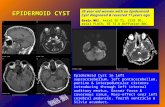
The structure carcinoids - Postgraduate Medical …carcinoid type, three (11%) cylindroid, one (3y.)...
Transcript of The structure carcinoids - Postgraduate Medical …carcinoid type, three (11%) cylindroid, one (3y.)...

Postgraduate Medical Journal (July 1977) 53, 360-363.
The cellular structure of bronchial carcinoids
CHARLES MARKSM.D., M.S., Ph.D., F.R.C.P., F.R.C.S., F.A.C.S.
J. LAMBERTYM.D.
Department of Surgery, L.S. U. Medical School, Charity Hospital andVeterans Administration Hospital, New Orleans
SummaryA histological and electronmicroscopic study ofbronchial carcinoids indicated that these tumours areargyrophilic in keeping with their occurrence in tissueof embryologically foregut origin. Normal bronchicontain cells that resemble, in their electronmicro-scopic content, intestinal Kultschitzky-type cells.
Characteristic of the carcinoid cell is the presenceof intracellular neurosecretory granules seen onelectronmicroscopy despite the apparent absence ofargentaffin granules under light microscopy.
IntroductionReisner (1928) and subsequently Kramer (1930)
suggested that bronchial adenomas arose from theductal epithelium of bronchial glands. Churchill(1937) as well as Womack and Graham (1938)considered their origin to be from residual embryonicpulmonary tissue derived from entodermal and meso-dermal elements. They explained the resemblance ofthe muco-epidermoid variety of bronchial adenomato mixed salivary gland tumours on this basis. Atthis time, they suggested that bronchial adenomas beconsidered malignant.
Willis (1940) postulated the probability that thesetumours arose from the mucous and mixed glands ofthe bronchial mucosa. Felton, Liebow and Lindskog(1953) held the view that the central and peripheralvarieties of bronchial adenomas had different origins.Although they concurred that proximal lesions arosefrom mucous glands, they believed that the periphe-ral tumours arose from the lining of the epithelium.The controversy that had been extant regarding theorigin and natural history of bronchial adenomasneeds to be viewed in the light that clear distinctionmust be made between the carcinoid type, the cylin-dromatous variety, and the muco-epidermoidtumour. The cylindroma also known as an adenoidcystic carcinoma is the most malignant of the threetypes, arising as it does from the basal or surfaceepithelium of the bronchial ducts. The term bron-chial adenoma, therefore, refers to three and possiblymore distinct neoplasms that differ structurally aswell as providing variations in their natural history.
Material and methodsDuring the period 1948-1973, twenty-eight
patients with bronchial adenomas were diagnosed atCharity Hospital and Veterans AdministrationHospital in New Orleans. The pathological classifi-cation of the adenomas were: twenty-four (86%/)carcinoid type, three (11%) cylindroid, one (3y.)muco-epidermoid tumour. A pathological study ofthe twenty-four patients with carcinoid bronchialtumours provided this basis for histological andelectronmicroscopic study of the carcinoid cell. Lightmicroscopic studies were carried out after stainingthe sections with haematoxylin and eosin. Tissue forelectronmicroscopic study was fixed in 4°o glutar-aldehyde and treated with 1% aqueous osmiumtetroxide. After dehydration by passage through aseries of alcohol it was embedded in Epon 812.Sections were appropriately cut, stained with leadcitrate and uranyl acetate and prepared for exami-nation.
ResultsThe bronchial carcinoid is a smooth, globoid
tumour that protrudes into a proximal bronchuswith its partial or complete occlusion (Fig. 1).Bronchoscopic examination generally discloses anintact mucous membrane without ulceration.Although the lesion is endobronchial it generallyextends beyond the bronchus and the extrabronchialcomponent may be large, even if the endobronchialcomponent is quite small. The cut surface of theneoplasm may vary from a pale pink, soft tumour toa tan-coloured lobulated firm lesion with its paren-chymal surface covered by a connective tissue cap-sule.
In the more invasive or frankly malignant bron-chial carcinoid the capsule is poorly defined. It isnoteworthy that a desmoplastic fibrous reaction ortissue necrosis is unusual in contrast to the frequencyof these features in carcinoids of the gastrointestinaltract.The carcinoid cell is typically uniform and small
with acidophilic cytoplasm demonstrable with hae-matoxylin and eosin stains. Although it is quite
copyright. on D
ecember 24, 2020 by guest. P
rotected byhttp://pm
j.bmj.com
/P
ostgrad Med J: first published as 10.1136/pgm
j.53.621.360 on 1 July 1977. Dow
nloaded from

Cellular structure of bronchial carcinoids 361
-w,
FIG. 1. Bronchial carcinoid tumour causing bronchial obstruction with atelectasisand bronchiectesis.
unusual to find nuclear mitoses or argentaffingranules, the nuclei contain finely stippled chroma-tin.The cells are arranged in sheets, strands, or
rounded masses resembling acini. The cell groups aresituated about sinusoidal vascular spaces which maybe so prominent as to suggest a haemangioma. Thisvascularity may lead to severe haemorrhage atbronchoscopic attempts to remove the endobronchialcomponent of the carcinoid.
Local invasiveness may be expressed by cellularpermeation of the capsule or by perivascular andperineural extension (Fig. 2). Despite this connota-tion of malignant potential, these cases are neverthe-less associated with prolonged survival. The presenceof tumour cells within bronchial or paratracheallymph nodes is not necessarily indicative of sub-sequent spread. Fragments of bone are occasionallynoted as a result of metaplasia in portions of theisolated bronchial cartilages.
Occasionally bulky cells are seen with granularor striated cytoplasm with small nuclei which con-tain a network of coarse chromatin. These oncocyticcells may be interspersed with carcinoid or transi-tional cells.
Histological criteria cannot be used to differentiatebetween benign and malignant tumours. A basis fordecision of malignancy needs to consider the degreeof local invasiveness, lymph node involvement, andmetastases.
ElectronmicroscopyCarcinoid tumour cells demonstrate a variable
ss s z F - iS-I
*.Xaw it
-a
* i.<e ~
-*. 4 <_ F.
FIG. 2. Histological appearance of bronchial carcinoidtumour with perineural and lymphatic invasion (H & Ex 90).
copyright. on D
ecember 24, 2020 by guest. P
rotected byhttp://pm
j.bmj.com
/P
ostgrad Med J: first published as 10.1136/pgm
j.53.621.360 on 1 July 1977. Dow
nloaded from

362 Charles Marks anid J. Lamberty
-,61_~~
r-
92 ~~~.i!o..;v ,i=~~ SK* | 9IEzni
FIG. 3. Electronmicroscopic study of five distinctcarcinoid tumour cells with intense nuclear proliferativeactivity. Electron dense granules surround lipofuscinvacuoles (uranyl acetate and lead citrate x 10 200).
degree of electron density with some cells demon-strating greater opacity than others. The resemblanceof the tumour cells to normal Kultschitzky cells isquite striking. The carcinoid cells possess villousprocesses up to 1 ,u in size which interdigitate withthose of adjacent cells. Although mitochondria arenumerous, the smooth endoplasmic reticulum isscanty though well developed (Fig. 3). Largedisternal spaces may be seen as a result of fusion ofseveral smaller cisternae. Aggregations of electrondense bodies may be seen, bound by limiting mem-branes of the phagolysosomal type. The lysosomalcharacter of these cells is confirmed by their highacid phosphatase content.
Characteristic of the carcinoid cell is the presenceof intracellular neurosecretory granules, 100-500 ,±in diameter, scattered throughout the cytoplasm(Fig. 4). Many granules are found directly beneaththe cell membrane and aggregations of the granulesare often found in the vicinity of the Golgi appara-tus. The granules are generally more numerous and
".,
S..,
*A O
'~~~~~0-~ ~ ~ ~ 10A
2.~~~~~~~~~N
FIG. 4. Electronmicroscopic study of carcinoid tumourdemonstrates the discrete arrangement of the electron-dense secretory granules. Note the lipofuscin vacuolewith surrounding granules (uranyl acetate and leadcitrate x 20400).
are more variable in size than in usual Kultschitzkycells.The electronmicroscopic examination of bronchial
carcinoids prepared by the Fontana silver impregna-tion technique of bulk fixed tissue demonstrates thatthe neurosecretory granules consist of dense aggre-gations of fine argentaffin granules. This is in con-trast to the fact that under light microscopy mostbronchial carcinoids do not contain demonstrableargentaffin granules, the majority of these cells beingargyrophilic. This apparent anomaly may depend onthe ability of the neurosecretory granules to storethis secretion with variable ability to reduce silvercompounds.
DiscussionAlthough most bronchial carcinoids fail to show
argentaffin granules under light microscopy, mostare argyrophilic. Williams and Sandler (1963) inclassifying carcinoid tumours distinguished three
copyright. on D
ecember 24, 2020 by guest. P
rotected byhttp://pm
j.bmj.com
/P
ostgrad Med J: first published as 10.1136/pgm
j.53.621.360 on 1 July 1977. Dow
nloaded from

Cellular structure of bronchial carcinoids 363
carcinoid groups, based on differing embryologicalderivations associated with varying biochemicalcharacteristics distinctive for each group: (1) foregutorigin: bronchus, stomach, duodenum, biliary tractand pancreas; (2) midgut origin: arising from themid-duodenum to mid-transverse colon: (3) hindgutorigin: descending colon and rectum.Although most bronchial carcinoids have a
benign clinical course with prolonged survival, evenin the absence of extirpative surgery, the tumourdoes on occasion transgress into normal restrictivebarriers with local invasiveness, infiltration of lymphnodes and haematogenous spread.When haematogenous spread does occur, the sites
of predilection are liver, adrenals, brain and bones.Although bone metastases are rare, when they dooccur, they are generally osteoblastic in nature.Pollard, Grainger and Fleming (1962) reported anunusual case of metastasizing bronchial adenomaassociated with the carcinoid syndrome and notedthe presence of numerous osteoblastic lesions.Toomey and Felson (1960) noted the similarity of theosteoblastic patterns of metastatic carcinoid lesionsresulting from primary tumours in both the gastro-intestinal tract and the bronchial tree. In a cumula-tive review of seventeen cases of bone metastasesfrom carcinoid tumours, the lesions were noted to beblastic in fourteen cases, mixed blastic and lytic intwo, and purely lytic in only one case.The infrequency of associated concomitant or
synchronous malignant disease in bronchial carci-noid may be contrasted with the frequent co-existence of evident adenocarcinoma in patients withcarcinoids of the gastrointestinal tract.Bensch et al. (1968) first demonstrated Kult-
schitzky-type cells in normal bronchi. Their ultra-structural electron microscopic study demonstratedcells in normal human bronchial mucosa that re-sembled the intestinal argentaffin cells. Gmelich,Benson and Liebow (1967) demonstrated that thesecells were incorporated between exocrine cellssituated within the limiting basement membrane andcould be identified by their containing small darkcytoplasmic granules. Similar cells were also foundbetween the columnar cells lining the peripheral
bronchioles. It is these Kultschitzky-type cells thatprobably provide origin for the peripheral carcinoidtumour. The cells are particularly prominent in thelarger bronchi, especially at sites of bronchial bi-furcation. The argentaffin cell was originally thoughtto be derived from entoderm, but is now generallyaccepted to have a neuroectodermal origin. Thesecells are probably derived from the neural crest andmigrate to the components ofthe foregut, representedby bronchus, stomach, biliary tract, pancreas, andproximal half of the duodenum; the midgut repre-sented by the intestine, from the mid-duodenum tothe mid-transverse colon, and the hindgut represent-ed by descending colon and rectum. The migrationcommences in the 12-week fetus, and occurs longbefore these cells mature to develop this silver reac-tion. The failure of some of these cells to develop thesilver reaction may represent an expression of bio-logical idiosyncrasy.
ReferencesBENSCH, K.G., CORRIN, G., PARIENTE, R. & SPENCER, H.
(1968) Oat cell carcinoma of the lung. Cancer, 22, 1163.CHURCHILL, E.D. (1937) Discussion of bronchial tumors of
Jackson, C.L. and Konzelmann, F.W. Journal of ThoracicSurgery, 6, 335.
FELTON, W.L., LIEBOW, A.A. & LINDSKOG, G.E. (1953)Peripheral and multiple bronchial adenomas. Cancer, 6,555.
GMELICH, J.T., BENSON, K.G. & LIEBOW, A.A. (1967) Cells ofKultschitzky type in bronchioles and their relation to theorigin of peripheral carcinoid tumors. Laboratory Investi-gation, 17, 88.
KRAMER, R. (1930) Adenoma of bronchus. Annals of Otclogy,Rhinology and Laryngology, 39, 689.
POLLARD, A., GRAINGER, R.G. & FLEMING, 0. (1962) Anunusual case of metastasizing bronchial adenoma asso-ciated with the carcinoid syndrome. Lancet, ii, 1084.
REISNER, D. (1928) Intrabronchial polypoid adenoma.Archives of Surgery, 16, 1201.
TOOMEY, F.B. & FELSON, B. (1960) Osteoblastic bone meta-stasis in gastrointestinal and bronchial carcinoids. AmericanJournal of Roentgenology, 83, 709.
WILLIAMS, E.D. & SANDLER, M.N. (1963) The classificationof carcinoid tumours. Lancet, i, 238.
WILLIS, R.A. (1940) Argentaffin carcinomata (carcinoids) ofthe small intestine. Medical Journal of Australia, 2, 400.
WOMACK, N.A. & GRAHAM, E.A. (1938) Mixed tumors of thelung: so-called bronchial or pulmonary adenomas.Archives of Pathology, 26, 165.
copyright. on D
ecember 24, 2020 by guest. P
rotected byhttp://pm
j.bmj.com
/P
ostgrad Med J: first published as 10.1136/pgm
j.53.621.360 on 1 July 1977. Dow
nloaded from