Title your next right side: Bonding Review Number your paper 1-22
-
Upload
daquan-neal -
Category
Documents
-
view
25 -
download
0
description
Transcript of Title your next right side: Bonding Review Number your paper 1-22

Title your next right side: Bonding Review
Number your paper 1-22
Go around the room and answer the questions about bonding. It does not matter which question you begin with.
Bonding Review

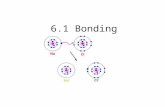
If lithium and oxygen bond together, what type of bond will
they form?
1.
Ionic1 metal + 1 nonmetal

What is the formula of dihydrogen monoxide?
2.
H O2
2 nonmetals = covalent
Prefixes

Draw the lewis dot structure of CS
2 3.
2 nonmetals = covalent
...
..
S C S.......
.. ..

What VSEPR shape does CS form?
24.
Linear






What type of bond do two nonmetals make?
5.
Covalent

What is the formula of carbon tetrachloride?
6.
CCl4

List the 7 elements that can form diatomic molecules.
7.
HydrogenFluorineNitrogenChlorineOxygen Bromine
Iodine

Write the formula of tin (II) sulfide.
8.
SnS
1 metal + 1 nonmetal = ionic
Swap & Drop
Sn S+2 -2

Write the formula of potassium nitrate.9.
K(NO )3
1 metal + 1 nonmetal = ionic
Swap & Drop
K (NO )+ -3

Do cations gain or lose
electrons?
10.
Lose electrons

The melting points of two substances are being compared.
Substance A melts at 1180°C. substance B melts at 120°C. Which
one is most likely an ionic compound?
11.
Sub. A

Write the name of FeO
12.
Iron (II) oxide
1 metal + 1 nonmetal = ionic
Reverse Swap & Drop

Name the following compound:
Li S2
13.
Lithium sulfide1 metal + 1 nonmetal = ionic

What VSEPR shape would this molecule form?
14.
Trigonal Pyramidal

Name the following compound:
NO3
15.
Nitrogen trioxide

Which is better at conducting electricity, ionic or covalent
compounds?
16.
Ionic

Which group on the periodic table has the electron configuration that all the other elements want? (this
group already fulfills the octet rule)
17.
Noble Gases

Why do molecules have the shapes they have? In other words, what is
the major idea behind VSEPR?
18.
Atoms & lone pairs of electrons want personal space

A powdery substance is stirred in water but doesn’t dissolve very
well. Is it most likely ionic or covalent?
19.
Covalent

What is unique about the physical structure of ionic
compounds?
20.
They form crystals

What type of bond is caused by a “sea of electrons”?
21.
Metallic

What VSEPR shape would this molecule form?
22.
Trigonal Planar
B
H
H
H