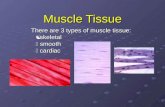
(a) Skeletal muscle (b) Smooth muscle - MIT OpenCourseWare · PDF fileSkeletal (striated) and...
-
Upload
truongcong -
Category
Documents
-
view
225 -
download
1
Transcript of (a) Skeletal muscle (b) Smooth muscle - MIT OpenCourseWare · PDF fileSkeletal (striated) and...

undergoing periodic contractions
Structure of muscle
Courtesy of Jan Lammerding. Used with permission.
Isolated cardiac myocyte in culture
1�
Image removed due tocopyright considerations.

Skeletal (striated) and smooth muscle�
Temporal patterns of muscle contraction •
•
• max)
l
(b) Smooth muscle(a) Skeletal muscle
Single twitch
Periodic sequence of excitations
Fused tetanus (F
2

Tension-length curves for a muscle fiber (relaxed and maximally stimulated)
l
Empirically determined force-velocity relationship obtained
Hill’s equation
from macroscopic measurements
v
vmax
= 1- F Fmax( )
1+ max( )
0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
0 0.2 0.4 0.6 0.8 1
v/vmax
F/
Fm
ax o
r P
/P
max
vF
vmaxFmax
= 1- ( Fmax F( ) + CC F F
F/Fmax
vF/Fmaxvmax
F Fmax)
3�

Source of energy for muscle�
Hydrolysis of adenosine triphosphate (ATP) creating adenosine diphosphate (ADP):
æATP æ Æ ææ ADP + Piactomy sin ATPase
Ê [ATP] ˆ DG = DG0 - kT lnÁ ˜
PiË [ADP][ ]¯
or approximately -25 kT (displacement ~ 5 nm)
Power/weight ~ same as an automobile engine
4

A rise in cytosolic Ca2+ triggers muscle contraction (part I)
2+
Step 1: An excitation signal travels along the efferent nervous pathways towards the muscle. Step 2: The excitation signal de-polarizes the cell membrane. This allows spread of the action potential along the
Step 3: The potential triggers the release of calcium into the
unit. Step 4:
chemical, mechanical, and electrostatic actions. Step 5: The stepping action of myosin along the adjacent
Step 6: reticulum (ATP-dependent) switches the contraction activity off.
A rise in cytosolic Ca triggers muscle contraction (part II)
sarcoplasmic reticulum.
sarcoplasmic matrix surrounding the filaments of the motor
This removes the hindrance (tropomyosin) for interactions between actin and myosin filaments through
actin filament causes the two to slide relative to each other, reducing the length of the sarcomere, producing contraction.
Sequestration of calcium ions in the sarcoplasmic
5�

array of actin and myosin Skeletal muscle contains a regular
6�

/
http://www.scripps.edu milligan/research/movies/myosin_text.html
couple ATP hydrolysis to movement Conformational changes in the myosin head
8�

http://www.sci.sdsu.edu/movies/actin_myosin.html
Image removed due to copyright considerations.
Rhodamine phalloidin labeled actin moves on a myosin coated cover slip (black)
observed with epifluorescence microscopy. This motility assay uses the antibody
capture technique.
D. A. Winkelmann, L. Bourdieu, A. Ott, F. Kinose, A. Libchaber: "Flexibility of
Myosin Attachment to Surfaces Influences F-Actin Motion" Biophys J. 68, 1995,
2444-2453.
9

The sliding filament model A. Huxley & Niedergerke, H. Huxley & Hanson,
Nature, 1954
A quantitative model A. Huxley 1957
See also textbooks by T. McMahon or J. Howard
• While in the bound state, the myosin head behaves as though loaded by linear springs with spring constant, k, and that it passes
through the necessary biochemical processes including binding of ATP,
ATP hydrolysis, and release of ADP. • Only the case of constant (time-invariant) relative sliding
velocity and force generation is considered. • The muscle is assumed to be maximally activated throughout.
• Attachment and detachment is assumed to obey simple kinetics.
• Effects of other elastic components in the muscle are ignored.
Myosin headMyosin filament
Actin binding site
Actin filament
x� As the actin filament moves past the (fixed) myosin filament, the myosin head can bind tok+ it at the red triangle. When it does, the springsk- are either stretched or compressed and a force kx acts at the binding
x site.
10

Equations governing the probability�n(x,t) that a cross-bridge is attached�
dn(x,t) ∂n(x,t) ∂n(x,t) = - v = [1- n(x,t)]k+ (x)- n(x,t)k (x)-dt ∂t ∂x Formation of new Detachment of existing
bonds bonds
At steady state [n = n(x)]
dn(x) -v = [1- n(x)]k+ (x)- n(x)k (x)-
kdx
+k+ = attachment rate; k- = k-�detachment rate; n =�probability of attachment� x
h
The sliding filament model
x > h:�In this region the actin binding site is approaching the free myosin head, unoccupied.�Since both k+ and k- are zero, no binding occurs:�
n(x) = n(h) = 0
h-x0 < x <h:�If binding is to occur, it has to do so (according to this simple model) within this narrow�region where the binding rate constant is large, described by the equation:�
-v dn
= (1 - n)k 0�
dx +�
k+ k-
Ê k 0 + x0
ˆ n(h - x0) = 1 - exp�Á- ˜�
Ë v ¯� xh
11

0 < x < h-x0
Both the attachment and detachment rate constants are zero, so the myosin head can
neither bind to nor detach from an actin filament, and the probability of attachment
remains constant:
n(x) = n(h-x0) = constant
x < 0
As the complex moves into the region x < 0, the force of interaction sustained at the
actin-myosin bond changes sign and its probability of attachment begins to fall, as
ˆ ˜¯
described by the equation:
-ÊÁË
dn 0kv n= -
˘˙˚
ˆ ˜¯
k+dx k-
k+ ÊÁË
0k0x n( x) n(0) exp = 1 exp -
x0 exp k0x
v v v -
x ÈÍÎ
h
ˆ ˜¯
-�
-�
ÊÁË
-
=
=
s
r
s
Work done by a single cross-bridge that attaches at x=a and detaches at x=b:
W
Askss = 2lA
s =
max
k
Ú-
r
a
Ú -
xdx = b
•�
•�
skh 2 s
ÊÁË
ÈÍÎ
(
˘˙˙̊
k 2
ˆ ˜¯
ÈÍÍÎ
a
n(x)xdx =
1 - 2
2 v
1max
)-
r
2 b2
Asks ˘ ˙˚
k]
-
h
Ú
r=
=
ˆ ˜¯
-
˘˙˚
s
ˆ ˜¯
[
ÊÁË
Ú-
+-
s
ÊÁË
˘ ˙˚
ˆ ˜¯
0
Ú-
+
ÈÍÎ
-
•
lA = n(x)r As /2 xdxs •
k 0 x n(0)x exp dx + n(0)xdx
• v 0
1 - exp k 0 x0
v
xskh2
0 maxk 0 s
4l v
hk0
vmax 2
ÈÍÎ
2lA
˘ ˙ ˙̊
ÊÁË
ˆ ˜¯ -
2 v
hk 0
- exp
4l
ÊÁË
-È Í ÍÎ
= 1v
12

Predicted force-velocity curve from cross-bridge model
F
Fmax
= 1-v
vmax
Ê
ËÁ
ˆ
¯˜
2È
Î
Í Í
˘
˚
˙ ˙
1- exp -k+
0x0
v
Ê
ËÁ
ˆ
¯˜
È
ÎÍ
˘
˚˙
-0.2
0
0.2
0.4
0.6
0.8
1
0 0.2 0.4 0.6 0.8 1
V/Vmax
F/
Fm
ax
0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
0.9
1
0 0.2 0.4 0.6 0.8 1
v/vmax
F/
Fm
ax o
r P
/P
max v
vmax
= 1- F Fmax( )
1 + max( )
Hill’s equation
C F F
13�