Y Pradeep Agarwal Academy( Taught By IITians) · PDF fileNucleophilic substitution reaction...
Transcript of Y Pradeep Agarwal Academy( Taught By IITians) · PDF fileNucleophilic substitution reaction...

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
1
REACTION MECHANISM1. NUCLEOPHILICITY& LEAVING GROUPABILITY
1.1 Nucleophiles : It is the electron rich species having atleast one lone pair of electrons. It can be
neutral or negativetely charged it is always a lewis base.
e.g. CN–, OH
–, Br
–, I
–, NH
3, H
2O
1.2 Basicity : It is the tendency to donate electron pair to ion.
(a)
(b)
1.3 Nucleophilicity : The tendency to give electron pair to an electron deficient carbon atom is defined
as nucleophilicity.
Criteria for Nucleophilicity :
– The factors which increases electron density at donor atom increases nucleophilicity.
– The more polarisable donar atom is a better nucleophile. Therefore, large size of donor atom increases
nucleophilicity.
Periodicity :
–Nucleophilicity decreases from left to right in a period.
–In a group, nucleophilicity increases from top to bottom due to increases in size of donor atom, but
basicity decreases from top to bottom.
Acid strength : HI > HBr > HCl > HF
Basic strength : F–
> Cl–
> Br–
> I–
Nucleophilicity : F–
< Cl–
< Br–
< I–
Steric effects on nucleophilicity
Stronger base, yet weaker Weaker base,
nucleophile cannot approach yet stronger
the carbon atom so easily. nucleophile
The effect of the solvent :
– In polar protic solvent large nucleophiles are good, and the halide ions show the following order
F–
< Cl–
< Br–
< I–
– In DMSO, the relative order of nucleophilicity of halide ions is
F–
> Cl–
> Br–
> I–
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
2
– This effect is related to the strength of the interaction between nucleophile and solvent molecules of
polar protic solvent forms hydrogen bond to nucleophiles in the following manner :
Relative nucleophilicity in polar protic solvent
> > > > > > > H2O
e.g. > More extensive resonance.
e.g. HO¯ > H2O Anion are better nucleophile than their neutral molecule
e.g. NH2¯ > NH
3
e.g. CF3SO
3¯ < PhCOO¯ < PhO¯ < RO¯
Nucleophilicity Basicity Remarks
1. CH3¯ > NH
2¯ > OH¯ > F¯ CH
3¯ > NH
2¯ > OH¯ > F¯ If donor atoms belong to
same period, nuclephilicity
and basicity order is same
2. SiH3¯ > PH
2¯ > SH¯ > Cl¯ SiH
3¯ > PH
2¯ > SH¯ > Cl¯ ––//––
3. F¯< Cl¯ < Br¯ < I¯ F¯> Cl¯ > Br¯ > I¯ In a group nucleophilicity
increases while basicity
decreases. on moving top to
bottom.
4. OH¯ < SH¯ OH¯ > SH¯ ––//––
5. RO¯ < RS¯ RO¯ > RS¯ ––//––
6. RO¯ > HO¯ RO¯ > HO¯ If donor atom is same,
nucleophilicity and basicity
have same order
1.4 Leaving group Ability / Nucleofugality :
The best leaving groups are those that become the most stable ion after they leave, because leaving
group generally leave as a negative ion, so those leaving group are good, which stabilise negative charge
most effectively and weak base do this best, so weaker bases are always good leaving groups.
– The leaving group should have lower bond energy with carbon.
– Negative charge should be more stable either by dispersal or delocalization.
– The weaker bases are better leaving groups.
Strongly basic ions rarely act as leaving group
(strong base / poor leaving group)
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
3
(It is not a leaving group)
Some other leaving groups are
e.g. F¯ < Cl¯ < Br¯ < I¯
e.g. CH3¯ < NH
2¯ < OH¯ < F¯
e.g. R–COO¯ > PhO¯ > HO¯ > RO¯
e.g. SH¯ > OH¯
1.5 Types of solvents
(a) Non polar
(b) Polar (These solvents are of two type - polar protic and polar aprotic)
Solvents Polar Protic Aprotic
1. H2O –
2. CH3OH –
3. CH3CH
2OH –
4. H–COOH –
5. CH3–COOH –
6. NH3
–
7. ×
8. × DMSO – Dimethyl sulphoxide
9. × DMF – Dimethyl formamide
10. × DMA – Dimethyl acetamide
11. ×
12. ×
13. C–C–C–C–C–C ×
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
4
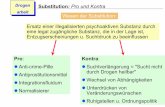
2. Nucleophilic substitution reaction (SN) :
Replacement (displacement) of an atom or group by an other atom or group in molecule is known assubstitution reaction. If substitution reaction is brought about by a nucleophile then it is known asnucleophilic substitution reaction. Generally substitution takes place at sp3 carbon.
R–g + R – Nu +
2.1 Unimolecular nucleophilic substitution reaction (SN1) :
Nucleophilic substitution which involves two step process(a) First step : - Slow step involves ionisation to form carbocation
R–g R+ + g –
(b) Second step : - Fast attack of nucleophile on carbocation to result into product .
R+ + Nu – R–Nu
2.1.1 SN1 Reaction of Alkyl halide
Mechanism :
)rds(stepSlow
halidealkylofIonisation
Characteristics of SN1 reactions :
1. It is unimolecular, two step process.2. Carbocation intermediate is formed so rearrangement is possible in S
N1 reaction.
3. It is first order reaction4. Kinetics of the reaction Rate [Alkyl halide]
Rate of SN1 reaction is independent of concentration and reactivity of nucleophile.
5. Energetics of the SN1
Figure : Free energy diagram for the SN1 reaction.
6. Factors affecting the rates of SN1
(i) The structure of the substrate : The rds of the SN1 reaction is ionization step, a carbocation is formed
in this step. This ionisation is strongly endothermic process, rate of SN1 reaction depends strongly on
carbocation stability because carbocation is the intermediate of SN1 reaction which determines the
energy of activation of the reaction.
SN1 reactivity : 3° > 2° > 1° > CH
3– X
(ii) Concentration and reactivity of the nucleophile The rate of SN1 reactions are unaffected by the
concentration and nature of the nucleophile# Weak, neutral, mostly solvents (protic) itself functions as nucleophiles in S
N1 reaction. So S
N1 reaction
are termed as solvolysis reaction.water hydrolysis ; C
2H
5OH ethanolysis
CH3COOH acetolysis ; NH
3 ammonolysis
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
5
(iii) Effect of the solvent : The ionising ability of the solvent :
Because to solvate cations and anions so effectively the use of a polar protic solvent will greatly increase
the rate of ionisation of an alkyl halide in any SN1 reaction. It does this because solvation stabilises the
transition state leading to the intermediate carbocation and halide ion more than it does the reactant,thus the energy of activation is lower.
R – X (Solvolysis)
Solvated ions
Table - : Dielectric constants () and ionisation rates of t-Butylchloride in common solvents
Solvent Relative rate
H2O 80 8000
CH3OH 33 1000
C2H5OH 24 200
(CH3)2CO 21 1
CH3CO2H 6 –
(iv) The nature of the leaving group In the SN1 reaction the leaving group begins to acquire a negative
charge as the transition state is reached stabilisation of this developing negative charge at the leavinggroup stabilises the transition state and this lowers the free energy of activation and there by increasesthe rate of reaction. Leaving ability of halogen is F¯ < Cl¯ < Br¯ < I¯
7. Stereochemistry of SN1 reactions In the S
N1 mechanism, the carbocation intermediate is sp2
hybridized and planar, A nucleophile can attack on the carbocation from either face, if reactant is chiralthan after attack of nucleophile from both faces gives both enantiomers as the product, which is calledracemisation.
Mechanism of racemisation (SN1
e.g. acetoneOH2 + HBr (3° alkyl halide)
e.g. Ag/OHCH3 (carbocation rearrangement)
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
6
2.1.2 SN1 Reaction of Alcohols
(A) Reaction with hydrogen halides
A common method is to treat the alcohol with a hydrohalic acid, usually HI or HBr. These acids areused to convert alcohols to the corresponding alkyl halides.
(i) In acidic solution, an alcohol is in equilibrium with its protonated form. Protonation converts the
hydroxy group from a poor leaving group to a good leaving group (H2O). If the alcohol is
protonated all the usual substitution and elimination reactions are feasible, depending on the struc-ture (1°, 2°, 3°) of the alcohol.
(ii) Halides are anions of strong acids, so they are weak bases. Solutions of HBr and HI contain
nucleophilic and ions.
(iii) Concentrated hydrobromic acid rapidly converts t-Butyl alcohol to t-Butyl bromide. The strong acidprotonates the hydroxyl group, converting it to a good leaving group. The hindered tertiary carbonatom cannot undergo S
N2 displacement, but it can ionise to a tertiary carbocation. Attack by
bromide ion gives the alkyl bromide. The mechanism is similar to other SN1 mechanism.
(iv) 1-Butanol reacts with sodium bromide in concentrated sulfuric acid to give 1-Bromobutane by an SN2
displacement.
oltanbu1OHCH)CH(CH 2223
42SOH,NaBr
%)90(etanbromobu1
BrCH)CH(CH 2223
Protonation converts the hydroxy group to a good leaving group, but ionization to a primary carbocationis unfavourable. The protonated unbranched primary alcohol is well suited for the S
N2 displacement.
(v) Secondary alcohols also react with HBr to form alkyl bromides usually by the SN1 mechanism.
e.g. HBr
(vi) HCl (Hydrochloric acid) reacts with alcohols in much the same way that as the hydrobromic acid.
(vii) Chloride ion is a weaker nucleophlile than bromide ion because it is smaller and less polarizable.Lewis acid, such as ZnCl
2, is sometimes necessary to promote the reaction of HCl with primary and
secondary alcohols.
Mechanism :
R –OH 2HOR
RDS
O2H–
R R – X
Reactivity of HX : HI > HBr > HCl
Reactivity of ROH : allyl benzyl > 3° > 2° > 1°
e.g.
alcoholIsopropylOH|CHCHCH 33
bromideIsopropylBr|CHCHCH 33
e.g.
3
23
3
CH|
OH–CHCCH|
CH
Br|
CH–CHCCH|
CH
323
3
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
7
Lucas Reagent
(i) A mixture of concentrated hydrochloric acid and zinc chloride is called the Lucas reagent.
(ii) Whether an alcohol is primary, secondary or tertiary is identified by the Lucas test, which is basedupon the difference in reactivity of the three classes of alcohol towards hydrogen halides.
(iii) Alcohol (of not more than six carbons in their molecule) are soluble in the Lucas reagent. Thecorresponding alkyl chlorides are insoluble.
(iv) Formation of a chloride from an alcohol is indicated by the cloudiness that appears when the chlorideseparates from the solution hence, the time required for cloudiness to appear is a measure of thereactivity of the alcohol.
(v) A tertiary alcohol reacts immediately with the Lucas reagent, a secondary alcohol reacts within fiveminutes and a primary alcohol does not react appreciably at room temperature.
e.g.
OH|
CHCHCHCHCH 3223 HCl/ZnCl.anhy 2
entaChloropen3Cl|
CHCHCHCHCH 3223
2.1.3 SN1 Reactions of Ethers
(A) Reaction with HXEthers are unreactive towards most bases, but they can react under acidic conditions. A protonatedether can undergo substitution or elimination with the expulsion of an alcohol. Ethers react with conc.
HBr and HI because these reagents are sufficiently acidic to protonate the ether, while bromide iodide
are good nucleophiles for the substitution.If R or R’ is 3º then mechanism will be S
N1 otherwise S
N2 .
Mechanism :
R – O – R` ROH–
X
R` – X
e.g. (CH3)
3COC(CH
3)
3
HCl
H|
)CH(COC)CH( 3333
C)CH( 33 + (CH
3)
3COH
(CH3)
3CCl
(CH3)
3COH + HCl (CH
3)
3CCl + H
2O
2.2 Bimolecular nucleophilic substitution reaction (SN2)
Nucleophilic substitution in which incoming group replaces leaving group in one step only.
2.2.1 SN2 Reaction of Alkyl halide :
Mechanism :
+
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
8
Characteristic of SN2
1. It is bimolecular, one step concerted process2. It is second order reaction because in the rds both species are involved
3. Kinetics of the reaction rate [alkyl halide] [nucleophile]
rate = k[alkyl halide] [nucleophile]
If the concentration of alkyl halide in the reaction mixture is doubled, the rate of the nucleophilic
substitution reaction is double. If the concentration of nucleophile is doubled the rate of reaction is also
double. If the concentration of both are doubled then the rate of the reaction quadriples.
4. Energetics of the reaction :
Figure : A free energy diagrams for SN2 reaction
5. No intermediates are formed in the SN2 reaction, the reaction proceeds through the formation of an
unstable arrangment of atoms or group called transition state.
6. The stereochemistry of SN2 reactions As we seen earlier, in an S
N2 mechanism the nucleo-
phile attacks from the back side, that is from the side directly opposite to the leaving group. This
mode of attack causes an inversion of configuration at the carbon atom that is the target of nucleo-
philic attack. This inversion is also known as Walden inversion.
Inversion
7. Factor's affecting the rate of SN2 reaction Number of factors affect the relative rate of S
N2
reaction, the most important factors are
(i) Effect of the structure of the substrate
SN2 reactivity CH
3> 1° > 2° >> 3° (unreactive)
The important reason behind this order of reactivity is a steric effect. Very large and bulky groups can
often hinder the formation of the required transition state and crowding raises the energy of the transition
state and slows down reaction.
Table : Relative rate of reactions of alkyl halide in SN2 reaction.
Substituent Compound Relative rate
Methyl CH3X 30
1° CH3CH2X 1
2° (CH3)2CHX 0.02
Neopentyl (CH3)3CCH2X 0.00001
3° (CH3)3CX ~ 0
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
9
(ii) Concentration and reactivity of the nucleophile
– As nucleophilicity of nucleophile increases rate of SN2 increases.
– Anionic nucleophiles mostly give SN2 reaction
– A stronger nucleophile attacks upon -carbon with faster rate than the rate of departing of leaving
group.
Table :
(iii) The effect of the solvent: Polar aprotic solvent have crowded positive centre, so they do not solvatethe anion appreciably therefore the rate of S
N2 reactions increased when they are carried out in polar
aprotic solvent.(iv) The nature of the leaving group Weaker bases are good leaving groups. A good leaving group alwaysstabilise the transition state and lowers its free energy of activation and there by increases the rate ofthe reaction. Order of leaving ability of halide ion F¯ < Cl¯ < Br¯ < I¯
Examples of SN2 reactions of alkyl halide
+ R – X – R + X¯
Nucleophile Product Class of Product
R – X + :¯..
..I :–R..
..I alkyl halide
R – X + HO:¯..
.. R – HO..
.. alcohol
R – X + RO:¯..
.. R – RO..
.. ether (Williamson synthesis)
R – X + HS:¯..
.. R – HS:
..
.. thiol (mercaptan)
R – X + RS:¯..
.. R – RS
..
.. thioether (sulphide)
R – X + 3NH: R –
NH3
X¯ amine
R – X + :¯NNN¯:....
R – :¯NNN....
azide
R – X + ¯:C C – R R – C C – R alkyne
R – X + ¯:C N: R – C N: nitrile
R – X + R – :¯OCO..
.. R – COO – R ester
R – X + :P(Ph)3
[R – PPh3]+
_
X phosphonium salt
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
10
e.g. (CH3)
2CHCH
2CH
2– Br
OH
KOH
2
(CH3)
2CHCH
2CH
2– OH
e.g.
e.g. MeOH Na BrPhCH2
2.2.2 SN2 Reaction of Alcohol
(A) Reaction with HX : The protonated unbranched primary alcohol is well suited for the SN2 reaction.
Mechanism :
R –OH 2HOR
X
R – X + H2O
e.galcoholPentyln
OHCHCHCHCHCH 22223
chloridePentyln
ClCHCHCHCHCH 22223
(B) Reaction with phosphorus trihalidesSeveral phosphorus halides are useful for converting alcohols to alkyl halides. PBr
3, PCl
3, & PCl
5work
well and are commercially available.Phosphorus halides produce good yields of most primary and secondary alkyl halides, but none works
well with tertiary alcohols. The two phosphorus halides used most often are PBr3and the P
4/I
2combi-
nation.
3R – OH + PX3
3R – X + H3PO
3
Mechanism :
Step : 1
Step : 2 RCH2X+ HOPX
2
RemarksThe mechanism for the reaction involves attack of the alcohol group on the phosphorus atom, displacinga halide ion and forming a protonated alkyl dihalophosphiteIn second step a halide ion acts as nucleophile to displace HOPX
2, a good leaving group due to the
electronegative atoms bonded to the phosphorus.
e.g.
oltanbu1Methyl2OH–CH–CH–CH–CH
|CH
223
3
3PBr
etanbromobu1Methyl2Br–CH–CH–CH–CH
|CH
223
3
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
11
e.g.alcoholEthyl
OHCHCH 23 2P I
iodideEthylCHCH 23 I
e.g.
(C) Reaction with thionyl chloride in presence of pyridineThionyl chloride (SOCl
2) is often the best reagent for converting an alcohol to an alkyl chloride. The by
products (gaseous SO2
and HCl) leave the reaction mixture and ensure that there can be no reversereaction.
R – OH + ClSCl||O
Pyridine
Heat R – Cl + SO
2+ HCl
Mechanism :
H–
R – O – SO
Cl
:..
.. ..
..
Chlorosulphite ester
+ HCl
R – Cl + SO2
In the first step, the nonbonding electrons of the hydroxy oxygen atom attack the electrophilic sulphuratom of thionyl chloride. A chloride ion is expelled a proton and gives test of chloro sulphite ester.Second step is an S
N2 mechanism
e.g.Py
SOCl2
Reaction with thionyl chloride
ROH + SOCl2 RCl + SO
2+ HCl
In this mechanism an internal nucleophile attacks from the same side of leaving group , means retensionof configuration . It is an S
Ni mechanism , where i means internal
Mechanism :
H–
R – O – SO
Cl
:..
.. ..
..
Chlorosulphite ester
+ HCl
R – Cl + SO2
e.g.
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
12
2.2.3 SN2 Reaction of Ether
(A) Reaction with HX
A protonated ether can undergo substitution reaction. Ether react with conc. HBr and HI because these
reagents are sufficiently acidic to protonate the ether. If R or R’ is 3º then mechanism will be SN1
otherwise SN2.
Mechanism :
+ halidealkyl
RX + HXX – R + X – R
e.g. CH3CH
2– O – CH
3 H
e.g. HBrexcess
e.g.
2.3 SN
Reaction of EpoxideEpoxides are much more reactive than ether because of angle strain in three membered ring thereforeepoxide readily undergo nucleophilic substitution reaction.
In basic medium mechanism is SN2. Nucleophile atacks on less hindered carbon.
Mechanism :
O|
CH—CH—R|Nu
2 H
OH|
CH—CH—R|Nu
2
e.g.
In acidic medium mechanism is SN1 type. Nucleophilic attacks on more substituted carbon.
Mechanism :
H
OH|CH—CH—R
|Nu
2
e.g.
H
OH2
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
13
3. Elimination reactions :In an elimination reaction two atoms or groups (YZ) are removed from the substrate and generally
resulting into formation of bond.
ZY||CC||
YZ
inationlimE
-elimination : When two groups are lost from the same carbon atom to give a carbene (or nitrene).
This is also called 1–1 elimination.
-elimination : When two groups are lost from adjacent atoms so that a new bond is formed. This
is also called 1–2 elimination.
–elimination : It is also called 1–3 elimination, In this a three membered ring is formed.
3.1 E1 Reaction : Proton and leaving group depart in two different step.
(a) First step : - Slow step involves ionisation to form carbocation
(b) Second step : Abstraction of proton
3.1.1 E1 Reaction of Alkyl halide :
Mechanism :
Step 1 :
Step 2 : + B – H
Characteristics of E1 reaction :
(i) It is unimolecular, two step process.
(ii) It is a first order reaction.
(iii) Reaction intermediate is carbocation, so rearrangment is possible
(iv) In the second step, a base abstracts a proton from the carbon atom adjacent to the carbocation,
and forms alkene.
(v) Kinetics Rate [Alkylhalide]
Rate = k [Alkylhalide]
(vi) Energetics The free energy diagram for the E1 reaction is similar to that for the SN1 reaction.
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
14
e.g.
Br|
CHCCHCH|CH
323
3
+
e.g.
3.1.2 E1 Reaction of AlcoholDehydration requires an acidic catalyst to protonate the hydroxyl group of the alcohol and convert it toa good leaving group. Loss of water, followed by loss of proton, gives the alkene. An equilibrium isestablished between reactants and products. For E1 mechanism reagents are (i) H
3PO
4/
(ii) H2SO
4/ 160º
OHH||CC||
acid CC||
+ H2O (Rearrangement may occur)
Mechanism :
Step 1 :
Step 2 : +
Step 3 +
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
15
RemarksIn first step, an acid-base reaction a proton is rapidly transferred from the acid to one of the unsharedelecton pairs of the alcohol.In second step the carbon oxygen bond breaks. The leaving group is molecule of water :Finally,in third step the carbocation transfers a proton to a molecule of water. The result is the formationof a hydronium ion and an alkene.
Reactivity of ROH : 3° > 2° > 1°
e.g.
e.g.
CH OH2
42SOH.Conc
CH2
( )I
Minor
+
e.g. H
bondof
MigrationH
3.1.3 E1 Reaction of Ether :Elimination is not a favourable reaction for ether, but however few reactions have been observed.E1 Elimination takes place via formation of stable carbocation.Ether undergoes dehydration reaction in the presence of conc. H
2SO
4/
e.g. /SOH.Conc 42
3.2 E2 Reaction :
3.2.1 E2 Reaction of Alkyl halide :Dehydrohalogenation is the elimination of a hydrogen and a halogen from an alkyl halide to form analkene. Dehydrohalogenation can take place by E1 and E2 mechanism.
Reagent
(i) Hot alcoholic solution of KOH or EtO¯ / EtOH (ii) NaNH2
(iii) t-BuO¯
K in t-BuOH
Mechanism :
+ BH
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
16
Characteristics of E2 reaction
1. This is a single step, bimolecular reaction
2. It is a second order reaction
3. Kinetics Rate [R – X] [Base]
Rate = k [R – X] [ ]
4. Rearrangment is not possible
5. For the lower energy of activation, transition state must be stable
6. E2 follows a concerted mechanism
7. The orientation of proton & leaving group should be antiperiplanar.
8. Here – H is eliminated by base hence called elimination
9. Positional orientation of elimination In most E1 and E2 eliminations where there are two or more
possible elimination products, the product with the most highly substituted double bond will predominate.
This rule is called the saytzeff or zaitsev rule.
Reactivity towards E2 R – I > R – Br > R – Cl > R – F
For example : Dehydrohalogenation of 2-bromo-2-methylbutane can yield two products.
e.g. +
e.g.
CH3
(major)
+
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
17
e.g.
Formation of the Hoffmann productBulky bases can also accomplish dehydrohalogenations that do not follow the saytzeff rule. Due tosteric hindrance, a bulky base abstracts a less hindered proton, often the one that leads to formationof the least highly substituted product, called the Hoffmann product.
HBrH|||CHCCCH
||CHH
23
3
+
+
Bulky base :
(CH CH ) N:3 2 3
Triethylamine
Stereospecific E2 reactionsThe E2 is stereospecific because it normally goes through an anti periplanar transition state. Theproducts are alkene.
HCH3H
Br
PhPh
H
CH3
H
Br
Ph
Ph
Base
=
Ph H
PhCH3
=Ph Ph
CH3
H
3.3 E1 cB Reaction (Unimolecular conjugate base reaction)
In the E1 cB, H leaves first and then the X. This is a two step process, the intermediate is a carbanion.
Mechanism :
Step 1 :
||XCC
||H
(conjugate base)
Step 2 : ||
–CC–
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
18
Remarks :First step consists of the removal of a proton, , by a base generating a carbanionIn second step carbanion looses a leaving group to form alkene
Condition : For the E1 cB, (i) substrate must be containing acidic hydrogens and (ii) poor leavinggroups.
e.g.
e.g. X2C = CF
2
e.g. R2C = O +
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
19
Q.1 In the given reaction, CH3CH
2– X + CH
3SNa
The fastest reaction occurs when ‘X’ is -(A) – OH (B) – F (C) – OCOCF
3(D) OCOCH
3
Ans. CSol. Leaving group ability Stability of anion
Q.2 Give the solvolysis products expected when each compound is heated in ethanol
(a) (b) (c) (d)
Ans. (a) (b) (c) (d)
Sol. (a)
(b)
(c)
(d)
Q.3 The rate of SN1 reaction is fastest with
(A) (B)
(C) (D)
Ans. (A)Sol. Rate of S
N1 reaction Stability of carbocation .
Miscellaneous Solved Examples
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
20
Q.4. Predict the compound in each pair that will undergo solvolysis (in aqueous ethanol) more rapidly.
Ans. (a) II > I (b) II > I (c) I > II (d) II > I
Sol. Rate of solvolysis Stability of carbocation .
Q.5
3
23
3
CH|
OHCHCCH|CH
IH? Write the structure of major product
Ans.
nemethylbuta-2-Iodo-2
|CH–CHCCH
|CH
323
3
I
Sol.
3
23
3
CH|
OHCHCCH|CH
IH
3
223
3
CH|
HOCHCCH|CH
323
3
CH–CHCCH|CH
nemethylbuta-2-Iodo-2
|CH–CHCCH
|CH
323
3
I
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
21
Q.6 3-Methyl-2-pentanol reacts with conc.HCl / ZnCl2
to gives 3-Chloro-3-methylpentane, explain themechanism.
Ans./Sol.
OH|
CHCHCHCHCH|CH
323
3
OH
H
2
323
3
CHCHCHCHCH|CH
3223
3
CHCHCCHCH|CH
Cl|
CHCHCCHCH|CH
3223
3
Q.7 Make distinction between following pairs of substances by using Lucas reagent.
Ans./Sol. Lucas
(a) (II) gives white turbidity immediately
(b) (II) gives white turbidity after 5-7 minute
(c) (II) gives white turbidity immediately
(d) (I) gives white turbidity after 5-7 minute
Q.8 What is the major product ?
+ HBr A (Major)
Ans.
Sol. + HBr
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
22
Q.9 Correct decreasing order of reactivity towards SN2 reaction
(I) CH3CH2 CH2Cl (II) CH3 CH2CH2Cl (III) CH3CH2CH2CH2Cl (IV) CH3CH2CH2
(A) IV > I > II > III (B) III > II > I > IV (C) IV > I > III > II (D) II > I > IV > IIIAns. B
Sol. As increases branching rate of SN2 reaction decreases.
Q.10 Write the structure of product ‘P’
OH
DMSO/KOH
2
P
Ans. Inversion of configuration is possible
Sol. It is a SN2 reaction.
Q.11 Give the decreasing order of reactivity of alkyl halides with MeO– (Williamsons synthesis).
)A(BrCCH)CH( 233
)B(CHCHClCH 22
)C(
CHCHClCH 322
)D(CHCHBrCH 322
Ans. B > D > C > ASol. It is a S
N2 reaction.
Q.12 Write mechanisms that account for the product of the following reaction :
NH2
– CH2
– CH2
– CH2
– CH2
– Br
Sol.
Q.13 products
What is the product of the given reaction ?
Ans. +
Sol. It is a SN2 reaction.
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
23
Q.14 Draw a fischer projection for the product of the following SN2 reaction
(a) Acetone/NaI (b)
Ans. (a) (b)
Sol. It is a SN2 reaction.
Q.15 To prepare t-butyl ethyl ether, t-butyl bromide and sodium ethoxide is not taken ; instead ethyl bromidesodium t-butoxide is taken. Explain.
Sol.
(CH3)
3CBr
2E
O2
CH3
CH
2SN (CH
3)
3COCH
2CH
3
Q.16
3
23
CH|CHOHCHCH A , Identify AA
Ans.
3
23
CH|CHOHCHCH
3
23
CH|CHBrCHCH
Sol. It is a SN2 reaction.
Q.17 Explain why ArOR ethers are cleaved to give RI and ArOH rather than ArI and ROH ?
Sol.
SN2 attack on a carbon of a benzene ring does not occur nor does the (phenyl) of high energy
form by an SN1 reaction. Hence, ArI can’t be form
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
24
Q.18 Which one of the following does not give white precipitate with aqueous silver nitrate solution?
(A) (B) CH2
= CH – Cl (C) CH2
= CH – CH2
– Cl (D)
Ans. A BSol. In (A) and (B) lone pair of Cl atom takes part in conjugation so partial double bond character between
C–Cl bond.
Q.19 Me3CCH
2OH + , Identify A and B
Ans. A : Me2C = CHMe
B :
Me|
MeCCHCH 22
Sol. Me3CCH
2OH Me
3C Me
2C = CHMe +
Me|
MeCCHCH 22
Q.20 Write the intermediate species as indicated :
3, 3 – dimethyl – 2 – butanol H .................. OH2 .................. Migration ..................
(A) (B) (C)
oxonium ion 20 carbocation 30 carbocation
-H+ -H+
.................... ....................
(E) (D)
Alkene Alkene
Sol. (A) = CH3 C CH CH3
CH3
CH3
OH2
+
(B) = CH3 C CH CH3
CH3
CH3
+(C) = CH3 C CH CH3
CH3
+
CH3
(D) =CH3 C C CH3
CH3 CH3
(E) =CH3 C CH CH2
CH3
CH3
Major product is (D)
Minor product is (E)
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

Prade
ep A
garw
al Aca
dem
y
( Tau
ght B
y IIT
ians..
...)
CHEMISTRY
25
Q.21 What are dehydration products of
Ans.
Sol. +
Q.22 Write the major product of the following reaction ,42SOH
Ans.
Sol. ,42SOH
Q.23 Predict the elimination products of the following reactions
(a) Sec. butyl bromide + (b) 2-Bromo-3-ethylpentane + MeONa
(c) 1-Bromo-2-methylcyclohexane + EtONa
Ans. (a) CH3
– CH = CH – CH3
(b) (c)
Sol. (a) It is an E2 reaction. (b) It is an E2 reaction. (c) It is an E2 reaction.
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220

CHEMISTRY
26
Q.24 major + minor
Write the structure of major and minor product.
Ans.
Sol. It is an E2 reaction.
Q.25 What are the various product due to loss of HBr from
Ans. ,
CH3
CH3
(minor)
,
Sol. It is an E2 reaction.
Q.26 What are the major products of dehydrohalogenation by alcoholic KOH of :
(i) CH3
– CH2
– CH2
– CH2
– CH2Cl (ii)
Cl|
CHCHCHCHCH 3223
(iii)
Cl|
CHCHCHCHCH 3223 (iv)
Cl
|
CHCCHCH
|
CH
323
3
(v)
3
33
CHCl
||
CHCHCHCH (vi)
33
33
CHCH||
CHCCHCH|
Cl
Ans. (i) gives CH3CH
2CH
2CH = CH
2, (ii) & (iii) CH
3CH
2CH = CHCH
3
(iv) & (v) (vi)
Sol. It is an E2 reaction.
Pradeep Agarwal Academy(Taught By IITians.....)
By: Anand Kumar Rai (IITian...) Mob:7840011220