Spindle cell rhabdomyosarcoma of the tongue in an infant: a case report with emphasis on...
-
Upload
anita-gupta -
Category
Documents
-
view
225 -
download
5
Transcript of Spindle cell rhabdomyosarcoma of the tongue in an infant: a case report with emphasis on...

Pathology – Research and Practice 200 (2004) 537–543
ARTICLE IN PRESS
*Correspondin
E-mail addres
pmchou@northw
0344-0338/$ - see
doi:10.1016/j.prp
www.elsevier.de/prp
TEACHING CASE
Spindle cell rhabdomyosarcoma of the tongue in an infant: a case report
with emphasis on differential diagnosis of childhood spindle cell lesions
Anita Guptab, John Maddalozzoa, Thanda Win Htinb, Anil Shaha, Pauline M. Choub,*aDepartment of Ear Nose and Throat, Children’s Memorial Hospital, Northwestern University, Chicago, IL 60614, USAbDepartment of Pathology, Children’s Memorial Hospital, 2300 Children’s Plaza, Northwestern University,
Chicago, IL 60614, USA
Received 23 October 2002; accepted 26 March 2004
Abstract
Malignant tumors are extremely uncommon in infants, specifically in the head and neck region. We present a three-day-old infant with a large, polypoid, soft tissue mass arising from the floor of the mouth. Histologically, thisneoplasm consisted of hypercellular and myxoid areas. A mixture of poorly oriented, small, undifferentiated,hyperchromatic, and round to elongate spindle cells was seen. A high degree of striated muscle differentiation waspresent, along with areas marked by a herringbone pattern, as well as hemangiopericytic vessels and rare mitosis.Immunohistochemical examinations revealed strong nuclear staining for myogenin and diffuse cytoplasmic stainingfor desmin and muscle-specific actin (HHF-35). The tumor did not stain for S-100. Based on histologic results andimmunostains, this lesion was diagnosed as spindle cell rhabdomyosarcoma. This type of lesion involving the tongue israrely seen in females, neither in association with a herringbone pattern nor with hemangiopericytic vessels.Furthermore, rare benign and malignant spindle lesions, such as cellular fibromatosis, fetal rhabdomyoma, infantilehemangiopericytoma, infantile rhabdomyofibrosarcoma, and infantile fibrosarcoma, should be in the differentialdiagnosis and excluded.r 2004 Published by Elsevier GmbH.
Keywords: Rhabdomyosarcoma; Rhabdomyofibrosarcoma; Spindle cell lesions; Tongue; Childhood tumors
Introduction
It is important to distinguish between benign andmalignant spindle cell tumors, particularly in the headand neck region where complete resection may not befeasible due to location. Pathologists should be aware ofsubtle differences in the histologic features of spindle celllesions, as they may show morphologically similarpatterns. The differential diagnosis for a spindle cellneoplasm should include both benign and malignantfibroblastic, myofibroblastic, neurogenic, and various
g author. Tel.: 773-880-4438; fax: 773-880-8127.
ses: [email protected],
estern.edu (P.M. Chou).
front matter r 2004 Published by Elsevier GmbH.
.2004.04.001
rhabdomyoblastic lesions as seen in childhood. In thiscase report, we present a polypoid, large, fleshy pink-red, soft congenital mass lesion arising from the base ofthe tongue and floor of the mouth of a three-day-oldbaby girl, using radiological, histological and immuno-histochemical findings, along with a review of variousdifferential diagnoses.
Case report
A three-day-old, full term, neonate female wasevaluated for a congenital lesion involving the rightbase of the tongue and floor of the mouth (Fig. 1).

ARTICLE IN PRESS
Fig. 1. Infant with a large, polypoid mass arising in the base of tongue and floor of the mouth.
A. Gupta et al. / Pathology – Research and Practice 200 (2004) 537–543538
Prenatal ultrasound did not reveal any abnormalities.She was born with Apgar score of 9 and 9 and had noairway distress or feeding difficulties. Examinationrevealed a 5� 3 cm2 firm, mobile mass not fixed to anysurrounding structures. A CT scan was performed whichrevealed a soft tissue mass localized to the anterior floorof mouth and anterior 2/3 of the tongue without anycontrast enhancement (Fig. 2). She was afebrile with anormal CBC, and, otherwise, asymptomatic. The slowgrowing mass was first biopsied and then re-excised twoweeks later through a transoral approach, preserving thelingual nerve. The mass was submucosal and grosslyappeared to be completely excised at the conclusion ofthe case. Frozen section suggested a malignancy due tothe finding of mitotic figures and hyperchromaticpleomorphic cells.Gross pathological findings revealed a 4.5� 3.0� 1.0
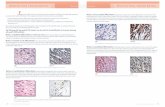
red-pink fleshy, polypoid, soft tissue mass. Cut sectionsrevealed an unencapsulated, tan-white to tan-pinkmulti-lobulated surface. Microscopy on low powerrevealed a varying degree of alternating, densely packed,hypercellular, and loosely textured myxoid areas
(Fig. 3a). Parallel fascicles or a herringbone pattern(Fig. 3b), along with numerous stag horn or angularvessels, were seen. Higher magnification revealed amixture of poorly oriented, small, round to oval,undifferentiated cells with hyperchromatic nuclei andelongated strap- or tadpole-shaped spindle cells witheosinophilic cytoplasm, and eccentrically placed nuclei(Fig. 3c). The lesion was infiltrating into the adjacentskeletal muscle (tongue) and was present at the marginof resection. The mitotic count ranged from 0 to 5 per 10high power fields.Immunohistochemical studies revealed a strong, uni-
form, nuclear stain for myogenin (Fig. 4, inset) using themonoclonal myf-4 antibody (Ventana, Tucson, Arizona)and diffuse cytoplasmic stain for desmin (Ventana) andmuscle-specific actin (HHF-35)(Ventana) (Fig. 4). Thetumor is negative for S-100 (Ventana). Tumor sent forcytogenetic study did not show any aberrant cytogeneticabnormality. The Intergroup RhabdomyosarcomaStudy, which reviewed the biopsy, concurred with ourdiagnosis. The oncology service was consulted, and anadditional operation was performed in an attempt to

ARTICLE IN PRESS
Fig. 2. CT scan showing a mass localized to the anterior floor of mouth and anterior 2/3 of tongue without any contrast
enhancement.
A. Gupta et al. / Pathology – Research and Practice 200 (2004) 537–543 539

ARTICLE IN PRESS
Fig. 3. (a) Light microscopy revealed a varying degree of
alternating densely packed, hypercellular spindle cells and
loosely textured myxoid areas (H&E). (b) and (c) Parallel
fascicles or a herringbone pattern along with elongated strap-
or tadpole-shaped eosinophilic rhabdomyoblasts with cross-
striations (H&E).
A. Gupta et al. / Pathology – Research and Practice 200 (2004) 537–543540
achieve tumor-free margins prior to chemotherapy. Thesurgical margins from the second surgery were negativefor tumor, but a random biopsy of the anterior 2/3 of
the tongue revealed microscopic disease involvement.The patient began a VAC (vincristine, actinomycin, andCytoxan) chemotherapy regimen two weeks aftersurgery. The patient had no sequelae postoperatively,including stridor or aspiration, and resumed bottle-feeding on postoperative day three. Three monthspost-surgery, a small residual tumor was noted and re-excised. Histologically, the tumor was identical to theprimary lesion at presentation. The patient will receivechemotherapy for one year and is likely not to receiveradiation because of age.
Discussion
Based on a spindle cell lesion with hemangiopericyticvessels as seen in this tumor, several benign andmalignant tumors peculiar to infancy and childhoodshould be considered. These tumors are rare and haveno clinical or morphological correlations in adults. Themost common tumor in consideration of location withinthis age group is a myofibroblastic tumor, solitarymyofibroma, or infantile myofibromatosis. These aregenerally non-aggressive lesions, unless they have multi-focal visceral involvement. Our case lacks the classicdiagnostic histologic feature in solitary myofibroma,namely the specific ‘‘zonation’’ with alternating hyper-cellular and hypocellular areas with plump myxoidspindle cells. Typically, these tumors also lack nuclearatypia. The central hypercellular region with hemangio-pericytoma-like vascular pattern suggests, to someauthors, that this tumor is a variant of infantilehemangiopericytoma, possibly at different stages ofmaturation [6,22].In general, fibromatosis is a major consideration in
our differential diagnosis, specifically gingival fibroma-tosis. Notably, up to 8% of gingival fibromatosis ispresent at birth, particularly the familial form. Thecellular nature of our tumor resembles infantile fibro-matosis, a counterpart of the desmoid fibromatosis inadults. These tumors show a predilection for lower jawand neck and may be infiltrative and destructive. Suchlesions are found at birth and are slightly more commonin boys. Although most (lesions) originate in the skeletalmuscles, the head and neck region is also a common site,including involvement of the tongue [21,8]. Although theinfiltrative borders of our case closely resemble suchtumors histologically, gingival fibromatosis is composedof bland fibroblasts that vary little in size withnormochromatic nuclei associated with a small tomoderate amount of collagen. Mitoses are seen, butare rarely abundant [8].The distinction from fetal rhabdomyoma, another
rare benign tumor, may be problematic as this tumornot only has a predilection for the head and neck region

ARTICLE IN PRESS
Fig. 4. Immunohistochemical studies revealed a strong, uniform, nuclear stain for myogenin using the monoclonal myf-4 antibody
(inset), and diffuse cytoplasmic stain for muscle-specific actin.
A. Gupta et al. / Pathology – Research and Practice 200 (2004) 537–543 541
(including the oral cavity), but can also be histologicallyindistinguishable [3,5,10]. The tumor, however, is fairlywell circumscribed, superficially located, and histologi-cally composed of muscle cells which vary little in sizeand shape, with no mitotic activity. Malignant trans-formation of cellular fetal rhabdomyoma has beenreported in the tongue of an 18-month-old infant aswell [11].The herringbone pattern and cellularity of the lesion
discussed in our case is a feature of several malignantspindle cell tumors uncommon in this location [19], suchas infantile rhabdomyofibrosarcoma, spindle cell rhab-domyosarcoma, and infantile fibrosarcoma. Infantilefibrosarcoma occurs primarily in the extremities but canbe seen in the head and neck region, and cases have beenreported at birth [17]. Microscopically, our tumorresembles infantile fibrosarcoma, as they both haveuniform, solidly packed spindle cells arranged in afascicular or herring-bone pattern with hemangioper-icytic vascular pattern [7]. Undoubtedly, without anyancillary studies, it would be extremely difficult to rule
out this diagnosis. However, one feature that is ofsignificance in our tumor is the presence of strap cellswith pleomorphic nuclei. Immunohistochemical stainswere helpful, as myogenin and MyoD1 do not stain ininfantile fibrosarcoma [2,12]. If available, cytogeneticand electron microscopic studies would be immenselyuseful in difficult cases [9].The next tumor (on the list) is not only rare, but also
controversial in existence. This so-called ‘‘Infantilerhabdomyofibrosarcoma’’ was first reported in 1993[15], with less than ten cases in the present literature.At the time of this report, infantile rhabdomyofibro-sarcoma appeared to be histologically similar to spindle-cellrhabdomyosarcoma, but may possibly [1,16] representan earlier form of rhabdomyogenic tumor. Anothertheory is that this tumor is merely an intermediate formbetween infantile fibrosarcoma and spindle cell rhabdo-myosarcoma. However, there is a substantial differencein their cytogenetic aberrations, although no recurringabnormality has yet been reported in this rare tumor. Atthe present time, the main difference lies in their

ARTICLE IN PRESSA. Gupta et al. / Pathology – Research and Practice 200 (2004) 537–543542
anatomical distribution, degree of myogenic differentia-tion, and prognosis [1,14,15]. Although this tumor hasbeen reported in the retroperitoneum, intrathoracic [16],and buttock, it is yet to be reported in the oral cavity.Lastly, spindle cell rhabdomyosarcoma is an uncom-
mon tumor primarily seen in the paratesticular region[13] and frequently found in the head and neck [14].Patients with spindle cell RMS of non-paratesticularsites usually have more extensive disease compared withpatients having paratesticular lesions, and two-thirds ofthese reported gross residual tumor after surgery ormetastatic tumor at diagnosis [13,14]. Notably, it isimportant to distinguish entrapped muscle fibres fromtumor cells. The presence of strap cells with distincteosinophilic cytoplasm, eccentric nuclei, and cross-striations suggests rhabdomyoblastic differentiation [4].Immunohistochemical studies are an integral part of
the workup of pediatric tumors. Many markers arecurrently available, some work better with moredifferentiated muscles than with others. As transcriptionfactors are the earliest markers of myogenesis, these arenaturally the most sensitive and specific markersavailable, even for the most primitive, poorlydifferentiated rhabdomyosarcoma [2,12]. Of these,myogenin has been reported to be highly specific and,therefore, considered the most reliable marker formyogenesis to date [12,20]. Although other tumors,such as ectomesenchymomas, nephroblastomas withmuscle differentiation, including the so-called ‘‘infantilerhabdomyofibrosarcoma’’, may show positivity, Kumaret al. claimed that myogenin is 100% specific forrhabdomyosarcoma [12]. Based on the evidence ofspindle cell lesion with a high degree of muscledifferentiation, cross-striations, and nuclear stainingfor myogenin, our tumor would best be diagnosed andclassified as an embryonal rhabdomyosarcoma, spindlecell subtype [18].This case report underlines the difficulty that pediatric
pathologists face in the investigation of spindle celltumors in infants. As different tumors carry differenttreatment protocols and prognosis, it is important tosecure adequate material for ancillary studies in suchcases. Nowadays, with the availability of molecularpathology studies, contemporary workup mandates theneed for securing appropriate tissue for such studies [9].Needless to say, this case illustrates the need to work ina multidisciplinary approach with emphasis on theprerequisite for pediatric pathologists to be familiarwith rare pediatric tumors having a spindle cellcomponent.
References
[1] A.O. Cavazzana, D. Schmidt, V. Ninfo, D. Harms, M.
Tollot, M. Carli, J. Treuner, R. Betto, G. Salviati, Spindle
cell rhabdomyosarcoma—a prognostically favorable var-
iant of rhabdomyosarcoma, Am. J. Surg. Pathol. 16 (3)
(1992) 229–235.
[2] M.H. Cessna, H. Zhou, S.L. Perkins, S.R. Tripp, L.
Layfield, C. Daines, C.M. Coffin, Are myogenin and
myo-D1 expression specific for rhabdomyosarcoma? A
study of 150 cases with emphasis on spindle cell mimics,
Am. J. Surg. Pathol. 25 (9) (2001) 1150–1157.
[3] E.B. Chung, Pitfalls in diagnosing soft tissue tumors in
infancy and childhood, Pathol. Ann. 20 (1985) 323–386.
[4] M.J. Cunningham, E.N. Myers, C.D. Bluestone, Malig-
nant tumors of the head and neck in children: a twenty-
year review, Int. J. Pediatr. Otolaryngol. 13 (1987) 279–292.
[5] L.P. Dehner, F.M. Enzinger, R.L. Font, Fetal rhabdo-
myoma. An analysis of nine cases, Cancer 30 (1) (1972)
160–166.
[6] M. Dictor, A. Elner, T. Andersson, et al., Myofibroma-
tosis like hemangiopericytoma metastasizing as differen-
tiated vascular smooth muscle and myosarcoma:
myopericytes as a subset of ‘‘myofibroblasts’’, Am.
J. Surg. Pathol. 16 (1992) 1239.
[7] F.M. Enzinger, S.W. Weiss, Soft tissue tumors, in: S.W.
Weiss, J.R. Goldglum (Eds.), Rhabdomyosarcoma, 4th
ed., Mosby Inc, Missouri, 2001, pp. 785–835.
[8] C.D. Fletcher, Distinctive soft tissue tumors of the head
and neck, Mod. Pathol. 15 (3) (2002) 324–330.
[9] D.A. Hill, M.J. O’Sullivan, X. Zhu, R.T. Vollmer, P.A.
Humphrey, L.P. Dehner, et al., Practical application of
molecular genetic testing as an aid to the surgical
pathologic diagnosis of sarcomas: a prospective study,
Am. J. Surg. Pathol. 26 (8) (2002) 965–977.
[10] S.B. Kapadia, J.M. Meis, D.M. Frisman, G.L. Ellis, D.K.
Heffner, Fetal rhabdomyoma of the head and neck: a
clinicopathologic and immunophenotypic study of 24
cases, Human Pathol. 24 (7) (1993) 754–765.
[11] R. Kodet, J. Fajstavr, Z. Kabelka, et al., Is fetal cellular
rhabdomyoma an entity or a differentiated rhabdomyo-
sarcoma? A study of patients with rhabdomyoma of the
tongue and sarcoma of the tongue enrolled in the
intergroup rhabdomyosarcoma studies I, II and III,
Cancer 67 (1991) 2907.
[12] S. Kumar, E. Perlman, C.A. Harris, M. Raffeld, M.
Tsokos, Myogenin is a specific marker for rhabdomyo-
sarcomas: an imunohistochemical study in paraffin—
embedded tissues, Mod. Pathol. 13 (9) (2000) 988–993.
[13] I. Leuschner, Spindle cell rhabdomyosarcoma: histologi-
cal variant of embryonal rhabdomyosarcoma with
association to favorable prognosis, Curr. Top. Pathol.
89 (1995) 261–272.
[14] I. Leuschner, W.A. Newton Jr., D. Schmidt, N. Sachs, L.
Asmar, A. Hamoudi, D. Harms, H.M. Maurer, Spindle
cell variants of embryonal rhabdomyosarcoma in para-
testicular region, A report of the intergroup rhabdomyo-
ma Study, Am. J. Surg. Pathol. 17 (3) (1993) 221–230.
[15] L. Lundgren, L. Angervall, G. Stenman, L.-G. Kindblom,
Infantile Rhabdomyofibrosarcoma: a high-grade sarcoma
distinguishable from infantile fibrosarcoma and rhabdo-
myosarcoma, Human Pathol. 24 (1993) 785–795.
[16] T. Mentzel, H.J. Mentzel, D. Katenkamp, Infantile
rhabdomyofibrosarcoma. An aggressive tumour in the

ARTICLE IN PRESSA. Gupta et al. / Pathology – Research and Practice 200 (2004) 537–543 543
spectrum of spindle-cell lesions of childhood, Pathologe
17 (1996) 296–300.
[17] H. Miki, S. Kobayashi, Y. Kushida, M. Aasaki, A case of
infantile fibrosarcoma with immunohistochemical, elec-
tron microscopic and genetic analyses, Human Pathol. 30
(1999) 1519–1522.
[18] D.M. Parham, Pathological classification of rhabdomyo-
sarcomas and correlations of molecular studies, Modern
Pathol. 14 (5) (2001) 506–514.
[19] M.W. Richardson, W.K. Funkhouser, B. Senior, B.W.
Weston, Spindle cell lesions of the head and neck
mimicking rhabdomyosarcoma in children, Pediatr. He-
matol. Oncol. 19 (2) (2002) 129–134.
[20] J. Rosai, Soft tissues, in: J. Rosai (Ed.), Ackerman’s
Surgical Pathology, 8th Edition, Mosby-Year book Inc,
Missouri, 1996, pp. 2080–2087.
[21] M. Takagi, H. Yamamoto, H. Mega, et al., Heterogeneity
in the gingival fibromatosis, Cancer 68 (1991) 2202.
[22] S. Variend, N.M.A. Bax, J. Van Gorp, Are infantile
myofibromatosis, congenital fibrosarcoma and congenital
hemangiopericytoma histogenetically related? Histo-
pathology 26 (1995) 57.