National University SUDA · 2019-03-26 · Take the final burette reading. Calculate the volume of...
Transcript of National University SUDA · 2019-03-26 · Take the final burette reading. Calculate the volume of...

Good Luck on the Exam
National University –SUDA Faculty of Clinical and Industrial Pharmacy
Second Year (Batch-PA-13)-Semester Four
Professional Skills-2- Laboratory Skills-1
(PA-SKILL-221)
Course Coordinator: Us. Ghada Mustafa Eltayeb,
Phone Number (00249-912242444 / 00249-990997710).
Assistant Coordinator: Us. Maria Alamin Hamed,
Phone Number (00249-913714903 / 00966567560185)
Ghada

Volumetric analysis involving
acids and alkalis

By the end of this lesson the student is expected ……..
Lecture, No-5
1. To prepare standard solutions
2. To practice acid-alkali titrations
3. To Calculate the concentration of
volumetric analysis
4. To Write a laboratory report on volumetric
analysis experiment
Lecture Objectives

Standard solutions
A solution of accurately known molar
concentration or molarity is called a
standard solution.
Key point
A _s_t_a_n_d_a_rd solution is a solution of
accurately known molar concentration or
molarity.

Apparatus for preparing standard solutions
Table 19.1 Names and uses of the apparatus commonly used in
the preparation of standard solutions.
Name Use
1. Weighing bottle
(a) Glass weighing bottle
(b) Plastic weighing bottle
cap cap
(a) (b)
It is used to hold the
sample to be
weighed.

Name Use
2. Electronic balance
(a) (b)
It is used to weigh
the solid quickly and
accurately (accuracy
up to (a) 0.01 g or
(b) 0.0001 g).
Names and uses of the apparatus commonly used in the
preparation of standard solutions.

Name Use
3. Volumetric flask It is used to make up
a solution to a
specific volume (e.g.
250.0 cm3)
accurately.
* Before use, rinse
the volumetric flask
with distilled water.
Table 19.1 Names and uses of the apparatus commonly used in
the preparation of standard solutions.

Table 19.1 Names and uses of the apparatus commonly used in
the preparation of standard solutions.
Name Use
4. Pipette
bulb
It is used to deliver a
specific volume (e.g.
25.0 cm3) of a
solution accurately.
* Before use, rinse
the pipette with
distilled water, then
with the solution it
is going to deliver.
19.1 Standard solutions
P. 8 / 74

Table 19.1 Names and uses of the apparatus commonly used in
the preparation of standard solutions.
Name Use
5. Pipette filler
(a) The simple type
(b) The traditional type with valves
valves
It is used to suck up
a solution into a
pipette.
* As a safety
precaution, always
fill a pipette with a
pipette filler. Do NOT
use your mouth.

Name Use
6. Beaker It is used to hold
solutions.
Names and uses of the apparatus commonly used in the
preparation of standard solutions.

Preparing standard solutions
Primary standard
A primary standard is a substance which can
be used to prepare a standard solution
directly.
Learning tip
Only very few substances can be used as a
primary standard. Examples include
anhydrous sodium carbonate and oxalic acid
crystals.

A good primary standard should have the following properties:
readily available with high purity
chemically stable (should not react
with
oxygen, carbon dioxide and water vapour in
air)
high solubility in water
does not absorb water vapour from air.
does not lose water of crystallization to
the atmosphere on exposure to air if it is
a hydrated substance
involatile and non-toxic

Preparing a standard solution by dissolving a pure solid
1000
250.0 dm3 106.0 g mol–1 = 0.10 mol dm–3
= 2.65 g
Steps for preparing 250.0 cm3 of an
approximately0.10 M sodium carbonate solution
Stage A: Weighing the pure solid
1. Calculate the mass of anhydrous sodium
carbonate required.
Mass of Na2CO3 required
= number of moles of Na2CO3 molar mass of Na2CO3

2. Use an electronic balance to weigh out accurately the required mass of anhydrous sodium carbonate.
weighing
bottle anhydrous
sodium
carbonate
Steps for preparing a standard
solution by dissolving a pure
solid in distilled water.
SBA note
Do not waste time trying to weigh out exactly 2.65
g of the solid. However, the mass weighed out
should be accurate (e.g. to be at least 0.01 g).

Stage B: Dissolving the pure solid
(3
)
anhydrous
sodium
carbonate
sodium
carbonate
solution
3. Dissolve the solid in about 100 cm3 of distilled
water in a beaker. Stir the solution with a glass
rod.
100 cm3 of
distilled
water
glass rod
beaker

Stage C: Making up the solution to 250.0
cm3 in a volumetric flask
(4)
4. Pour the solution to a 250.0 cm3 volumetric
flask through a filter funnel.
glass rod
sodium
carbonate
solution filter funnel
volumetric flask
(250.0 cm3
type)

5. Wash the beaker and the glass rod with distilled water several times. Pour all the washing to the volumetric flask.
plastic
wash
bottle distille
d
water
washin
g
sodium
carbonate
solution
distille
d
water
(5)

beake
r distille
d
water
6. Add distilled water to the volumetric flask but stop about 2 cm below the graduation mark.
7. Use a dropper to add distilled water until the
bottom of the meniscus reaches the graduation
mark. droppe
r
250.0 cm3
graduatio
n mark
distille
d
water
(6
)
(7
)

8. Stopper the flask and invert it several times to mix the contents well.
stoppe
r
meniscus
invert it
several
times
standard sodium
carbonate
solution
(8)
Example 19.2

Preparing a standard solution by diluting a
concentrated solution of known molarity
Steps for preparing 250.0 cm3 of 0.0120 M
sodium carbonate solution from
0.120 M sodium carbonate solution
1. Calculate the volume of the original solution
required for dilution. number of moles of Na2CO3 = number of moles of Na2CO3
before dilution after dilution
M1V1 = M2V2
∴ V1 = 25.0 cm3
0.120 V1
1000 = 0.0120 250.0
1000

2. Use a pipette to transfer 25.0 cm3 of the original solution to a 250.0 cm3 volumetric
flask. pipett
e
filler
0.120 M sodium
carbonate
solution
25.0
cm3
pipette
meniscu
s
25.0
cm3
pipette
0.120 M sodium
carbonate
solution
0.120 M
sodium
carbonat
e
solution
250.0 cm3
volumetric
flask
(1) and (2)
Steps for preparing a standard solution by diluting a
concentrated solution.

NOTE You should use the index finger instead
of the thumb to control the flow of solution in a pipette.

3. Add distilled water to the volumetric flask but stop about 2 cm below the graduation mark.
4. Use a dropper to add distilled water until the
bottom of the meniscus reaches the graduation
mark. beake
r distille
d
water
droppe
r distille
d
water 250.0 cm3
graduatio
n mark
(4
)
(3
)

invert it
several
times stopper
meniscus
0.0120 M sodium
carbonate
solution (5)
5. Stopper the flask and then invert it several times to mix
the contents well.

19.2 Acid-alkali titrations
Volumetric analysis and titration
Volumetric analysis
is a method to determine the amount (or
concentration) of a substance in a
sample.
requires the measurement of volumes of
the solutions involved.
is also known as titrimetric analysis as it
is done by titration.

Titration is an experimental technique used for
determining the amount (or concentration) of
a substance in a solution.
In a titration, a solution of known concentration
(i.e. a standard solution) is slowly added to
another solution of known volume, until the two
solutions have just reacted completely.

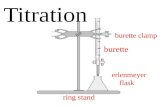
Apparatus for titration
Name Use
1. Burette
stopcock
It is used to deliver
variable volumes (ranging
from 0.05 to 50.00 cm3) of
a solution accurately.
* Before use, rinse the
burette with distilled
water and then with the
solution it is going to
deliver.
Names and uses of some apparatus used in acid-alkali titration.

Name Use
2. Conical flask It is used to hold a
solution to be titrated. Its
conical shape allows it to
be swirled gently without
spilling out the solution.
* Before use, rinse
the conical flask with
distilled water.
Table19.2 Names and uses of some apparatus used in acid-alkali
titration.

Name Use
3. Stand and clamp They are used to support
the burette.

Name Use
4. White tile It is used to allow clear
observation of the colour
change of the indicator.

Procedure for acid-alkali titration
A standard solution of an acid (or an alkali) is
slowly added to a known volume of a solution of
an alkali (or an acid), until the acid and alkali
have completely reacted with each other.
The stage at which the acid and the alkali have
just reacted completely during titration is called
the equivalence point.
Learning tip
The procedure for an acid-carbonate (e.g. HCl
and Na2CO3) titration is more or less the same
as that for the acid-alkali titration.

Key point
The equivalence point of an acid-alkali titration
is the point at which an acid and an alkali have
just
_c_o_m_p_l_e_t_e_ly_ reacted with each other.
The equivalence point of an acid-alkali titration
can be estimated by using a pH meter or a
data- logger connected with a pH sensor.
It is not possible to detect the equivalence
point using an acid-base indicator.

During the acid-alkali titration, a suitable acid-
base indicator is usually added.
The colour of the indicator changes sharply at
or very near the equivalence point, then the
end point of titration is reached.
Key point
_En_d_p_o_i_n_t_ is the point at which the
indicator changes colour sharply.

Detection of end point by using an acid-base indicator
alkal
i
conical
flask alkali
alkal
i
1. Fill the 25.0 cm3 pipette (using a pipette filler)
with the alkali until the graduation mark is
reached. Transfer 25.0 cm3 of the alkali to a
conical flask.
25.0
cm3
pipette
(1
)
Procedure for performing an acid-alkali titration.

2. Fill the burette with the acid. Make sure that the jet is filled completely (no air bubbles inside the jet).
Learning tip
The solution in the burette is often known as
titrant.
white
tile
clam
p
stan
d aci
d
burett
e
(2
)

3. Add a few drops of an acid-base indicator to the alkali. Note the colour
of the solution. 4. Take the initial burette reading, with your eyes
on the same level as the meniscus.
indicato
r
alkal
i (3
)
meniscu
s
eye at
prope
r
level
read as 0.6 cm3

5. Slowly add the acid to the alkali until the mixture just turns to another persistent colour. This is the end point of titration.
burett
e
acid
solution mixture + indicator
(5)

6. Take the final burette reading. Calculate the volume of the acid added to reach the end
point. 7. Repeat the titration (steps 1 to 6) at least three
times, until we get three sets of data for
calculating the average volume of the acid
used.
Learning tip
The volume of titrant used to react completely
with the solution in the conical flask is called
titre.

methyl
orange
pH = 2 pH =12
phenolphthalei
n
Choosing a suitable acid-base indicator
Methyl orange and phenolphthalein can give a sharp colour change at the end point of titration. Litmus is not used because it does not give a sharp colour change.
(a) pH = 2 pH = 12 (b
)
Figure The sharp colour changes of (a) methyl orange and
(b) phenolphthalein in solutions of pH = 2 and pH = 12.
(There is often a marked change in pH at the end point of
titration. The change from pH = 2 to pH = 12 is used as an
example here.)

Titration type Example Suitable
indicator(s)
Strong acid – strong alkali
HCl(aq) vs. NaOH(aq)
methyl orange;
phenolphthalein
Strong acid – weak alkali
(Strong acid – carbonate)
HNO3(aq) vs. NH3(aq)
HCl(aq) vs. Na2CO3(aq)
methyl orange
Weak acid – strong alkali
CH3COOH(aq) vs. KOH(aq)
phenolphthalein
Weak acid – weak alkali
CH3COOH(aq) vs. NH3(aq)
(no indicator
would be suitable)
The correct choice of indicators for different types of acid-alkali
titrations.
19.2 Acid-alkali titrations















![Gnc suda[1]](https://static.fdocuments.net/doc/165x107/55bef06cbb61eb32538b45bf/gnc-suda1.jpg)



