined molecules called catenanes• Catenanes and are separated prior to cell division Replicatio
nDecatenization

DNA
REPLICATION
IN
EUKARYOTES

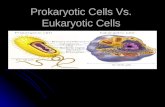
DNA Replication in Eukaryotes Eukaryotic DNA replication is not as well
understood as bacterial replication.
• The two processes do have extensive
similarities,
• Many of the bacterial enzymes described
above have also been found in eukaryotes
Nevertheless, DNA replication in eukaryotes
is more complex due to:
• Large linear chromosomes
• Multiple origins of replication per
chromosome
• Tight packaging of the DNA around
proteins

Linear eukaryotic chromosomes also have
telomeres at both ends
The term telomere refers to the complex of
repetitive DNA sequences found at the terminal
ends of eukaryotic chromosomes as well as the
proteins that recognize this sequence and bind
the DNA there.
Telomeric sequences consist of
• Moderately repetitive tandem arrays
• 3’ overhang that is 12-16 nucleotides long that
results from the loss of the RNA primer at the 5’
end of each strand that cannot be replaced

Therefore if this problem is not solved:
• The linear chromosome becomes
progressively shorter with each round of
DNA replication
• Indeed, some cells solve this problem by
adding DNA sequences to the ends of
telomeres following replication
• This requires a specialized mechanism
catalyzed by the enzyme telomerase
• All single-celled eukaryotes have active
telomerase enzyme because if they
didn’t successive generations of the
organism would have shortened
telomeres
Eventually, this would result in the
loss of important genes and the
death of the species

However, most somatic cells in multicellular
organisms do not express telomerase and the
telomeres shorten every time the cells
replicate.
• Most human somatic cells can only
replicate about 30 times before their
telomeres are so shortened that the
cell dies
This sets an upper limit on the life
span of the organism
Telomerase is active in the germ line cells,
maintaining telomere length from one
generation to the next.
Telomerase is also often abnormally
activated in cancer cells. This is why tumors
don’t eventually replicate themselves to
death. Once a tumor cell has activated
telomerase, it is immortalized.

Immortalized somatic cells are extremely
dangerous in multicellular organisms. If a cell
suffers a mutation in a gene controlling the cell
cycle, the cell can begin to replicate much
faster than the surrounding cells.
Normally, such cells will die off when their
telomeres get too short
Immortalized cells will continue to cycle and
the tissue will grow
The result can be a cancerous tumor that is
life-threatening to the organism

EUKARYOTIC DNA REPLICATION
DNA replication is the process of producing two
identical replicas from one original DNA molecule.
This biological process occurs in all living organisms
and is the basis for biological inheritance. DNA is
made up of two strands and each strand of the original
DNA molecule serves as template for the production
of the complementary strand, a process referred to as
semiconservative replication. The fundamental
mechanism of eukaryotic replication is same as
prokaryotic DNA Replication but some variation also
there.

The replications in eukaryotes are more complex. Because
DNA molecule of eukaryote
Eukaryotic genomes are quite complex
Considerably larger than bacterial DNA
Organized into complex nucleoprotein structure
(chromatin)
Essential features of DNA replication are the same in
prokaryotes and eukaryotes, Similarities of prokaryotes and
eukaryotic replication
Replication process is fundamentally similar in both
prokaryotes and eukaryotes. Process that are similar Include
Formation of replication fork
Simi conservative replication
Movement of replication fork bidirectional
Primer synthesis
Okazaki fragment synthesis in lagging strand
Primer removal
Gap bridging between newly synthesized DNA
fragments.

Difference between prokaryotic and eukaryotic replication
Overall process of eukaryotic replication is bit more complex.
Important differences are due to
• Larger size of eukaryotic DNA (105-106 Kb) compared to
prokaryotic DNA 15x103 kb in E.Coli
• Distinct package of eukaryotic DNA in the term of chromatin
• Slower rate of fork movement in eukaryotes
For DNA to become available to DNA polymerase, nucleotide
must dissemble. This step slows the Rate of fork movement.
Replication rate:
Prokaryotes: An E.Coli replication fork progresses at
approximately 1000 bp / sec.
Eukaryotes: Replication rate ten times slower than prokaryotes
50 nucleotides / sec.

Enzymes and proteins required for eukaryotic DNA
replication
Eukaryotic DNA polymerase:
In eukaryotes there are five different polymerases and they
differ in
Intracellular compartmentation
Kinetic property
Response to inhibitor
DNA pol location functionDNA Pol α nucleus lagging strand
synthesisDNA Pol β nucleus DNA repair
DNA Pol ϒ mitochondria mitochondrial replication
DNA Pol δ nucleus leading strand synthesis
DNA Pol Ɛ nucleus gap filling between okazaki fragments

DNA polymerases location function
DNA Pol alpha nucleusDNA replication initiation (both leading
and lagging strand)
DNA Pol Delta nucleus lagging strand synthesis
DNA Pol Epsilon nucleusleading strand synthesis

DNA polymerase Alpha
- Located in nucleus
- Catalysis the initiation of replication on both
leading and lagging strand synthesis
- Tetramer – 4 subunits POLA 1 (catalytic) POLA 1
(regulatory) POLA 3 ,4 (Primase)
- larger subunit - 5´-3´ polymerization activity
-Two smaller subunit – primase activity
- one subunit – assist in other three
subunits
- RNA primer 5-15 nucleotides are subsequently
extended by DNA Pol α.

DNA polymerase Delta
- Located in nucleus
- Catalyzes the synthesis of lagging strand
- Having four subunits – POLD 1,2,3,4
- larger subunits catalyzes 5´-3´ polymerization
activity
- Smaller subunits catalyzes 3´-5´ exonuclease
activity (proof reading activity)
- High processivity when interacting with PCNA
(Proliferating cell nuclear antigen).

PCNA
- Molecular weight 25,000; PCNA is important for both
DNA synthesis and DNA repair
- Multimeric protein
- Found in large amount in nuclei of proliferating
cells.
- Act as “clamp” to keep DNA pol δ from
dissociating off the leading DNA strand. “Clamp”
consist of 3 PCNA molecules each containing two
topologically identical domains that are tightly associated to
form closed ring.
- PCNA helps hold DNA polymerase epsilon (Pol ε)
to DNA.
- DNA pol δ improves fidelity of replication by a
factor of 102 due to its proof reading action. It contributes in
limiting the rates of overall error to 10-9 to 10-12.
- DNA Pol δ is also associated with helicase activity.

DNA polymerase Epsilon - Є
located in nucleus
Having four subunits – POLE 1, (Catalytic)
2,3,4 (subunits)associated with - 5´- 3´ polymerization activity 5’- 3’ exonuclease activity (to remove RNA primer) 3’- 5’ exonuclease activity (to proof read)
DNA pol Є catalyzes the repair mechanism
and catalyzes the removal of primer and
filing the primer gap in Okazaki fragments.
Replicating factor A/ Replicating protein
A (RPA/RFA)
RPA/ RFA are similar to single strand binding
protein. They bind to SS DNA and prevent
the re-annealing of parental DNA.

Replication factor C (RFC)
RFC also called as clamp loader or match
maker.
RFC assist in DNA pol δ to form clamp
between DNA and PCNA.
RFC also plays important role in setting up a
link between DNA pol δ and DNA pol α, so
that the leading strand synthesis and lagging
strand synthesis in eukaryotes can take place
simultaneously.

Histone Dissociation and Association Since DNA is present in packaged form as chromatin, DNA replication is sandwiched between two additional steps in eukaryotes.1. Carefully ordered and in complete dissociation of the chromatin.2. Re-association of DNA with the histone octomers to form nucleosome.Dissociation of histone: methylation at the fifth position of cytosine residues by a DNA methyl transferase appears to functioning by loosening up the chromatin structure. This allows DNA access to proteins and enzymes needed for DNA replication.Synthesis of histone: the synthesis of new histone occurs simultaneously with DNA replication.

SEQUENTIAL STEPS IN EUKARYOTIC DNA REPLICATIONDNA replication is a very complicated process that involves several enzymes and other proteins. It occurs in following stages
• Pre-initiation• Initiation• Elongation• Termination• Telomerase function
PRE-INITIATION
Actually during pre-initiation stage, replicator
selection occurs. Replicator selection is the process of
identifying the sequences that will direct the initiation
of replication and occur in G1 phase (prior to S
phase). This process leads to the assembly of a
multiprotein complex at each replicator in the
genome. Origin activation only occurs after cells enter
S phase and triggers the Replicator – associated
protein complex to initiate DNA unwinding and DNA
polymerase recruitment.

Replicator selection is mediated by the formation
of pre-replicative complexes (pre-RCs). The first
step in the formation of the pre-RC is the
recognition of the replicator by the eukaryotic
initiator, ORC (Origin recognition Complex). Once
ORC is bound, it recruits two helicase loading
proteins (cell division cycle protein - Cdc6 and
Cdtl). Together, ORC and the loading proteins
recruit a protein that is thought to be the
eukaryotic replication fork helicase (the Mem 2-7
complex). Formation of the pre-RC does not lead
to the immediate unwinding of origin DNA or the
recruitment of DNA polymerases. Instead the pre-
RCs that are formed during Gl are only activated
to initiate replication after cells pass from the Gl
to the S phase of the cell cycle.

Figure - The steps in the formation of pre-replicative complex (pre-RC)The assembly of the pre-RC is an ordered process that is initiated by the association of the ORC with the replicator. Once bound to the replicator ORC recruits at least two additional proteins Cdc6 and Cdt1 (cell division cycle proteins). These three proteins function together to recruit the putative eukaryotic DNA helicase- the MCM 2-7 (multi-chromatin maintenance protein) complex to complete the formation of the pre-RC

INITIATION
ARS (Autonomously Replicating
Sequences)
In eukaryotes the DNA replication is initiated at
specific site known as ARS
(Autonomously Replication Sequences) or
replicators.
ARS (Origin of chromosome in eukaryotes)
contains
- A central core sequence which contains
highly conserved 11 bp sequence (AT rich
sequence)
- Flanking sequences.

ARS – is 100- 150 long (generally it span about
150 bp)
There are multiple origins in eukaryotes. Eg:
yeast contains 400 ARS. The multiple origins are
spaced 30 -300 kb apart. The sequence between
two origins of replication is called replicons. An
average human chromosome contains as many as
100 replicons and replication may proceed
simultaneously at as many as 200 forks.
- The central core sequence contains 11 bp
elements known as “ARS consensus sequence”
rich in AT pair (It is similar to AT rich 13 mers
present in E.Coli Ori C). It is also
called as ORE (Origin replication element)
- The flanking sequences consist of
overlapping sequence that include varients of core
sequences

ORE (Origin Replicating Element) and
ORC (Origin Recognition Complex)
At the origin there is an association of
sequence specified – ds DNA binding
sequence.
ORE (11 bp sequence in core sequence) binds
to a set of proteins (DNA pol α, DNA pol δ,
RFC, PCNA, RFA, SSB and helicase)
collectively called as ORC Origin Recognition
Complex
ORC is a multimeric protein. Initiation of
replication in all eukaryotes requires this
multimeric subunit protein (ORC) which binds
to several sequences within the replicator.

DUE (DNA Unwinding Element)
ORE located adjacent to approximately 80 bp
AT rich sequence that is easy to unwind.
This is called DUE (DNA Unwinding
Element) Binding of ORC to ORE causes
unwinding at DUE.
Events in replication fork:
When ORC (DNA pol α, DNA pol δ, RFC,
RFA, PCNA, SSB helicase into the origin of
replication especially at ORE, the DNA
synthesis is initiated. The replication fork
moves bi-directionally and replication
proceeds simultaneously as many as 200
forks.

Formation of replication fork:
The replication fork in eukaryotes consists of
four components that form in the following
sequence.
DNA helicase and DNA pol δ (due to its
associated helicase activity) unwinds short
segment of parental DNA at 80 bp AT rich
sequence called DUE (DNA unwinding elements)
which is located adjacent to ORE.
DNA pol α initiated the synthesis of RNA primer.
(DNA pol α is also having primase activity) The
primer is approximately 10 bp.
DNA pol ε in lagging strand and DNA pol δ in
leading strand initiates the daughter strand
synthesis.
SSB and RFA bind to SS DNA and prevent re-
annealing of SS DNA.

In addition to the above, two additional factors play important role in replication of eukaryotesPCNA (proliferating cell nuclear antigen) act as a ‘’clamp’’ to keep DNA pol δ from dissociating off the leading strand and thus increasing the processing of DNA pol ε.RFC also called as ‘clamp loader’ or ‘match maker’.RFC assist in - DNA pol δ to form clamp between DNA and PCNA and - setting up a link between DNA pol δ and DNA pol ε so that the leading Strand and lagging strand synthesis in eukaryotes can take place simultaneously.

Rate of Replication fork Movement
The rate of replication fork movement in
eukaryote (approximately 50 nucleotide /sec) is
only one tenth that observed in E.Coli at this
rate, replication of an average human
chromosome proceeding from a single origin
would take more than 500 hours. Instead of
that, replication of human chromosome
proceeds bi-directionally from multiple origins
spaced 30-300 kb apart and completed within an
hour.
DNA sequence between two origins of
replication is called replicons. An average
chromosome contains nearly 100 replicons and
thus replication proceeds simultaneously at as
many as 200 forks.

ELONGATION
During elongation, an enzyme called DNA
polymerase adds DNA nucleotides to the 3' end
of the newly synthesized polynucleotide strand.
The template strand specifies which of the four
DNA nucleotides (A, T, C, or G) is added at each
position along the new chain. Only the
nucleotide complementary to the template
nucleotide at that position is added to the new
strand. For example, when DNA polymerase
meets an adenosine nucleotide on the template
strand, it adds a thymidine to the 3' end of the
newly synthesized strand, and then moves to
the next nucleotide on the template strand. This
process will continue until the DNA polymerase
reaches the end of the template strand.


All newly synthesized polynucleotide strands
must be initiated by a specialized RNA
polymerase called primase. Primase initiates
polynucleotide synthesis and by creating a short
RNA polynucleotide strand complementary to
template DNA strand. This short stretch of RNA
nucleotides is called the primer. Once RNA
primer has been synthesized at the template
DNA, primase exits, and DNA polymerase extends
the new strand with nucleotides complementary
to the template DNA. Eventually, the RNA
nucleotides in the primer are removed and
replaced with DNA nucleotides. Once DNA
replication is finished, the daughter molecules
are made entirely of continuous DNA nucleotides,
with no RNA portions.

The Leading and Lagging Strands
DNA polymerase can only synthesize new
strands in the 5' to 3' direction. Therefore, the
two newly synthesized strands grow in opposite
directions because the template strands at each
replication fork are antiparallel. The "leading
strand" is synthesized continuously toward the
replication fork as helicase unwinds the template
double stranded DNA.
The "lagging strand" is synthesized in the
direction away from the replication fork and
away from the DNA helicase unwinds. This
lagging strand is synthesized in pieces because
the DNA polymerase can only synthesize in the 5'
to 3' direction, and so it constantly encounters
the previously synthesized new strand. The
pieces are called Okazaki fragments, and each
fragment begins with its own RNA primer.

Leading strand synthesis:- Leading strand synthesis is initiated upon RNA primer, synthesized by the primase subunit of DNA pol α. The RNA primer contains 10-15 nucleotides. - Then DNA pol α adds a stretch of DNA to the primer.- At this point replication factor C (RFC) carries out a process called polymerase switching.- RFC removes DNA pol α and assembles PCNA in the region of primer strand terminus.- Then DNA pol epsilon binds to PCNA and carries out highly processive leading strand synthesis due to its 5’-3’ polymerization activity.- After the addition of several nucleotides in the daughter strand, primer is removed. DNA pol Є due to its 5’-3’ exonuclease activity removes the primer and the gap is filled by the same DNA pol Є due to its 5’-3’ polymerization activity.- Then the nick is sealed by DNA ligase. DNA pol δ improves the fidelity of replication due to its proof reading activity.

Lagging strand synthesis:Lagging strand synthesis of Okazaki fragment initiated same way as leading strand synthesis. An Okazaki fragment contains 150-200 nucleotides.RNA primer is synthesised by DNA pol α due to its primase activity.The primer is then extended by DNA pol delta due to its 5’-3’ polymerization activity (lagging strand synthesis), using deoxy ribonucleotides (dNTPs).Priming is a frequent event in lagging strand synthesis with RNA primers placed every 50 or 80 nucleotides.All but one of the ribonucleotides in RNA primer is removed by RNase H1.Then exonuclease activity of FEN 1/ RTH 1 complex removes the one remaining nucleotide. The gap is filled by DNA pol Є by its 5’-3’ polymerase activity.DNA ligase joins the Okazaki fragment of the growing DNA strand.


Combined activity of DNA pol delta and DNA
pol epsilon:-
Looping of lagging strand allows a combined
polymerase delta and polymerase epsilon
asymmetric dimer to assemble and elongate both
leading and lagging strands in the same overall
direction of fork movement.
TERMINATION
When the replication forks meet each other, then
termination occurs. It will result in the formation
of two duplex DNA. Even though replication
terminated, 5’ end of telomeric part of the newly
synthesized DNA found to have shorter DNA strand
than the template parent strand. This shortage
corrected by the action of telomerase enzyme and
then only the actual replication completed.

TELOMERES
Eukaryotic chromosomes are linear. The ends
of chromosomes have specialized structures
known as ‘Telomeres’.
Telomeres are – short (5-8 bp)
- tandem repeated and
- GC rich nucleotide
sequence.
- Telomeres form protective cap 7-12 kbp
long in the ends of chromosome. Telomeres
are necessary for chromosome maintenance
and stability. They are responsible for
maintaining chromosome integrity by
protecting against DNA degradation and
rearrangement.

Problem in the completion of replication of
lagging strand:
- Linear genomes including those of
several viruses as well as the chromosomes of
eukaryotic cells force a special problem
completion of replication of the lagging strand.
- Excision of an RNA primer from the 5’
end of a linear molecule would leave a gap
(primer gap). This gap cannot be filled by DNA
polymerase action, because of the absence of a
primer terminus to extend. If the DNA could not
be replicated, the chromosome would shorten a
bit with each round of replication.
- This problem has been solved by
Telomerase.

Telomerase:- Telomerase is ribonucleoprotein. It contains a RNA component which has repeat of 9 to 30 nucleotides long. This RNA component serves as the template for the synthesis of telomeric repeats at the parental DNA ends.- Telomerase is a RNA dependent DNA polymerase with a RNA component.
Telomerase uses the - 3’ end of parental DNA strand as primer,- RNA component of telomerase as template,- adds successive telomeric repeats to the parental DNA strand at its 3’ end due to its 5’-3’ RNA dependent DNA polymerase activity.

Regeneration of telomeres:Telomeric DNA consists of simple tandemly repeated sequences like those shown as below:Telomeric repeats sequence at 5’end -
Organism RepeatHuman AGGGTTHigher plant AGGGTTTAlgae AGGGTTTTprotozoan GGGGTTTTYeast GGGT
These sequences are repeatedly added to the 3’ termini of chromosomal DNAs by ‘Telomerase’. Telomerase uses its RNA component as template and parental DNA as primer. Then by its RNA dependent DNA polymerase activity it repeatedly adds telomeric sequences to the 3’ termini of parental DNA. - Then the telomerase is released.- Finally the RNA primer, (of telomerase) is bound near the lagging strand and it is extended by DNA polymerase. Thus the lagging strand synthesis is completed.

In Linear eukaryotic chromosome, once the first primer on each strand is remove, then it appears that there is no way to fill in the gaps, since DNA cannot be extended in the 3′–>5′ direction and there is no 3′ end upstream available as there would be in a circle DNA. If this were actually the situation, the DNA strand would get shorter every time they replicated and genes would be lost forever.

Elizabeth Blackburn and her colleagues have
provided the answer to fill up the gaps with the
help of enzyme telomerase. So, that the genes
at the ends, are conserved. Telomerase is a
ribonucleoprotein (RNP) i.e. it has RNA with
repetitive sequence. Repetitive sequence
varies depending upon the species example
Tetrahymena thermophilia RNA has AACCCC
sequence and in Oxytrica it has AAAACCCC.
Telomerase otherwise known as modified
Reverse Transcriptase. In human, the RNA
template contains AAUCCC repeats. This
enzyme was also known as telomere terminal
transferase..
.

The 3′-end of the lagging strand template
basepairs with a unique region of the telomerase
associated RNA. Hybridization facilitated by the
match between the sequence at the 3′-end of
telomere and the sequence at the 3′-end of the
RNA. The telomerase catalytic site then adds
deoxy ribonucleotides using RNA molecule as a
template, this reverse transcription proceeds to
position 35 of the RNA template. Telomerase then
translocates to the new 3′-end by pairing with
RNA template and it continues reverse
transcription. When the G-rich strand sufficiently
long, Primase can make an RNA primer,
complementary to the 3′-end of the telomere’s G-
rich strand. DNA polymerase uses the newly
made primer to prime synthesis of DNA to fill in
the remaining gap on the progeny DNA. The
primer is removed and the nick between
fragments sealed by DNA ligase

V. Magendira ManiAssistant Professor, PG & Research Department of Biochemistry,Islamiah College (Autonomous),Vaniyambadi,Vellore District – 6357512,Tamilnadu, [email protected] ;
https://tvuni.academia.edu/mvinayagam