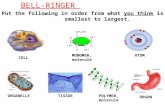
Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro-...
-
Upload
russell-norton -
Category
Documents
-
view
221 -
download
0
description
Transcript of Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro-...
![Page 1: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/1.jpg)
Unit 3 – Lecture 2
![Page 2: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/2.jpg)
Levels of Organization – reviewAtomMoleculeBiomolecule
[aka macro-molecule]
Organelle
CellTissueOrganOrgan systemOrganism
![Page 3: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/3.jpg)
MoleculesMolecule – composed of two or more atoms
can be made of a single element or multiple elements
has no overall chargeexample: H2 – two atoms of hydrogen
![Page 4: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/4.jpg)
Molecules – cont’dCompound – a molecule made of two or more
elementsmy abbreviation = cmpd(s)MUST have more than one element presentEx: H2O [2 atoms of hydrogen, 1 atom of
oxygen]ionic compounds have a positive or
negative chargeEx: NH4+
![Page 5: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/5.jpg)
Molecules – cont’dTypes of BondsCovalent Bonds
formed when atoms share valence electrons“co” – together valence electron – electron in the outermost
shell of an atoma strong bondEx: H2O
![Page 6: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/6.jpg)
Molecules – cont’dTypes of BondsIonic Bonds
formed when atoms gain or lose electrons by transfer
weaker than a covalent bondEx: NaCl
![Page 7: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/7.jpg)
Properties of WaterApproximately 2/3 of your body is water…so
apparently it’s quite important…
What makes water so important??
![Page 8: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/8.jpg)
Water is a Polar MoleculePolar Molecule: has no overall charge [hence…
“molecule”] but has a side that is more electronegative
electronegativity – the ability of an atom to attract electrons to itselfuneven distribution of electrons leads to a
negative pull on one side
![Page 9: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/9.jpg)
Water is a Polar MoleculeThe oxygen side of the molecule is much
more electronegative than the hydrogen side
![Page 10: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/10.jpg)
Water is a Polar MoleculeIt is the “Universal Solvent”
water given this term because it is able to dissolve many substancesthis is because of its polarity
solvent: a substance that is used to dissolve a solute.
solute: a substance that is dissolved by the solvent
Ex: water [solvent] dissolves sugar [solute]
![Page 11: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/11.jpg)
Water is a Polar MoleculePolar is aka Hydrophilic
hydrophilic: literally, “water-loving”“hydro” = water“philos / phil / philia” = to lovedissolve easily in water [like sugar]
hydrophobic: literally, “water- fearing”“phobia / phobic / phobos” = to fearaka non-polar do not dissolve or mix easily in water [like
lipids]
![Page 12: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/12.jpg)
Water Performs H-BondingHydrogen Bond: bond formed between a H
and a more electronegative atomis very weakis not a true “bond” but an attractionusually takes place between oxygen [O], nitrogen [N], or fluorine [F]
![Page 13: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/13.jpg)
Water Performs H-BondingAdhesion – the ability of water to form H-bonds
with other molecules [to “stick” to other things ]
think “adhesive”ex: straw in water
water climbs above the surface levelaka capillary action
major way blood flows through the capillaries in your body and through the roots of plants.
![Page 14: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/14.jpg)
Water Performs H-BondingCohesion – the ability of water to form H-bonds
with other water moleculescauses surface tensionEx: water drops form small spheres, can
overfill a glass, some bugs can walk on water
![Page 15: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/15.jpg)
Water Expands when Freezingis less dense when frozen; most substances are more denseallows it to float on water when frozenif ice sunk, lakes & ponds would freeze solid
in winter
![Page 16: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/16.jpg)
Water has a High Specific HeatSpecific Heat – amount of energy it takes for
the temperature of a substance to increase or decrease
makes homeostasis MUCH easier
![Page 17: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/17.jpg)
MixturesMixture – a combination of multiple substances
that…can be physically separatedare not combined chemicallycan be homogenous or heterogenous
![Page 18: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/18.jpg)
Mixtures – cont’dMixtures can be homogenous or heterogeneoushomogenous – appearance and consistency
are the same throughout “genous” – typetypically called solutions
heterogeneous – appearance & consistency are different throughoutEx: …?
![Page 19: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/19.jpg)
Mixtures – cont’dSolution – a mixture that…is homogenousis in a single phase
solid, liquid, or gascan have different phases dissolved in one
anothergas in liquid [carbonated water]solid in solid [alloys – carbon/iron = steel;
copper/zinc = brass]solid in liquid [sugar water]etc.
![Page 20: Unit 3 – Lecture 2. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://reader035.fdocuments.net/reader035/viewer/2022062504/5a4d1b587f8b9ab0599aa293/html5/thumbnails/20.jpg)
Mixtures – cont’duses the term “solute” & “solvent”solubility – the ability of a substance to
dissolve in another






![Unit 3 – Lecture 4. Levels of Organization – review Atom Molecule Biomolecule [aka macro- molecule] Organelle Cell Tissue Organ Organ system Organism.](https://static.fdocuments.net/doc/165x107/56649e455503460f94b394f3/unit-3-lecture-4-levels-of-organization-review-atom-molecule-biomolecule.jpg)