The Challenges and Pitfalls in Diagnosing or Misdiagnosing Tuberculosis: Are the Days of the TB Skin...
-
Upload
marcia-lucas -
Category
Documents
-
view
213 -
download
0
Transcript of The Challenges and Pitfalls in Diagnosing or Misdiagnosing Tuberculosis: Are the Days of the TB Skin...
The Challenges and Pitfalls in Diagnosing or Misdiagnosing
Tuberculosis: Are the Days of the TB Skin Test Over?
Theodore F. Them, MD, MS, PhD, MPH, FACOEM Chief, Section of Occupational
& Environmental MedicineGuthrie Clinic, Ltd
Sayre, Pennsylvania
Roy F. Chemaly, MD, MPH, FIDSA, FACPProfessor of Medicine
Director, Infection Control SectionDirector, Antimicrobial Stewardship Program
University of Texas – MD Anderson Cancer CenterHouston, Texas
Objectives
By the end of this presentation, the attendee should be able to:•Explain the consequences of misdiagnosing active or latent TB. •Describe the currently available tests and their place in TB diagnosis. •Recognize the limitations of different TB screening strategies. •Discuss how certain patient populations might benefit from the use of an IGRA in place of the TST for TB screening.
2
Tuberculosis Testing
Currently 2 methods are available for the detection of M. tuberculosis infection in the US:•Mantoux tuberculin skin test (TST)•Interferon-gamma release assays (IGRAs)• CDC guidelines1 allow the use of IGRAs or the TST for screening healthcare workers
3
Issues Affecting TST Utility1
• False-Positive Results– Foreign-born persons (BCG vaccinated) account for 64.6% of all TB
cases in US– Exposure to other mycobacteria– Unknown cause
• False-Negative Results– Immunosuppression
• AIDS• Cancer• Anti-TNF• Transplant
• Noncompliance– Failure to return for TST interpretation
41. Centers for Disease Control and Prevention. MMWR. 2014.
Tuberculin Skin Test (TST) vsInterferon-Gamma Release Assays (IGRAs)
TST•2 visits required (minimum)•Method: injection into skin•Results affected by BCG•Results in 48−72 hours•Subjective results•Can cause booster phenomenon
IGRAs•1 visit required•Method: blood draw•Results not affected by BCG•Next-day results•Objective results•Does not cause booster phenomenon
5
Commercially Available IGRAsQuantiFERON®-TB Gold1
•ELISA technology•Measures IFN-γ release•“One and done”•PI sensitivity: 88.2%•PI specificity: 99.1%•“Real-world” specificity: 98%−98.9%3,4
•3 specialized tubes•Provides qualitative results•Sample stability: 16 hours•No FDA-approved borderline category•Can be run in hospital lab•Available nationally through reference laboratories (eg, Quest)
The T-SPOT®.TB Test2
•ELISPOT technology•Enumerates effector T cells•“One and done”•PI sensitivity: 95.6%•PI specificity: 97.1%•“Real-world” specificity: 98.9%−99.1%5,6
•1 standard tube•Provides quantitative and qualitative results•FDA-approved borderline category•Sample stability: 32 hours•Can be run in hospital lab•Available nationally through Oxford Diagnostic Laboratories®
1. QuantiFERON-TB Gold Package Insert. Cellestis, Inc. Valencia, CA. Doc. No. US05990301L, March 2013.2. T-SPOT.TB Package Insert. Marlborough, MA: Oxford Immunotec; 2010. 3. Schablon A, et al. BMC Infect Dis. 2011;(11):245-252. 4. Pai M, et al. Ann Intern Med. 2008;149(3):177-184.
5. Wang SH, et al. Scand J Infect Dis. 2010;42(11-12):845-850. 6. Bienek DR, et al. Int J Tuberc Lung Dis. 2009;13(11):1416-1421.T-SPOT and Oxford Diagnostic Laboratories are registered trademarks of Oxford Immunotec, Ltd.QuantiFERON is a registered trademark of Cellestis, Inc. 6
Blood Collection for QFT® Testing1
• Collection tubes include: – Nil control (grey cap)– TB antigen (red cap)– Mitogen control (purple cap)
• Tubes require shaking (10 times each) to mix blood with antigens coated on the inside of the tubes, but too much shaking could cause aberrant results.
• Blood in collection tubes must be incubated for 16−24 hours at 37°C within 16 hours of collection.2,3
1. QuantiFERON-TB Gold Package Insert. Cellestis, Inc. Valencia, CA. Doc. No. US05990301L, March 2013.2. Herrera V, et al. J Clin Microbiol. 2010;48(8):2672-2676. 3. Doberne D, et al. J Clin Microbiol. 2011;49(8):3061-3064.QFT is a registered trademark of Cellestis, Inc. 8
Blood Collection for T-SPOT®.TB1
• Standard phlebotomy techniques• Uses a standard lithium or sodium heparin tube• Less sensitive to preanalytical variables than QFT– Time from collection to analysis– No specialized tubes needed– No specific order of draw– No shaking of tubes– No incubation required – Specimens maintained at room temperature
for up to 32 hours
1. T-SPOT.TB Package Insert. Marlborough, MA: Oxford Immunotec; 2013.T-SPOT is a registered trademark of Oxford Immunotec, Ltd. 10
Consideration of IGRA Logistics
1. QuantiFERON-TB Gold Package Insert. Cellestis, Inc. Valencia, CA. Doc. No. US05990301L, March 2013.2. T-SPOT.TB Package Insert. Marlborough, MA: Oxford Immunotec; 2010.T-SPOT is a registered trademark of Oxford Immunotec, Ltd. QuantiFERON is a registered trademark of Cellestis, Inc.
Phlebotomy Steps QuantiFERON®-TB Gold1 T-SPOT®.TB Test2
Collection tubes 3 specialized tubes Standard tube
Tubes drawn in specific order
Required; Nil, TB antigen, mitogen N/A
Blood volume 1 mL (0.8−1.2 mL); under- or overfilling outside the 0.8- to 1.2-mL range may lead to erroneous results
Fill 6-mL tube
Shake collection tubes Required; shake the tubes up and down 10 times
Not required
Purge tube with butterfly Required when a butterfly needle is used
Not required
Sample stability Specimens must be incubated as soon as possible but within 16 hours
Up to 32 hours
11
Targeted TB Testing• Close contacts of persons known or suspected to have TB
disease• Foreign-born from areas of high incidence TB disease (Latin
America, Africa, Asia, Eastern Europe, and Russia)
• Persons who travel to areas with a high prevalence of TB disease
• Residents and employees of high-risk congregate settings (correctional facilities, long-term care facilities, homeless shelters)
• Healthcare workers who serve clients or patients who are at increased risk for TB disease
• Specific populations defined locally as having increased incidence of latent M-TB (possibly including medically underserved, low-income populations, or persons who abuse drugs or alcohol)
13
14
• 9588 new TB cases in US (case rate of 3.0)
• TB rate among foreign-born (FB) was 13x higher than US-born
• 4 states account for one-half of all reported cases: – California– Texas – New York – Florida
TB in the US: 2012 Data1
1. Centers for Disease Control and Prevention. www.cdc.gov/mmwr/preview/mmwrhtml/mm6211a2.htm?s_cid=mm6211a2_e.
TB in the US: 2013 Data¹TB population statistics: •54.2% of 6172 FB persons with TB originated from Mexico, Philippines, India, Vietnam, and China •6.8% reported as HIV+•5.7% reported being homeless in past year•3.9% confined to correctional facility at time of diagnosis•Every 100 contacts not treated will lead to 3 new cases of TB in 1−2 years.1
1. Centers for Disease Control and Prevention. Trends in Tuberculosis – United States, 2013. MMWR. 2014.
Screening TB Contacts: Acceptance of Diagnosis1
• The results of this study: Grinsdale J, et al. Int J Tuberc Lung Dis. 2011;15(12):1614–1619.
• Contacts tested with QFT were more likely to complete evaluation (64% vs 56%).
• Infected contacts started (89% vs 72%) and completed (70% vs 53%) LTBI treatment more often in group tested with QFT.
• Positive QFT results, but not positive TST results, correlated with the intensity, proximity, and duration of TB exposure.
1. .16
Case Study• 41-year-old male Indian nurse who started work at a Cancer Center
September 2012. Was referred with a diagnosis of pneumonia. • PMHx: February 2012, he started having a cough with off and on
sputum production. He was diagnosed with bronchitis and received 2 courses of azithromycin with no improvement.
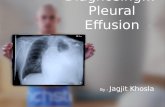
• Referred to a pulmonologist had chest x-ray done in March 2012 and a PPD skin test, which were both negative ; received cefuroxime and later on Levaquin with no improvement.
• At that point, he was diagnosed with possible asthma, and was started on inhaled steroids.
• His cough got a little bit better initially and started to get worse again.
Case Study
• December 2012: Worsening cough and hemoptysis now.
• Saw another pulmonologist in February 2013 for a second opinion. No chest x-ray was done. Was prescribed another course of an antibiotic with no improvement.
Case Study• Past Social History: – Born and raised in India. He immigrated to Canada in
2005 and then moved to the US few years later. – Working since September 2012 as a nurse on the
Stem Cell Transplant Units. – Exposure to an active case of TB 35 years ago when he
was a child (his uncle had active TB). – The patient had multiple PPD skin tests, which were
negative. He received the BCG vaccine as well. – He is married and he has 2 kids (9-month-old and 6-
year-old).
Case Study
• May 2013: He went back to India for a vacation. He saw an internist who did a Ziehl-Neelsen stain on 3 sputum specimens, which came back positive for AFB. His chest x-ray showed bilateral infiltrates.
• After return to the USA had a positive Quantiferon -TB test done.
Issues Affecting LTBI Diagnosis1
• LTBI is difficult to diagnose in immunocompromised patients.
• Risk for progression to active TB is greater in immunocompromised patients.
• No gold standard exists to diagnose LTBI.– TST performs particularly poorly in
immunocompromised patients because of an increased likelihood for false-negative results.
1. Redelman-Sidi G, et al. Am J Respir Crit Care Med. 2013;188(4):422-431.
Advantages of IGRAs
• Results are numerical and thus less subject to reader bias.
• No need for a follow-up visit for reading of results.
• Not affected by BCG vaccination status as they use TB-specific antigens that are not present in BCG.
• Whatever test for TB you use, understand the variables that affect test results, so that you can manage them and get the most accurate results possible.