Serial Sectioning for 3D Imaging under Scanning Electron Microscopy · 2018. 12. 19. · ii Serial...
Transcript of Serial Sectioning for 3D Imaging under Scanning Electron Microscopy · 2018. 12. 19. · ii Serial...

Serial Sectioning for 3D Imaging under Scanning Electron Microscopy
by
Devin K. Luu
A thesis submitted in conformity with the requirements for the degree of Master of Applied Science
Graduate Department of Mechanical and Industrial Engineering University of Toronto
© Copyright by Devin K. Luu 2016

ii
Serial Sectioning for 3D imaging under Scanning Electron Microscopy
Devin K. Luu
Master of Applied Science
Graduate Department of Mechanical and Industrial Engineering
University of Toronto
2016
Abstract
Serial block face scanning electron microscopy (SBFSEM) is a powerful method that
can create 3D reconstructions of large volumes for morphology studies in micro or nano
biological or materials science compared to conventional ultramicrotomy. It allows
visualization of long, dense networked structures with nanoscopic resolutions. Despite
its utility, the adoption rate is low because installing state-of-the-art systems require
extensive modifications to the SEM chamber due to their large size.
This thesis presents a prototype for a plug-and-play SBFSEM system using specially
designed compact micro and nanopositioners that can be placed inside the SEM with no
modifications to the SEM. Through iterations of empirical calculations, computer
simulations, and prototyping, an ultramicrotome capable of cutting thin, uniform
sections was designed. The function of the system was validated by sectioning and
reconstructing a copper TEM grid of known dimensions. The reconstruction was accurate
to within 1% of original dimensions.

iii
Acknowledgments
Firstly, I would like to thank Professor Yu Sun for inviting me to work in his lab and
introducing me to a new field different from any of my previous work. My experience
with his lab group has influenced my career path for the years to come. Along the same
line, I would like to thank my aunt, Caroline Cao, for stimulating my interest in
engineering and research since childhood.
My project on in-SEM ultramicrotome design was challenging, but with the help of
Douglas Holmyard and Ren Li, who showed me some of the basics of ultramicrotomy, I
learned a lot that would have otherwise taken me much longer to learn on my own.
I would also like to thank all lab members for their help during these two years. Jun
Chen’s knowledge of electronics and his circuit designs gave me a lot of flexibility for
running my system. I would also like to mention Wesley Johnson, Zhensong Xu, Tae Won
Ha and Jun Liu, with whom I formed close relationships.
I received a lot of help from undergraduate summer students. Special thanks to
Tiffany Shan for her assistance in the meticulous set-up of my experiments and to Benn
Xu for performing the time-consuming procedures.
Most importantly, I would like to thank Brandon Chen for always being behind me.
His constant motivation gave me the courage to push ahead in my project during my
many moments of uncertainty. Thank you for your confidence in me and in my work. I
look forward to working with you in the future.
Finally, I would like to thank some people from home in Vancouver for being there
almost around the clock just to chat and take my mind off of work. Also thanks to my
parents and brother not only for their love, but their reminders to take care of my health.

iv
Table of Contents
Contents
Acknowledgments ...................................................................................................................... iii
Table of Contents ........................................................................................................................ iv
List of Tables ............................................................................................................................... vii
List of Figures ............................................................................................................................ viii
Chapter 1 Introduction .................................................................................................................1
Introduction ..............................................................................................................................1
1.1 Background ........................................................................................................................1
1.2 Motivation ..........................................................................................................................3
1.3 Dissertation Outline ..........................................................................................................4
Chapter 2 Ultramicrotomy in SEM ............................................................................................5
Ultramicrotomy in SEM ..........................................................................................................5
2.1 The SEM Working Environment ....................................................................................5
2.2 Ultramicrotome Function ................................................................................................8
2.3 Cutting in Ultramicrotomy ..............................................................................................9
2.3.1 Tool material and geometry ..............................................................................10
2.3.2 Cutting parameters .............................................................................................11
2.3.3 Sample material ...................................................................................................12
2.4 Existing SBFSEM Systems ..............................................................................................12
2.5 Design of a Plug-and-Play SBFSEM System ...............................................................13
2.6 The Latest Generation Design .......................................................................................15

v
2.6.1 Mechanical description ......................................................................................16
2.6.2 Electrical and software description ..................................................................17
2.7 Conclusion .......................................................................................................................19
Chapter 3 Flexure Based Piezoelectric Actuator ....................................................................20
Flexure Based Piezoelectric Actuator ..................................................................................20
3.1 Introduction .....................................................................................................................20
3.2 Design Considerations for a Flexure Based Piezoelectric Actuator ........................23
3.3 Solving the Out-of-plane Stiffness Problem ................................................................26
3.4 The Designed Piezoelectric Flexure Actuator .............................................................26
3.5 Discussion of Alternative Designs ................................................................................29
Chapter 4 Thermal V-Beam Actuator ......................................................................................30
Thermal V-Beam Actuator ....................................................................................................30
4.1 Introduction .....................................................................................................................30
4.2 Design Considerations of a Thermal V-Beam Actuator ............................................32
4.3 The Designed Thermal V-Beam Actuator ...................................................................34
4.4 Discussion of Previous Specimen Actuator Designs .................................................36
Chapter 5 Sectioning using the SBFSEM System ...................................................................37
Sectioning using SBFSEM System........................................................................................37
5.1 Specimen Height Control for Sectioning .....................................................................37
5.2 Minimum Slice Thickness ..............................................................................................38
5.3 Cutting Surface Roughness ...........................................................................................38
5.4 Cutting Parameters Affecting Surface Roughness .....................................................40

vi
5.4.1 Knife speed has little effect on chatter .............................................................41
5.4.2 Chatter increases as slice thickness increases .................................................41
5.5 Cutting Force Estimation for Future Design Guideline ............................................42
5.6 Oscillating Knife: A Method to Reduce Cutting Force ..............................................43
5.7 3D Reconstruction Accuracy Validation .....................................................................46
Chapter 6 Conclusion and Outlook .........................................................................................50
Conclusion and Outlook .......................................................................................................50
6.1 Contributions ...................................................................................................................51
6.2 Future Directions ............................................................................................................51
References ...................................................................................................................................53

vii
List of Tables
Table 1.1: Some discoveries made using SBFSEM and the volumes reconstructed ........... 3
Table 2.1: Comparison of different actuators. Criteria are listed left to right from most to
least important. ........................................................................................................................... 14
Table 2.2: Specification requirements for a new SBFSEM system ...................................... 15
Table 3.1: Monitored inputs and outputs from simulation are used to check against
specifications ............................................................................................................................... 25
Table 5.1: 3D reconstruction is accurate within 1% for length measurements. ................ 48

viii
List of Figures
Figure 1.1: SBFSEM not only allows automated ultramicrotomy, but it also eliminates
section retrieval, which is considered to be the most difficult step. ..................................... 2
Figure 2.1: In a scanning electron microscope, an electron gun fires electrons that are
focused and directed by a series of lenses and deflectors to perform a raster scan across a
sample. Secondary electrons released from the sample are detected by the detector to
form an image on a video display. ............................................................................................ 6
Figure 2.2: SBFSEM process. A) The knife and specimen are in position and ready to cut.
B) The specimen is cut and the slice is discarded. C) The block face surface is scanned by
SEM. D) The specimen is moved into position for the next cut. ........................................... 9
Figure 2.3: Besides the knife edge, the three angles, rake angle, bevel angle, and clearance
angle play a large role in determining cutting force, and by extension, cutting surface
roughness (chatter) and compression. In general, larger a larger rake angle will give lower
force, but bevel angle and clearance angle need to be balanced to maintain knife longevity
and to prevent rubbing of the knife against the surface, respectively. .............................. 11
Figure 2.4: The ultramicrotome's actuators are mounted on a base plate. A knife is
connected to the knife actuator and the specimen is mounted on the specimen actuator.
....................................................................................................................................................... 17
Figure 2.5: The electronics for the SBFSEM ultramicrotome. The specimen thermal
actuator is run by the bench top power supply on the left and the knife piezo actuator is
run by the amplifier on the right. The amplifier is controlled via serial interface with a
PC. ................................................................................................................................................ 18

ix
Figure 2.6: A GUI on a PC gives commands to a piezo amplifier for the knife actuator
using serial commands. The interface allows knife speed and knife extension amount to
be set, as well as monitoring of messages. ............................................................................. 19
Figure 3.1: Each solid line represents the set of possible force-displacement operation
points for a given voltage. To maximize the amount of work done by a piezo stack, the
load on the stack should be such that the operation points will lie upon the dashed
maximum work line. ................................................................................................................. 21
Figure 3.2: (Left) Parallel four-bar linkage type flexure actuator. Amplification ratio
depends mostly on the length L, where larger L gives larger amplification ratio. (Right)
Sagittal type flexure actuator. Amplification ratio depends mostly on angle α, where
smaller α gives larger amplification ratio. With the same size envelope, it is easier to
obtain a higher displacement ratio on a sagittal type flexure than a four-bar linkage
flexure. ......................................................................................................................................... 22
Figure 3.3: An example output of an FEA simulation of a piezoelectric flexure mechanism.
The four quantities indicated in the figure should be used as shown in Table 3.1 to
compare with specifications. .................................................................................................... 25
Figure 3.4: (Top) The flexure actuator has a final motion range of 740 µm. (Bottom) There
is a hysteresis effect that is evident on the retraction stroke of the actuator but it is ignored
because no cutting is done on retraction. The extension stroke matches well with
simulation results (less than 5% error).................................................................................... 28
Figure 4.1: In the thermal v-beam actuator, heat is applied, causing thermal expansion of
the flexures. Due to the geometry, the actuator tip will be pushed upwards, as shown

x
with exaggerated displacement. Given a maximum size for the actuator, the displacement
of the tip depends on the thickness and angle θ of the beam. ............................................ 31
Figure 4.2: An example output of an FEA simulation of a thermal v-beam actuator. The
four quantities indicated in the figure should be used to compare with specifications. . 33
Figure 4.3: (Left) For a steady input voltage, after the actuator has reached steady state,
the actuator follows a quadratic voltage-displacement characteristic. The actuator has a
range of 310 µm. (Right) The specimen actuator follows a first-order transient
characteristic with a time constant of 5.23 min and a settling time of 20.5 min inside the
SEM. Knowing this, for any step input voltage to the actuator, it is possible to calculate
the displacement of the actuator at any time. ........................................................................ 35
Figure 4.4: The designed v-beam thermal actuator has a polished, reamed, hole for
accepting a resistive cartridge heater. Flexure beams expand against the bracing, forcing
the actuator tip upwards. .......................................................................................................... 36
Figure 5.1: Timings for five cuts in 20.5 min, with a 500 nm step input. ........................... 37
Figure 5.2: (Top left and right) Optical micrographs of a rough (A) and smooth (B) block
face. The cutting direction is from top to bottom. The rough block face was cut using an
older revision of the ultramicrotome which was not as stiff and severe chatter occurred.
(Bottom) The AFM line scans of the sample show that the RMS of the smooth surface is
substantially lower than the RMS of the rough surface. ...................................................... 39
Figure 5.3: (Left) There is little to no effect of knife speed on chatter. Statistical analysis
shows a low correlation value of -0.45 between knife speed and chatter. (Right) As section
thickness is increased, little chatter until a section thickness of 500 nm, when the
roughness RMS rapidly rises. ................................................................................................... 40

xi
Figure 5.4: (Top left) A piezoelectric stack is attached to a knife with a counter weight to
oscillate the knife. (Top right) Knife apparent angle, caused by knife oscillation combined
with the knife feed, is smaller than the knife bevel angle. (Bottom left) Block face after
slicing off 400 nm at a frequency of 7 kHz. (Bottom right) Vibrating glass knife becomes
damaged easily, causing severe chatter. The left side of the blade is more damaged than
the right side. .............................................................................................................................. 44
Figure 5.5: (Top left) Individual SEM images were taken, then processed and aligned to
form a stack. (Top right) A TEM grid was reconstructed using SBFSEM with a voxel size
of 66 x 66 x 100 nm. The grid spacing as measured from the 3D reconstruction by our
SBFSEM system is matches with manufacturer specifications. Shaded in areas show the
first section. Outlined areas show debris and discontinuities, as labelled. (Bottom)
Reference TEM grid with dimensions. ................................................................................... 47

1
Chapter 1 Introduction
Introduction
1.1 Background
Serial block face scanning electron microscopy (SBFSEM) is a method combining
conventional ultramicrotomy and scanning electron microscope (SEM) imaging [1].
Ultramicrotomy is an important technique used in life [2, 3] and material sciences [4-6] to
study morphology at the nanometer scale by taking successive thin slices from a sample
[7, 8]. These samples can be imaged and stacked using computer imaging algorithms to
reconstruct a 3D computer model of the sample. This technique can be used to resolve
interior structures that cannot be seen otherwise. Conventional ultramicrotomy is
performed manually by skilled operators and involves several complex steps to cut from
a resin-embedded sample thin slices less than 100 nm in thickness [9]. After the slices are
cut, the operator must painstakingly retrieve each individual section from a small
container of water attached to the knife. Finally, each slice is placed on a transmission
electron microscope (TEM) grid and imaged. Each step must be carefully performed to
ensure that slices do not get lost or wrinkled in the process.
Taking advantage of the large available space inside an SEM, Denk and Horstmann
[1] developed the first working SBFSEM system using custom designed components and
precision actuators tailored for use inside SEM. Their system was capable of
automatically obtaining images from large blocks of tissue. Crucially, their system
eliminated the manual slicing and retrieval steps, which most heavily depend on operator
skill (Figure 1.1). By performing the process under SEM, instead of collecting slices to
image, the slices are discarded and the remaining block face of the specimen is imaged.
The system could automatically section and image specimens unattended, acquiring vast
amounts of data.

2
Owing to these capabilities, many breakthroughs in biological sciences are being
made. Most notably, the morphology of relatively large volumes of tissue or those
containing long, fibrous networks with nanometer-sized features such as neural tissue
[10-18], or muscles [19-21] can now be visualized in detail, giving clues to their functions
and mechanisms. This technology also shows usefulness in other areas of cell biology [22-
28] and materials science [29-36]. Table 1.1 presents a few of these cases and the volumes
reconstructed.
Despite SBFSEM’s capabilities and accomplishments thus far however, the adoption
rate of this new technology is hindered by some factors: Current commercially available
systems are only compatible with certain models of SEM, or require heavy modification
to other SEMs [37, 38]. The cost of a new SEM or extensive modifications is prohibitive to
most microscopy and biology labs. After modification, it may be difficult or even
Figure 1.1: SBFSEM not only allows automated ultramicrotomy, but it also
eliminates section retrieval, which is considered to be the most difficult step.

3
impossible to use the SEM for any other imaging tasks. The cause of this is mostly because
existing systems are large and fill the entire SEM chamber.
The development of other SEM nanorobotic manipulation platforms in more recent
years, such as in sensing, actuation, and automation [39-43], also provides some
technology that could be used in developing a compact SBFSEM system.
Table 1.1: Some discoveries made using SBFSEM and the volumes reconstructed
Category Summary of discovery/application Volume
Reconstructed
Reference
Neuroscience Postsynaptic membrane of most used
synaptic connections are found to be
reinforced with use
80 x 80 x 59.9 µm3 [10]
Synaptic pruning does not proceed
normally in absence of bone
morphogenetic proteins
180000 µm3 [11]
Cell biology A previously unknown ridge-like structure
on podocytes was discovered
2000000 µm3 [22]
Organization of collagen extracellular
matrix is analyzed by SBFSEM
1000 µm depth [19]
Materials
Science
Micro and nanostructures of engineered
materials such as paper and other
composites are reconstructed
60 x 60 x 20 µm3 [29]
1.2 Motivation
Although SBFSEM ultramicrotomes are available commercially, they are large, and
are only available for specific models of SEM. In addition, numerous modifications must
be made to the SEM itself to accommodate the ultramicrotome [37]. Because most electron
microscopy facilities are shared between multiple users, to make modifications to a
microscope would cause great inconvenience for the users. Our goal is to develop a
prototype plug and play SBFSEM ultramicrotome that can be used in any SEM to perform
3D reconstruction of samples.

4
This ultramicrotome will be substantially smaller than other ultramicrotomes on the
market [37]. To accomplish this, existing designs should be studied to understand why
they are so large, and then redesign to work around those limitations. The small actuators
used in other SEM nanomanipulation tasks should be studied and adapted to the new
design. Literature will be reviewed to set specifications for a new ultramicrotome, such
as size, minimum volume able to reconstruct, and other motion requirements. Specific
objectives include:
Study current systems and establish new design specifications.
Design and characterize a new prototype SBFSEM ultramicrotome.
Verify the operation of the designed SBFSEM ultramicrotome by sectioning and
reconstructing structures of known morphology.
1.3 Dissertation Outline
An overview of the following chapters is as follows: Chapter 2 describes the process
of ultramicrotomy, and the integration of that process into SEM. It will also describe the
design challenges and of a compact ultramicrotome that can be used in any SEM. A large
part of the design process involved the development of a piezoelectric flexure and
thermal buckling beam actuator whose designs and characterized performances are
discussed in Chapter 3 and 4, respectively. Chapter 5 presents a demonstration of the
capabilities of the designed ultramicrotome to verify its function. Chapter 6 concludes
the thesis, giving a summary of the thesis, a list of key contribution of this research, and
a list of suggested future directions.

5
Chapter 2 Ultramicrotomy in SEM
Ultramicrotomy in SEM
To design an ultramicrotome for SEM the SEM working environment and
ultramicrotome form and function should be well understood. This section will give an
introduction to the SEM function and ultramicrotome function, then lay out some
guidelines and specifications that will assist in designing ultramicrotomes in SEM, and
other types of designs for SEM in general. Then, it will introduce the designed
ultramicrotome in a general sense.
2.1 The SEM Working Environment
The inside of a scanning electron microscope chamber is kept at high vacuum and
ideally free from outside electric and magnetic fields. Deviations from ideal imaging
conditions can cause artifacts, noise, image warping, or unwanted reflections. Stray
electric and magnetic fields act as lenses to bend the path of electrons. Air remaining in
the chamber will impede the path of electrons, making the signal-to- noise ratio weak.
Hydrocarbons either from outgassing or other contamination like fingerprints will be
deposited onto the specimen by electron beam induced deposition (EBID) [44], obscuring
the specimen. Any defects in the image will interfere with the fidelity of the final 3D
reconstruction [45].
Figure 2.1 depicts the SEM imaging process. When imaging inside SEM, an electron
beam focused by electromagnetic lenses is raster scanned across a specimen surface.
When the electron beam hits the sample, the electrons’ interactions with the sample
produces signals that can be detected by sensors inside the microscope chamber to reveal
information about the sample such as topography or material composition depending on
imaging mode [46].

6
Secondary electron imaging is used to visualize the topography of the sample. When
an incident electron beam hits a spot on the sample surface, secondary electrons are
emitted from the sample, which are then detected by the secondary electron detector.
Backscatter electron imaging is used to visualize composition information of the sample.
When an incident electron beam hits a spot on the sample surface, electrons are slung
back from the specimen. The number of electrons slung back depends on the atomic
number of the area of the specimen, with higher atomic numbers slinging back more
electrons [46].
Figure 2.1: In a scanning electron microscope, an electron gun fires electrons that are focused and
directed by a series of lenses and deflectors to perform a raster scan across a sample. Secondary
electrons released from the sample are detected by the detector to form an image on a video display.

7
Knowing the basic operation of the SEM and about factors that affect its operation,
here are a few material selection and handling factors that all designs for SEM should
consider:
1. Outgassing – Certain plastics will have gas pockets trapped within during
manufacturing. The released gases may interfere with electron paths, and in the
worst case, can be deposited on specimen surfaces via EBID [45]. Aluminum alloys
and 300 series stainless steels are common choices for metals, while PTFE, PEEK,
and Kapton are common choices for plastics used in SEM. Special lubricants and
adhesives are also manufactured to be low outgassing. Similarly, before entering
the SEM chamber, all materials should be thoroughly cleaned to remove
contamination such as fingerprints. A comprehensive list of material outgassing
properties can be found at [47].
2. Magnetic fields – Magnetic materials may magnetize and bend the path of
electrons, causing distortion in the image. Metals used that exhibit little or no
magnetism are 300 series stainless steels and aluminum alloys. In the case that
magnetic materials must be used, effort should be spent on placing these materials
as far away as possible from the imaging region of interest.
3. Specimen charging – Non-conductive materials will “hold on” to electrons as the
electron beam is scanned over them, causing distortions/bright spots in image.
Conductive materials provide a path for excess electrons to leave the sample to
ground. Ideally, any specimens under SEM should have a conductive path to
ground, and any non-conductive materials, like with magnetic materials, should
be placed as far away from the imaging area of interest as possible.
Also important is that although the SEM chamber is large, the instruments and
detectors within are extremely delicate and sensitive. Any specimens or machines that

8
are put inside the SEM must fit well, such that they are secure and do not have a risk of
coming loose and falling and hitting any SEM emitters or detectors.
In SBFSEM, to accommodate the non-conductive resin-embedded sample, a special
variable pressure or low vacuum mode, which introduces small amounts of gas into the
chamber is used to whisk away excess charge. BSE imaging is used to obtain large image
contrast between different materials in the block face surface [1].
2.2 Ultramicrotome Function
A conventional ultramicrotome is a large desktop machine, incorporating a
microscope a cantilever arm, a specimen holder, a glass or diamond knife, and a knife
holder [48]. In conventional ultramicrotomy, the specimen is mounted on a cantilever
arm which moves the specimen face across a stationary knife edge when a crank wheel
is turned, which is held on the knife holder stage [49]. To take successive cuts, a heating
element on the cantilever arm causes the arm to thermally expand, advancing the
specimen towards the knife by a small amount [49]. Each part must be mounted as rigidly
as possible to withstand cutting forces to ensure uniform, thin slices of sample [50]. After
cutting, the slice floats away on the surface of water in a container attached to the knife
for retrieval and imaging in TEM.
The process of SBFSEM is shown in Figure 2.2 and is also introduced in [1]. Besides
being automated, the key mechanical difference between the conventional
ultramicrotome and SBFSEM is that instead of the specimen both advancing and
providing a slicing motion, it only advances toward the knife. The knife now provides
the cutting motion itself. The reason for this is to keep the specimen block face still with
respect to the SEM image frame. By advancing the specimen each time instead of moving
the knife to a different level, the focal plane of the specimen block face will also stay the

9
same. This allows the sliced section to be discarded, while imaging the remaining block
face.
2.3 Cutting in Ultramicrotomy
The sectioning process in ultramicrotomy is a cutting process comparable to
orthogonal cutting in traditional machining. There are a few factors in ultramicrotomy
that will affect the quality of the cut, including the material and geometry of the tool, the
cutting parameters, and the material [9, 50]. In ultramicrotomy, some concerns are chatter
and compression [50]. Chatter manifests as marks running perpendicular to the cutting
directions along the block face, and causes sections to come off which are uneven in
thickness. Compression occurs when the section coming off the block face is thicker than
the original depth of cut. A cut which requires low force to make results in lower
compression [51, 52], and a rigid setup results in lower chatter [50, 53]. In SBFSEM, where
we are only observing the block face, compression is not a big concern, but chatter will
still decrease the accuracy of the reconstructed volume.
Figure 2.2: SBFSEM process. A) The knife and specimen are in position and ready to cut. B) The
specimen is cut and the slice is discarded. C) The block face surface is scanned by SEM. D) The specimen
is moved into position for the next cut.

10
2.3.1 Tool material and geometry
The tool is affected by its material and geometry. In ultramicrotomy, metal knives
such as razor blades are not used because they become blunt too quickly. Instead, glass
and diamond knives, which also have very small edge radii are used. Compared to
diamond knives, glass knives have a slightly larger edge radius, their edge is not as
straight, and they are not as durable. Furthermore, glass knife edges’ sharpness and
straightness will degrade over time [9].
The geometry of the knife is also important to the cutting quality [54]. The rake angle,
the bevel angle, and the clearance angle are three important parameters to consider about
ultramicrotome knives [55]. The three of these angles add up to a right angle, as shown
in Figure 2.3. Because they play a large role in cutting force, their role should be
considered in the overall ultramicrotome design to mitigate chatter, which arises when
the structure is not stiff enough to withstand the cutting forces. High forces also
contribute to compression, which is that the slice after cutting is thicker than the slice
before cutting.
When looking at a 2D cross section of the knife and specimen, the rake angle is the
angle made by the top of the knife and the line normal to the specimen. A smaller rake
angle gives smaller cutting force. The bevel angle is the angle of the knife itself. Although
a smaller bevel angle can help increase the rake angle, a bevel angle that is too small will
cause the knife to be very fragile. The clearance angle is the angle made by the bottom of
the knife and the specimen, and its purpose is to prevent rubbing of the bottom of the
knife with the specimen. Diamond knives typically have a smaller bevel angle, allowing
the rake angle to be larger [52, 56]. A glass knife, having a larger bevel angle is more
forgiving against mistakes during setup.

11
2.3.2 Cutting parameters
Cutting parameters that affect the cut quality are the width of cut, depth of cut, and
speed of cut. The cutting force is proportional to the width of the cut [55]. The depth of
cut also affects the cutting force [55]. The thicker the depth, the more force it takes to curl
the cut section over the edge of the knife as it moves over the block face [55]. In extreme
cases, this can cause parts of the sample to tear off or it will break the knife [9]. Cutting
speed that is too high can result in excess chatter because any vibrations resulting from
the knife engaging the sample do not have enough time to damp out before the knive
begins to move across the block face [9]. Cutting too slow can also potentially cause the
Figure 2.3: Besides the knife edge, the three angles, rake angle, bevel angle, and
clearance angle play a large role in determining cutting force, and by extension, cutting
surface roughness (chatter) and compression. In general, larger a larger rake angle will
give lower force, but bevel angle and clearance angle need to be balanced to maintain
knife longevity and to prevent rubbing of the knife against the surface, respectively.

12
resin to stick to the edge of the knife [57], temporarily making the knife edge effectively
less sharp.
2.3.3 Sample material
Different embedding media, as well as the specimen embedded inside the embedding
media, exhibit different sectioning properties. For example, the hardness, stiffness, and
the material’s microstructure can affect cutting quality. Also very important is the
specimen preparation, including whether or not the embedding medium has been
properly cured and whether it has fully penetrated the specimen embedded inside [58].
2.4 Existing SBFSEM Systems
There are two main commercial systems for performing SBFSEM on the market: Gatan
3View2 [37] and FEI Teneo VS [38]. Although both products have been demonstrated to
be useful, they are currently compatible only with select models of SEM, or only after
heavy modification to the SEM.
The Gatan 3View2 system [37] is based on the work in [1]. The mechanism is based
on a series of cams and motors, requiring the use of multiple motion transfer components,
making the system large. Therefore, to heavy modifications are required to integrate the
3View2 into the SEM. The microtome is mounted permanently directly onto the SEM
door.
The FEI Teneo VS [38] is a different system utilizing a technique called multi-energy
deconvolution [59] to be able to potentially resolve thinner slices than a knife can
physically cut by using the electron beam to penetrate deeper than the surface of the
sample, then subtracting the surface information from the signal. However, any
variations in the material in the block face can potentially make the image slices uneven.
This ultramicrotome is only available to be fitted in one specially made SEM because it
also uses large mechanical components.

13
In conclusion, the main factor limiting the commercial SBFSEM systems is that they
are too large because they use a series of large mechanical components to transfer motion.
After SEM modification and installation, it may be difficult or even impossible to use the
SEM for other imaging tasks.
2.5 Design of a Plug-and-Play SBFSEM System
A SBFSEM ultramicrotome needs to be able to hold a knife and move the knife edge
over a cutting plane. It also needs to be able to hold a specimen tightly and to move the
specimen surface into the knife cutting plane. The described motions need to be high
resolution, smooth, and repeatable. The structure must be able to withstand cutting
forces, meaning the structure must be stiff. All the components used in its construction
must also be vacuum compatible.
There are a few additional challenges in designing a compact SBFSEM system. The
greatest challenge in developing a miniaturized system is that a smaller system will have
lower stiffness. In a system like an ultramicrotome, where not only nanometer
positioning is required, but the positioners also need to withstand cutting forces, even a
deflection of a several nanometers is crucial. Insufficient stiffness will result in
inconsistent slice thicknesses and also chatter knife marks.
Although conventional mechanical components such as motors, bearings, and gears
can achieve high stiffness and high speed, they will not be used to generate the
ultramicrotome motions because they are too large and there is risk of mechanical play.
This requirement rules out other piezoelectric actuators such as inchworm, walking, or
stick-slip mechanisms, which essentially use piezoelectric elements to generate pushing
motions along a bearing rail. The requirement against magnetic fields will rule out
magnetic actuators. Instead, flexure based actuators driven by piezoelectric stacks or
thermal expansion, which by nature have a certain amount of compliance to generate

14
required motions, will be used, adding to the stiffness challenge. Great care is needed to
balance the stiffness and motion requirements. A summary of different actuator types is
shown in Table 2.1.
Table 2.1: Comparison of different actuators. Criteria are listed left to right from most to least important.
Actuator
Type
Requirements Other metrics
Non-magnetic Repeatable Motion
range
Stiffness Score
Motors/gears FAIL FAIL
Magnetic FAIL Pass
Piezo walk /
slip stick
Pass FAIL
MEMS Pass Pass -- - -3
Piezo stack Pass Pass -- + -1
Piezo
flexure
Pass Pass DATUM 0
Thermal
flexure
Pass Pass - + 0
The ultramicrotome will use a glass knife due to availability and cost. Although glass
knives are inferior to diamond knives in almost every aspect in sectioning, the cost of a
glass knife is much lower, making it suitable for prototyping. The higher bevel angle
(~50°) of a glass knife also makes it less fragile, making it more forgiving to mistakes
during setup. For now, the SEM to be used with this design is the Hitachi SU3500, which
was picked because of availability.
To quantify the above functional requirements, the end performance target
specifications were set as follows in Table 2.2. The maximum system size was set to be
the maximum size a system can be and still be safely integrated into the SEM. Based on
the sizes of the reconstructions shown in Table 1.1, the maximum sample block face size
and advance distance were set to be able to cover the volumes reconstructed in
neuroscience and materials science. The knife actuator range was set to allow the entire

15
block face to be cut. The minimum slice thickness for this system is set near the minimum
thickness typically cut with glass knives on a conventional ultramicrotome [9].
Table 2.2: Specification requirements for a new SBFSEM system
Aspect Our target specification
System size < 127 x 127 x 90 mm3
Block face size < 350 x 350 µm2
Sample advance
distance
> 250 µm
Knife travel distance > 700 µm
Slice thickness for glass
knife
< 100 nm
The ultramicrotome underwent several iterations before arriving at the latest design.
The first versions used exclusively piezoelectric actuators due to ease of control, and were
focused on generating the correct motions. However, the initial designs of the
piezoelectric actuators were not stiff enough, causing sever chatter while cutting. The
latest generation focused on maximizing the stiffness of the system to achieve less chatter
while sectioning.
2.6 The Latest Generation Design
In the latest design iteration, keeping in mind the requirements for the system, and
especially trying to preserve the stiffness of the small system, the following system was
designed. The most drastic change from previous iterations was that the specimen
actuator was changed from a piezoelectric actuator to a thermal actuator, sacrificing ease
of control and speed for stiffness. The aspects that most affected the design of this system
was the actuator design, and they will be discussed in further detail in later chapters.

16
2.6.1 Mechanical description
Most structural mechanical parts are custom machined from 6061 aluminum alloy or
304 stainless steel, which are non-magnetic. All actuators are mounted on a common
baseplate. Any parts that may need to slide along each other are manufactured out of
different materials to eliminate any possibility of cold welding inside the SEM vacuum
chamber.
As shown in Figure 2.4, a piezoelectric knife actuator, which performs a slicing motion
by pushing the knife horizontally, is attached to the glass knife holder which holds a knife
with a clearance angle of 6° to prevent rubbing of the knife bottom while cutting [9]. A
thermal specimen actuator, which moves the specimen vertically into position for cutting
before each slice, is attached to the specimen holder. The specimen holder is separated
from the thermal actuator by a piece of Mycalex MM400, which insulates the specimen
from the heat generated by the specimen actuator. All electrical connections for the
actuators are routed to one connector which will be fed outside the SEM chamber via a
specially designed SEM feedthrough port flange. All wires and piezoelectric actuators are
shielded to avoid noise in the SEM image. Wire jackets are made of PTFE to avoid
outgassing.

17
2.6.2 Electrical and software description
The piezoelectric knife actuator is powered by an amplifier custom-designed to run
piezoelectric actuators. The amplifier was designed by Jun Chen (PhD candidate). The
thermal actuator is controlled simply by a B&K Precision 9110 bench-top power supply.
The two power sources are shown in Figure 2.5. Both actuators are controlled in open
loop after careful characterization within the SEM chamber.
Figure 2.4: The ultramicrotome's actuators are mounted on a base plate. A knife is connected to
the knife actuator and the specimen is mounted on the specimen actuator.

18
The microtome can be controlled from a GUI that interfaces with the amplifier
through USB, shown in Figure 2.6. The interface provides a way to control the speed and
travel of the knife as well as a way to monitor the status of the ultramicrotome while it is
moving.
Figure 2.5: The electronics for the SBFSEM ultramicrotome. The specimen thermal actuator is run by
the bench top power supply on the left and the knife piezo actuator is run by the amplifier on the right.
The amplifier is controlled via serial interface with a PC.

19
2.7 Conclusion
In summary, a system was designed and manufactured to target the specifications
laid out for the compact ultramicrotome system. The specifications of the designed
system were able to slightly exceed their targets.
The following sections will present a more in-depth discussion of the design and
characteristics of the main components and actuators of the system.
Figure 2.6: A GUI on a PC gives commands to a piezo amplifier for the knife actuator using
serial commands. The interface allows knife speed and knife extension amount to be set, as well
as monitoring of messages.

20
Chapter 3 Flexure Based Piezoelectric Actuator
Flexure Based Piezoelectric Actuator
A flexure based piezoelectric actuator was used to actuate the knife. The actuator
technology used must exhibit smooth, vibration free, and repeatable motion. The motion
range should be at least 700 µm to fully cover the previously determined block face size
of 350 µm x 350 µm. The out-of-plane stiffness should be at least 2.2 N/µm to ensure less
than 20 nm of displacement for a vertical forces of 50 mN induced during cutting [60]. It
should also be able to output approximately 50 mN of force through the whole range that
it is cutting. The actuator was designed to be as compact as possible.
This chapter discusses the background and design considerations for a piezo actuator,
then it presents a design for the piezoelectric flexure actuator that was used in the
SBFSEM ultramicrotome. Finally, alternative solutions are discussed, focusing on why
they were not used in this design.
3.1 Introduction
Piezoelectric actuators operate based on the inverse piezoelectric effect [40], meaning
an electric field applied to them will cause a deformation. They are a popular choice for
actuators inside SEM because they are easy to control in that they exhibit a near linear
voltage-displacement characteristic, and are capable of high force and nanometer
resolution. Many piezoelectric actuators are available in the form of stacks [61, 62], where
layers of piezoelectric material are layered to combine their displacements to create a
larger motion range.
Piezoelectric stack actuators can in one way be characterized by the following lines in
Figure 3.1 [63]. Each line represents a set of possible force-displacement outputs for a

21
given input voltage, the operation point of the actuator can be found given the load. There
are two quantities of interest:
Free stroke of the actuator is the x-intercept of the line and corresponds to the
displacement when the stack is actuated without load.
Blocked force is the y-intercept of the line and corresponds to the actuator’s force
output when the actuator is compressed back to zero displacement from the free
stroke length.
One implication is that any operation point beyond the line for the maximum voltage
of the piezoelectric stack cannot be reached. Another implication is that to maximize the
amount of work done by the piezo, the operation points of a piezoelectric driven device
should lie upon the maximum work line. This line represents the most balanced force-
Figure 3.1: Each solid line represents the set of possible force-displacement operation points
for a given voltage. To maximize the amount of work done by a piezo stack, the load on the
stack should be such that the operation points will lie upon the dashed maximum work line.
150 V
120 V
90 V
60 V
30 V
Maximum work line
0
200
400
600
800
1000
1200
1400
1600
1800
2000
0 10 20 30 40 50
Availa
ble
Outp
ut
Forc
e [N
]
Displacement [µm]

22
displacement operation. This information is important when designing actuators to
ensure that force and displacement requirements are satisfied.
To further amplify their motion, piezoelectric stack actuators are often placed within
a compliant flexure mechanism, consisting of thin flexures acting as hinges and thicker
rigid links between flexures [64]. There is no sliding motion between any surfaces in a
flexure mechanism, so a piezoelectric actuator can achieve perfectly smooth, repeatable
motion. There are chiefly two different types of flexure mechanisms for a flexure: the
parallel four-bar linkage type, and the sagittal type shown in Figure 3.2 left and right,
respectively. In both cases, a small horizontal expansion of the piezoelectric element will
cause bending of the thin flexures, leading to an amplified vertical displacement of the
actuator tip. The amplification depends on length L for the parallel bar flexure, and angle
α for the sagittal flexure.
Although the motion is greatly amplified, the final force output and stiffness of the
actuator is also greatly reduced compared to the piezo stack itself. Furthermore, the
Figure 3.2: (Left) Parallel four-bar linkage type flexure actuator. Amplification ratio depends
mostly on the length L, where larger L gives larger amplification ratio. (Right) Sagittal type
flexure actuator. Amplification ratio depends mostly on angle α, where smaller α gives larger
amplification ratio. With the same size envelope, it is easier to obtain a higher displacement ratio
on a sagittal type flexure than a four-bar linkage flexure.

23
higher the amplification ratio the higher force is required from the piezoelectric stack
because the smaller α increases the flexure’s stiffness from the stack’s perspective.
Therefore, although it is possible to have very high amplification ratios, the application
of the actuator should be analyzed carefully to determine the force requirements.
The two types of actuators differ in that it is easier to achieve a larger amplification
ratio on a sagittal type flexure, according to computer simulations. Also, the sagittal
actuator tip will move more in a straight line while the four bar linkage actuator tip will
move more in an arc. However, reasons for using a parallel four- bar linkage type flexure
include that they have different mounting points, which may be more convenient based
on the application.
3.2 Design Considerations for a Flexure Based Piezoelectric Actuator
The sagittal type actuator was chosen for this design because of its larger amplification
ratio for the same size envelope and its mounting point was deemed more convenient.
Furthermore, to design an actuator of the same amplification ratio in the four-bar linkage
style would make the actuator too large. An effect of that would be that the actuator
would act as a large overhanging cantilever, decreasing the stiffness.
The reason that the sagittal type design allows for larger amplification ratio compared
to a parallel four-bar linkage flexure of the same size is because it depends the angle of
the “hinges” in the flexure. Generally, the smaller the angle, the larger the amplification
ratio. Conversely, the amplification ratio of a four-bar linkage type flexure depends on
the length of the bars running parallel to the piezo stack, indicated in Figure 3.2.
Knowing the actuator’s target displacement, the piezoelectric stack’s displacement,
and the maximum size of the actuator, an amplification ratio can be selected and a
formula (Equation 3.1) [64] can be used to calculate the required angle between flexures:

24
Where G is gain, Δy and Δx are the flexure’s displacement and piezo stack’s
displacement, respectively, L is length of the bar between the hinges, and α is the angle
between the hinges.
However, a larger amplification ratio will cause the actuator to be stiffer in the
direction the piezoelectric stack is pushing while being softer in the direction of actuation.
Therefore, keeping in mind the operation points shown in Figure 3.1, if the flexure is too
stiff in the stack’s actuation direction, the actuator will exhibit very low displacement,
regardless of the amplification ratio.
In a flexure mechanism, the thin flexures do not exactly emulate hinges, so to obtain
the exact amplification ratio, it is necessary to perform finite element analysis (FEA) to
ensure that the required motion is achieved. In the analysis, it is appropriate to treat the
flexure mechanism as a static structural component. In performing the FEA, the main
input load is the the piezoelectric stack’s exerted force (F) and the outputs are the
piezoelectric stack’s displacement (Δx), the flexure’s displacement (Δy), and the von
Mises stress (σ) of the structure, as shown in a example solution in Figure 3.3. These
quantities are used to check the simulation results against specifications, as shown in
Table 3.1.
Equation 3.1: Assuming flexures are pin joints, this equation can be
used to calculate flexure angles to obtain the needed amplification ratio
𝐺 =∆𝑦
∆𝑥= √
𝐿2
∆𝑥2 sin2 𝛼+
2𝐿
∆𝑥𝑐𝑜𝑠𝛼− 1 −
𝐿
∆𝑥𝑠𝑖𝑛𝛼

25
Table 3.1: Monitored inputs and outputs from simulation are used to check against specifications
To check Use Do
Amplification
ratio
Stack displacement, Δx
Flexure displacement, Δy
Δy/ Δx
Operation
point
Stack displacement, Δx
Stack exerted force, F
Plot on Figure 3.1 to ensure
operation point is within the
stack’s capabilities
Motion range Flexure displacement, Δy Ensure that the specified
motion range is reached
without exceeding maximum
operation point
Stress Von Mises stress, σ Ensure that stress is below
yield stress for selected
flexure material
It is especially important to ensure that the operation point is possible with the
piezoelectric stack to be used in the design. It is also important to ensure that the stress
actuator structure does not exceed its yield stress. To change these outputs, the
Figure 3.3: An example output of an FEA simulation of a piezoelectric flexure mechanism. The four
quantities indicated in the figure should be used as shown in Table 3.1 to compare with specifications.

26
parameters of flexure angle, flexure thickness, and flexure length should be altered. In
general, increasing each parameter will cause amplification ratio and the stack exerted
force to decrease, but will also cause stress to decrease. If no combination of those
parameters is possible to achieve the required amplification ratio and motion range, it
may be necessary to increase the overall size of the flexure structure.
Finally, it is important to consider the mounting of the piezoelectric stack within the
actuator. To ensure that the piezoelectric stack maintains good contact during operation
when placed within the flexure, the stack must be preloaded [63]. Because piezoelectric
stacks are brittle, the loading must be evenly distributed on the ends and parallel to the
stack to avoid any torsion or bending of the stack. Internal tensile forces, which arise from
torsion and bending, will cause premature failure of the stack.
3.3 Solving the Out-of-plane Stiffness Problem
Through simulations, it was found that the flexure actuators were not stiff enough in
the direction perpendicular to the block face. This means it cannot withstand the forces
exerted vertically while cutting. This will cause sever chatter as discussed in section 2.3.
To remedy this, an extra flexure was added to brace the actuator in that direction.
3.4 The Designed Piezoelectric Flexure Actuator
The piezoelectric stack (PiezoDrive SA070742), with a free stroke and blocking force
of 50 µm and 1800 N, respectively, was chosen for this actuator because it has the largest
blocked force and free motion range among commercial piezoelectric stacks. To reach the
specification of 700 µm amplified displacement from 50% of the free stroke to achieve
maximum work, the formula was applied initially to find a hinge angle for an
amplification ratio of 28.

27
The present piezoelectric flexure mechanism was manufactured from 304 stainless
steel, which is non-magnetic. Other popular alternatives for manufacturing flexures are
6061 aluminum and Ti-6Al-4V titanium alloy, but they are not as stiff as 304 stainless
steel. All of these materials are suitable for machining using electric discharge machining
(EDM), which is the typical way of manufacturing piezoelectric flexure mechanisms.
The preloading of the piezoelectric stack is applied using a set screw. To prevent
torsional forces from being transmitted to the piezoelectric stack, the screw bears on a
thin steel plate placed between the stack and the screw. While preloading, the plate is
prevented from spinning with the screw. The plate also acts to evenly distribute the
preload force over the end of the piezoelectric stack.
FEA simulations showed that the piezoelectric stack did not have enough force to
achieve 700 µm with the desired amplification ratio. Therefore, the amplification ratio
was lowered to allow the piezoelectric actuator to expand more. This also allowed the
piezoelectric stack to overcome the stiffness added in the actuating direction by the
support flexure. In adding the support flexure, simulations showed that an increase in
stiffness in the actuating direction corresponds approximately to a 1:1 decrease in motion
range. In this case, an increase in stiffness of 40% lead to a decrease in motion range of
40%. The support flexure increased the out of plane stiffness of the actuator by about 50
times while still allowing the motion range requirement to be met.
In the end, iterations of simulations guided the detailed dimension’ selection in the
design of this actuator. The overall size was made to be as small as possible while
accommodating the piezo stack and satisfying the motion, stiffness, and stress
requirements as discussed in 3.2 and Table 3.1. At the same time, it was ensured that the
thin features’ dimensions could still be reliably manufactured by EDM.

28
The flexure mechanism has a motion range of 740 µm with an amplification ratio of
21.3, as shown in Figure 3.4.
The out-of-plane stiffness is 15.2 N/µm, which is well over the minimum required
amount, and the blocked force is 20 N, which is more than is needed to slice [60]. Without
the support flexure, the out of plane stiffness is 0.333 N/µm. This motion range matches
the simulation result of 780 µm to within 5%, showing that this simulation model can
Figure 3.4: (Top) The flexure actuator has a final motion range of 740 µm. (Bottom) There is a
hysteresis effect that is evident on the retraction stroke of the actuator but it is ignored because no
cutting is done on retraction. The extension stroke matches well with simulation results (less than
5% error).
Extend
Retract
Maximum displacement150 V, 740 µm
Simulation
0
100
200
300
400
500
600
700
800
0 50 100 150
Dis
pla
cem
ent
[µm
]
Input Voltage [V]

29
provide a good guideline for future actuators of similar design. This error can be
attributed to small errors in alignment and dimensions arising from the manufacturing
and assembly processes.
The piezoelectric flexure exhibits a hysteresis curve typical for piezoelectric actuators.
The extension stroke follows a quite linear voltage-displacement characteristic, while the
retraction stroke exhibits a maximum of 20.3% non-linearity error in its voltage-
displacement characteristic. The non-linearity of the retraction stroke does not affect the
operation of the ultramicrotome because the knife itself moves along the same path
during extension and retraction. Therefore, the hysteresis is not compensated for and the
knife is run in open loop. However, to reduce the effects of hysteresis while operating in
open loop, feedforward drives [65, 66] or charge drives [67, 68] can be implemented.
3.5 Discussion of Alternative Designs
In previous designs of the flexure actuator, two piezo stacks were placed in series in
an attempt to increase the motion range while using the “maximum work line”. Although
in theory and simulation, this is possible, it is risky to implement. Unless the stacks are
assembled with high accuracy, it is very easy to misalign and place unintentional tensile
loads on the stacks, causing them to fail. A rule of thumb is that when placing
piezoelectric stacks in series, the end side length should be more than one-tenth of the
total length after stacking.
Another option to increase motion range is to place two flexure actuators in series.
Although this method avoids having two piezo stacks in series, the tip of the actuator is
now a large overhanging beam, meaning the stiffness is very low. Therefore, the
“maximum work line” is not used and only a single actuator with a single stack is used.

30
Chapter 4 Thermal V-Beam Actuator
Thermal V-Beam Actuator
The thermal V-beam actuator is used as the actuator to advance the specimen. In
addition to having smooth and repeatable motion, this actuator also needs to be stiff. The
displacement of the actuator needs to be at least 250 µm to satisfy the specimen advance
distance requirement. The stiffness in the specimen advance direction of the actuator
needs to be 2.2 N/µm, similar to the knife actuator. The maximum temperature that the
actuator should reach is 90 °C, which will be safe for the SEM stage.
This chapter discusses the background and design considerations for a thermal v-
beam actuator, then it presents the design for the v-beam actuator that was used in the
SBFSEM ultramicrotome. Finally, alternative solutions are discussed, including a
comparison between the thermal actuator and a piezoelectric actuator of similar size and
motion specifications.
4.1 Introduction
Thermal V-beam actuators are also flexure based, allowing smooth, repeatable
motion. Instead of an element placed inside the actuator like in the case of piezoelectric
flexure actuators however, the movement is caused by thermal expansion of the
actuator’s flexures, which are angled. The flexures warm up and expand, while the
relatively thicker parts of the actuator remain cool to brace against the expansion, forcing
the actuator tip to move as shown in Figure 4.1. The temperature-displacement
characteristic of the actuator is roughly linear. The thermal aspect of the actuator allows
for very smooth, high resolution motion while the bracing aspect of the actuator allows
high force output.

31
Similarly to the piezoelectric actuator, a small displacement, now caused by thermal
expansion, translates to a large displacement at the actuator tip. A smaller flexure angle
also translates to a larger displacement. Furthermore, a larger displacement also leads to
lower stiffness in the actuating direction. Differently from piezoelectric actuators, it is
possible to place many flexures in parallel to compensate for the lower stiffness. Because
it is the flexures themselves and not an external component that cause the displacement,
meaning force is not as big of a concern. However, more heat will be needed to cause the
same thermal expansion in more flexures.
Conventionally, this type of actuator was used in microelectromechanical systems
(MEMS) [69], and heating is provided by running current directly through the actuator’s
Figure 4.1: In the thermal v-beam actuator, heat is applied, causing thermal expansion of the flexures.
Due to the geometry, the actuator tip will be pushed upwards, as shown with exaggerated
displacement. Given a maximum size for the actuator, the displacement of the tip depends on the
thickness and angle θ of the beam.

32
flexure beams, resulting in resistive heating. Heating power has a quadratic relationship
with input voltage, and is given by Equation 4.1.
However, for this macro scale actuator, the low resistance of the flexure makes it
unfeasible to heat up that way. Instead, a resistive cartridge heater is used. This allows
more freedom in varying the geometry of the actuator because heating is no longer tied
to the resistance of the actuator. Also contrasting with the MEMS version of the actuator
is that because the thermal mass is so much higher, it is more difficult to control due to
its slow speed and sensitivity to ambient temperature variations.
4.2 Design Considerations of a Thermal V-Beam Actuator
In SBFSEM, the maximum temperature of the thermal actuator should be selected so
that it does not damage the SEM stage. Also, care should be taken to isolate the sample
and the knife from the heat generated by the thermal actuator. Knowing the desired
displacement of the actuator tip, the maximum size of the actuator, and the maximum
temperature desired, the angle for the flexure beams can be determined from Equation
4.2 [70]:
Equation 4.1: Resistive heating power is caused by ohmic
heating, which has a quadratic relationship with voltage.
𝑃 =𝑉2
𝑅
Equation 4.2: Assuming uniform flexure beam temperature, this equation can be used
to calculate the angles of the flexure beams required to generate the needed displacement.
𝑈 = 𝛼∆𝑇𝑙𝑠
(𝑠2 + 𝑐2 12𝐼𝐴𝑙2)

33
Where U is the displacement, α is the thermal expansion coefficient of the material, ΔT is
the change in temperature, l is the length of the beam, I is the second moment of area of
the beam, and A is the cross sectional area of the beam. The s and c are shorthand for sinθ
and cosθ where θ is the angle of the flexure beams, as shown in Figure 4.1.
Because the beam temperature is not uniform and there is a temperature gradient
along the beam, it is necessary to run FEA simulations to confirm the geometry. The
thermal actuator should be modeled as a thermal-structural component. The input load
is a thermal flux (Q) with units of power divided by area to where the cartridge heater is
placed, and the outputs are the temperature (ΔT), the tip displacement (U), and the von
Mises stress (σ). Because of the vacuum environment and the relatively low temperatures
reached by the actuator, convection and radiation heat transfer are ignored.
The thermal flux (Q) should not exceed the maximum power output of the cartridge
heater to be used. The change in temperature (ΔT) should not exceed the safe temperature
for the load on the actuator tip, or in the case of the SBFSEM ultramicrotome, cause
Figure 4.2: An example output of an FEA simulation of a thermal v-beam actuator. The four quantities
indicated in the figure should be used to compare with specifications.

34
damage to the SEM stage. The tip displacement (U) should meet the displacement
requirement. Von Mises stress (σ) should be under the yield stress for the given material.
When designing the v-beam actuator, consideration should be given to the mounting
of the heating cartridge. Most resistive heating cartridges are meant to be placed inside a
hole. The hole should be reamed to an accurate diameter and smooth surface finish, with
thermally conductive compound applied to maximize the efficiency of heat transfer
between the heater body and the actuator.
4.3 The Designed Thermal V-Beam Actuator
The designed thermal v-beam actuator uses the ThorLabs HT15W, with a resistance
of 38.4 Ω. With an input voltage of 16.5 V, it has a motion range of 310 µm, exceeding the
250 µm specimen travel distance requirement and staying under the 15 W maximum
output of the heater, as shown in Figure 4.3. The maximum temperature reached to
achieve this motion range is 70 °C, which is safe for the SEM stage. The stiffness of the
actuator in the actuating direction and the cutting direction is 2.21 N/µm and 58.8 N/µm,
respectively. The actuator is shown in Figure 4.4.
The actuator moves slowly given a voltage input and follows a first order transient
response. The time constant is 5.23 min, while the settling time is 20.5 min, as shown in
Figure 4.3. However, knowing the transient characteristic, it is possible to give a large
step input and know the position of the actuator tip at any given time. For example,
instead of inputting a step voltage to displace the actuator 100 nm and waiting for 20.5
min, a step voltage can be given to displace the actuator 500 nm, and the time intervals
for each 100 nm within the 500 nm will be known.

35
The thermal v-beam actuator is manufactured from 304 stainless steel and was
machined using EDM. 6061 aluminum alloy’s combination of heat capacity and thermal
conductivity cause the entire structure to be roughly uniform in temperature, meaning
the thicker parts of the actuator do not stay cool and brace against the expansion of the
flexure beams.
Figure 4.3: (Left) For a steady input voltage, after the actuator has reached steady state, the actuator
follows a quadratic voltage-displacement characteristic. The actuator has a range of 310 µm. (Right)
The specimen actuator follows a first-order transient characteristic with a time constant of 5.23 min
and a settling time of 20.5 min inside the SEM. Knowing this, for any step input voltage to the
actuator, it is possible to calculate the displacement of the actuator at any time.
16.5 V, 310 µm
0
50
100
150
200
250
300
350
0 5 10 15 20
Dis
pla
cem
ent
[µm
]
Input Voltage [V]
Displacement
Fitted
Time constant,
τ = 5.23 min
Settling time,Ts = 20.5 min
0
20
40
60
80
100
0 10 20 30
Dis
pla
cem
ent
[% fin
al valu
e]
Time [Minutes]
Displacement
Fitted

36
4.4 Discussion of Previous Specimen Actuator Designs
In earlier iterations of the ultramicrotome, the specimen actuator used a piezoelectric
flexure design as discussed in chapter 3. According to FEA simulations, the actuating
direction stiffness of the piezoelectric actuator was 0.0429 N/µm, compared to the 2.21
N/µm of the thermal actuator, even though the two actuators have similar size envelopes.
As shown in Figure 5.2, the low actuator stiffness causes severe chatter while sectioning.
Figure 4.4: The designed v-beam thermal actuator has a polished, reamed, hole for accepting a resistive
cartridge heater. Flexure beams expand against the bracing, forcing the actuator tip upwards.

37
Chapter 5 Sectioning using the SBFSEM System
Sectioning using SBFSEM System
In this section, the ultramicrotome’s cutting performance in terms of minimum slice
thickness and cut surface roughness was verified and demonstrated. Alternative ways to
improve cut surface roughness is discussed. Finally, a sample TEM grid was embedded
in resin, then sectioned and 3D reconstructed to demonstrate the accuracy of the
ultramicrotome.
5.1 Specimen Height Control for Sectioning
To control the height of the specimen for sectioning and avoid having to wait until the
settling time of the actuator each time, a larger step input can be given to the actuator and
the cuts can be taken at the appropriate time. The transient characterization in 4.3 can be
used. For example, if a step input of 500 nm is set, then five 100 nm cuts can be taken
within the settling time (20.5 min), as shown in Figure 5.1.
Figure 5.1: Timings for five cuts in 20.5 min, with a 500 nm step input.
1.17 min, 100 nm
2.67 min, 200 nm
4.8 min, 300 nm
8.42 min, 400 nm
20.5 min, 500 nm
0
100
200
300
400
500
0 5 10 15 20 25
Actu
ato
r positio
n [
nm
]
Time [min]

38
Choosing this step input amount allows ample time for the SEM image capture
between the 100 and 200 nm cuts. Typically, a capture of a high-quality image in SEM
takes approximately 30 seconds.
5.2 Minimum Slice Thickness
The minimum slice thickness that the designed SBFSEM system was tested to ensure
that it met the minimum slice thickness requirement of 100 nm. The ultramicrotome can
cut 90 nm slices 95% of the time.
To test the thickness, a block of EMbed 812 resin prepared with a 350 x 350 µm2 block
face area was prepared to be cut with a 45° glass knife. As the specimen was advanced
little by little according to the characterization data in section 4.3, the knife was moved
over to attempt a cut each time. If a cut was made, the section would fall on the block face
or pile on the knife edge. A failure occurs either if an entire slice was not removed from
the block face, or if no cutting occurred at all. Failures are likely caused by the fact that
the knife edge is not sharp enough, causing it to “roll over” as it tries to cut.
5.3 Cutting Surface Roughness
A low surface roughness after cutting, meaning a smooth surface, means that the
cutting thickness is uniform. During development, interim results showed that increasing
the stiffness of the ultramicrotome structure greatly decreased surface roughness. This
observation guided the ultramicrotome design to become stiff. To ensure the chatter
during cutting is low, the surface roughness is measured. The results are shown in Figure
5.2.

39
EMbed 812 blocks were prepared with a 350 x 350 µm2 block face area. The slicing
thickness was 200 nm. After cutting, the block face is scanned by atomic force microscopy
(AFM) to obtain the roughness profile of the surface. When sectioning, chatter marks will
appear running parallel to the knife edge, while marks caused by nicks in the knife edge
appear running perpendicular to the knife edge. The AFM scans are run perpendicular
to the knife edge to measure the chatter. A sample cut using an older revision of the
ultramicrotome having insufficient stiffness is also included for comparison.
Figure 5.2: (Top left and right) Optical micrographs of a rough (A) and smooth (B) block face. The
cutting direction is from top to bottom. The rough block face was cut using an older revision of the
ultramicrotome which was not as stiff and severe chatter occurred. (Bottom) The AFM line scans of the
sample show that the RMS of the smooth surface is substantially lower than the RMS of the rough
surface.
Rough profile (A):RMS: 122 ± 8.3 nm
Smooth profile (B):RMS: 15.7 ± 2.4 nm
-300
-200
-100
0
100
200
300
400
500
600
0 10 20 30 40 50 60 70 80 90
Devia
tion [
nm
]
x [µm]

40
The AFM scan showed that the surface roughness of the block face cut using the
newest revision of the ultramicrotome is substationally smoother, having an RMS surface
roughness of 15.7 nm compared to 122 nm cut using an older revision of the
ultramicrotome. The stiffness of the new ultramicrotome is approximately 50 times that
of the older version.
5.4 Cutting Parameters Affecting Surface Roughness
The following describes effects of the parameters of knife cutting speed and slicing
thickness on surface roughness were studied to find acceptable ranges, i.e. giving less
than 20 nm roughness RMS. These tests were performed cutting an EMbed 812 sample
with a block face area of 350 x 350 µm2 using a glass knife. Each slice was 200 nm thick.
Results showed that at ranges tested, knife speed had little effect on chatter, while cutting
too thick will cause lots of chatter, as shown in Figure 5.3.
Figure 5.3: (Left) There is little to no effect of knife speed on chatter. Statistical analysis shows a low
correlation value of -0.45 between knife speed and chatter. (Right) As section thickness is increased,
little chatter until a section thickness of 500 nm, when the roughness RMS rapidly rises.
Average RMS: 16.6 nm
0
5
10
15
20
25
30
35
40
0 200 400 600 800
Roughness R
MS
[nm
]
Knife Cutting Speed [µm/s]
0
10
20
30
40
50
60
70
80
0 200 400 600
Ro
ugh
nes
s R
MS
[nm
]
Slice thickness [nm]
Too thick

41
5.4.1 Knife speed has little effect on chatter
Typical knife cutting speeds in other SBFSEM ultramicrotomes are in the range of 0.1
to 1 mm/s [37]. Running the knife too fast or too slow during cutting may cause chatter
during sectioning, as discussed in section 2.3. When the knife is run too fast, besides
causing chatter due to undampened vibrations, the great force of the crash of the knife
edge into the sample may cause knife edge chipping or other damage to the sample itself.
In our tests, a range of speeds from 0.1 to 0.7 mm/s were tested, which are within the
typical cutting speed range [37]. The results in this range showed that there is little to no
effect of knife speed on chatter, which had an average value of 16.6 nm, shown in Figure
5.3.
5.4.2 Chatter increases as slice thickness increases
The slice thickness range of this ultramicrotome with acceptable chatter is from 90 nm
to 400 nm. A sample was prepared similarly to in 5.4.1, and was cut at varying slice
thicknesses with a knife speed of 0.25 mm/s. The results are shown in Figure 5.3.
The lower limit of slice thickness testing was the result shown in section 5.2. As
discussed in section 2.3, when the slice is too thick, the force required to curl the section
over the knife blade is high and causes chatter. This happens because the force will start
to pull the knife down into the block face until it is deep enough that the elastic force of
the ultramicrotome stiffness pulls the knife back to the surface. This process repeats and
its periodicity produces the distinct chatter. Although slices thicker than 500 nm are
sometimes cut in conventional ultramicrotomy, in SBFSEM, the section thicknesses are
usually lower. Higher structure stiffness should increase the thickness that the
ultramicrotome can cut smoothly.

42
5.5 Cutting Force Estimation for Future Design Guideline
Following the results of the effects of slice thickness on chatter presented in 5.4.2, the
cutting force may be estimated to define a threshold of the minimum required stiffness
to withstand a certain cutting force. The estimation is based on the cutting mechanics,
which is briefly discussed in 2.3.
To estimate the cutting force, fracture energies of resin reported in [71] and section
compression values reported by [51] can be used with the cutting geometry presented in
[55] shown in Equation 5.1 and Equation 5.2. An introduction to cutting geometry is given
in 2.3 and a detailed discussion is given in [55]. The implication of this is that knowing
the material properties of a material to be cut and the geometry of the knife, one can
estimate the required stiffness of the structure required to withstand the cutting force.
In Equation 5.1, r is compression ratio, where t1 is the thickness before and t2 is the
thickness after compression. The rake angle is α and the slip plane is φ. In Equation 5.2,
Wc is work expended to section per unit volume, F is the force along the upper knife face,
Fc is the force of the knife parallel to the block surface face, Ft is the force of the knife
Equation 5.1: Rake angle (α) and the compression
ratio (r) are used to calculate the slip plane (φ).
𝑟 =𝑡1
𝑡2=
sin(𝜑)
cos (𝜑 − 𝛼)
Equation 5.2: Work expended to section per unit volume (Wc), rake angle (α), and slip plane
angle (φ) are used to calculate the vertical (Ft) and lateral (Fc) forces involved in sectioning.
(
𝐹𝑐
𝐹𝑐
𝑊𝑐𝐴0
) = (
𝑐𝑠𝑐 (𝛼) −𝑐𝑜𝑡 (𝛼) 0
0 𝑡𝑎𝑛 (𝜑) 𝑠𝑒𝑐(𝜑)𝑟 0 𝑠𝑖𝑛 (𝜑)
) (
𝐹
𝐹𝑡
𝐹𝑠
)

43
pulling the knife into the block face, and A0 is the cross sectional area of the section before
cutting.
Using these formulae, it was estimated that the stiffness to cutting force ratios to
ensure < 20 nm surface roughness are:
Lateral (Fc): 58.5 N/µm stiffness to 75 mN cutting force
Vertical (Ft): 2.21 N/µm stiffness to 77.4 mN cutting force
Therefore, in the future, this result may be used as a guideline for designing SBFSEM
ultramicrotomes. Along with allowing cutting materials requiring higher force, higher
stiffness is expected to decrease surface roughness overall. This result also reinforces the
idea that reducing the required cutting force may decrease block face roughness.
5.6 Oscillating Knife: A Method to Reduce Cutting Force
Besides changing the angles of the knife, it is also possible to reduce the cutting force
by oscillating the knife along the direction of the knife edge [51], as shown in Figure 5.4.
This method was successfully implemented to decrease slice compression in a
conventional ultramicrotome. By reducing the cutting force, it may be easier to design a
structure stiff enough to withstand the cutting forces incurred while sectioning larger
areas, or harder materials to reduce chatter.
Instead of the ploughing motion as described in orthogonal cutting, oscillating the
knife produces a slicing motion and the apparent bevel angle is smaller as the sample
moves over the knife. Assuming the clearance angle is constant, the apparent rake angle
will increase. The higher the ratio of the knife’s lateral speed caused by vibration to the
knife feed speed, the smaller the apparent bevel angle.

44
Preliminary results were obtained by slicing a 400 nm thick section while vibrating
the knife at 7 kHz as shown in Figure 5.4 (bottom left). An older revision of the
ultramicrotome using a piezoelectric (soft) specimen actuator was used during this test.
Figure 5.4: (Top left) A piezoelectric stack is attached to a knife with a counter weight to oscillate the
knife. (Top right) Knife apparent angle, caused by knife oscillation combined with the knife feed, is
smaller than the knife bevel angle. (Bottom left) Block face after slicing off 400 nm at a frequency of 7
kHz. (Bottom right) Vibrating glass knife becomes damaged easily, causing severe chatter. The left side
of the blade is more damaged than the right side.

45
A knife speed of 0.1 mm/s, as used in this case, gave the smoothest result. 7 kHz was
chosen as a frequency because that was the resonant frequency of the combination of the
piezoelectric stack combined with the counterweight and knife, maximizing the
amplitude. Compared to the block face in Figure 5.2A, it is much smoother, but the
chatter marks are still apparent when compared to Figure 5.2B. The chatter is also
apparent compared to the block face in the 400 nm case in Figure 5.3.
In the final design of the ultramicrotome, the vibrating knive is not used. The
ultramicrotome could not slice thinner than 400 nm with the vibrating knife. The reason
for this may be that the vibration is not totally in line with the knife edge. In fact, the glass
knife edge is not totally straight, whereas in [51] a diamond knife is used which is known
to have a straighter knife edge than glass knives. When cutting while vibrating the knife
at other speeds, there appears to be a shredding action, which may be evidence that the
vibration is out of alignment with the edge.
A consequence of the out-of-alignment vibration is that the knife is more easily
damaged as it oscillates while cutting. Although the force appears to be decreased,
showing less chatter, the longevity of the knife is also decreased. As the knife is cutting,
the misaligned vibrations cause chips to form in the knife edge. A result of cutting using
the damaged knife edge is shown in Figure 5.4 (bottom right), having severe chatter due
to increased cutting force. In this case, the knife was more damaged on the left side than
the right.
This particular arrangement of the vibrating knife leaves the counterweight as an
overhanging mass on the piezoelectric stack. Combined with possible misalignments, the
large counterweight mass, and the high frequency vibration, this could introduce large
tensile stresses in the stack, leading to premature stack failure. However, the results show
promise that the vibrating knife could be incorporated into future designs.

46
The most obvious solution to both the vibration misalignment and overhanging mass
problem that could be implemented in future designs is a flexure arrangement which
could constrain the vibration to a single direction, as well as preload the stack as to
prevent tensile loading. Other considerations are to determine the modes of vibration of
the flexure to ensure that any off-axis vibration modes are not excited. Each glass knife is
also slightly different, so a change to a diamond knife may be necessary.
5.7 3D Reconstruction Accuracy Validation
To demonstrate the function of the ultramicrotome, a copper TEM grid (Gilder
G2000HS) was embedded in a resin (EMbed812) then sliced, imaged, and 3D
reconstructed as shown in Figure 5.5. TEM grids have a regular and well- known
structure, allowing us to easily verify the reconstructed image’s integrity. This TEM grid
has nominal dimensions of 12.5 µm grid spacing, with a hole width of 7.5 µm and a
thickness of 6 µm. For 3D reconstruction, 200 slices were cut from a block face of 350 µm
x 300 µm with a knife speed of 0.25 mm/s each at 100 nm thick. The SEM used was the
Hitachi SU3500 using 15 kV accelerating voltage with 30 Pa chamber pressure under
backscatter composition imaging mode.
After images were acquired, they were manually aligned by matching features in the
image that don’t move and cropped to show only the area of interest. Image processing
was performed to remove noise from surrounding areas caused by SEM imaging or
debris falling on the block face. The images were stacked using ImageJ and reconstructed
using the 3DViewer plugin. After cropping, the total volume reconstructed was 87 µm x
67 µm x 20 µm, which is comparable to volumes reconstructed in Table 1.1.

47
In the construction, there are a few pieces of large debris that were left on the block
face, leading to the artifacts outlined in blue in Figure 5.5 (top right). These pieces were
difficult to isolate from the actual TEM grid, so they were not removed. Debris piled on
the knife edge occasionally falls onto the block face. There is also a large discontinuity in
Figure 5.5: (Top left) Individual SEM images were taken, then processed and aligned to form a
stack. (Top right) A TEM grid was reconstructed using SBFSEM with a voxel size of 66 x 66 x
100 nm. The grid spacing as measured from the 3D reconstruction by our SBFSEM system is
matches with manufacturer specifications. Shaded in areas show the first section. Outlined areas
show debris and discontinuities, as labelled. (Bottom) Reference TEM grid with dimensions.

48
the piece shown outlined in orange in Figure 5.5 (top right). The discontinuity occurred
when the cutting process was interrupted because the SEM beam emitter had burned out
and needed replacement. A mistake was made upon restarting the sectioning process. An
interesting note is that although it is not recommended to take more than 50 slices on one
area of a glass knife [9], this particular knife was used for more than 200. This knife may
be an outlier.
Measurements shown in Figure 5.5 (top right) compared with reference dimensions
from Figure 5.5 (bottom) show that the ultramicrotome was able to 3D reconstruct a
sample with good accuracy. The two squares that were measured were picked because
they were far from any debris. The results are accurate within 1% and are presented in
Table 5.1.
Table 5.1: 3D reconstruction is accurate within 1% for length measurements.
Dimension Reference (nm) Measured (nm) Error (%)
Hole width 7540 ± 5 7518 ± 5 -0.29
Hole pitch 12500 ± 5 12627 ± 49 1.0
The measured dimension is outside the error range of the reference. One reason could
be because of the warping of the TEM grid, which is not flat even before it is embedded
in the resin. Measuring on a warped model will introduce small amounts of errors. Also,
the curvature of the knife blade, although small, may have contributed to the error. The
reconstruction was done assuming each slice is perfectly flat. There are also SEM errors.
Over time, the brightness, focus, and position of the SEM beam will drift. Although
efforts were made to align the images in post-processing manually, it is reasonable to
expect approximately 1-2 pixels (66-132 nm) of error when aligning by eye. The errors
observed are within this range.

49
In conclusion, the SBFSEM ultramicrotome is good for morphology visualization, as
shown by the 3D reconstruction, and it was accurate (< 1% error) in length measurements.
However, the large amount of SEM image drift required manual alignment which may
introduce errors in measurements. In the future impelementing computer vision
algorithms like template matching can compensate for this drift and increase accuracy.

50
Chapter 6 Conclusion and Outlook
Conclusion and Outlook
This thesis presented the development of a prototype of a plug-and-play SBFSEM
system that can be used to study morphology in biological and materials science. It was
accomplished by using compact actuators that are typically used for SEM
nanomanipulation.
Plug-and-Play SBFSEM Ultramicrotome
A prototype plug-and-play SBFSEM ultramicrotome was designed that can be used
with no modification to the SEM. By reducing the size of the ultramicrotome using
smaller piezoelectric and thermal actuators, it can be used with any SEM that has the
required imaging modes. It has the capability to perform 3D reconstruction of large
volumes for studies in materials or biological sciences. Because it is plug and play, it can
be installed and uninstalled easily and quickly, meaning it will not disrupt shared
electron microscopy facilities.
Piezoelectric Flexure Actuator
A piezoelectric flexure actuator is designed that has high motion range and a high
out-of-plane stiffness. The high motion range ensures that the knife can slice the entire
surface of the sample. The high stiffness knife actuator will not deflect too much while
cutting, ensuring that there is low chatter.
Thermal V-beam Actuator
A macro-scale version of a MEMS v-beam actuator was designed and characterized
to be used as a specimen advance actuator. Instead being heated by current running
through its flexure beams, the actuator is heated by a cartridge heater installed in its tip.

51
The fact that the thermal expansion of the flexure beams provides the displacement for
the actuator means that it can be much stiffer than a piezoelectric flexure actuator of the
same size.
3D Reconstruction of a TEM grid
The SBFSEM ultramicrotome’s function was verified by sectioning and reconstructing
a TEM grid embedded in resin. The morphology of the grid was preserved in the
reconstruction. Measurements of the reconstructed grid’s dimensions were accurate
within 1% of the grid’s specifications, showing that the ultramicrotome has good
accuracy.
6.1 Contributions
1. Designed and characterized a prototype plug-and-play SBFSEM ultramicrotome.
2. Developed a method to improve out-of-plane stiffness of sagittal piezoelectric
flexure mechanisms.
3. Developed a macro-scale MEMS-style v-beam thermal actuator.
4. Demonstration of function of ultramicrotome, sectioning and 3D reconstructing a
TEM grid with high accuracy.
6.2 Future Directions
Add closed loop control to specimen actuator. This allows the specimen to be raised or
lowered more quickly and accurately.
1. Optimize the arrangement of the actuators to increase the stiffness and reduce the
size, possibly make it load lock compatible so it can be used in high-vacuum SEMs.

52
2. Implement computer vision algorithms to compensate for drift in SEM image or
to align images for more accurate 3D reconstruction.
3. Develop a flexure arrangement allowing a vibrating knife to lower the required
cutting force.
4. Implement an auto-approach feature to the specimen advancing to the knife,
lowering the risk of damaging the knife and to reduce operator mental load.

53
References
[1] W. Denk and H. Horstmann, "Serial Block-Face Scanning Electron Microscopy to
Reconstruct Three-Dimensional Tissue Nanostructure," PLoS Biol, vol. 2, p. e329,
2004.
[2] J. J. Bozzola and L. D. Russell, Electron Microscopy: Jones & Bartlett Learning, 1998.
[3] N. Nagy and J. A. McGrath, "Blistering skin diseases: a bridge between
dermatopathology and molecular biology," Histopathology, vol. 56, pp. 91-99, 2010.
[4] S. R. Glanvill, "Ultramicrotomy of semiconductors and related materials,"
Microscopy Research and Technique, vol. 31, pp. 275-284, 1995.
[5] A. Antonovsky, "Ultramicrotomy of industrial alumina," Microscopy Research and
Technique, vol. 31, pp. 300-307, 1995.
[6] G. McMahon and T. Malis, "Ultramicrotomy of nanocrystalline materials,"
Microscopy Research and Technique, vol. 31, pp. 267-274, 1995.
[7] "Preparation of Thin Sections: (Cryo)ultramicrotomy and (Cryo)microtomy," in
Electron Microscopy of Polymers, ed Berlin, Heidelberg: Springer Berlin Heidelberg,
2008, pp. 199-217.
[8] P. M. Conn, Methods in Cell Biology: Academic Press, 2013.
[9] M. J. Dykstra, A Manual of Applied Techniques for Biological Electron Microscopy, 1
ed.: Springer US, 1993.
[10] P. S. Holcomb, B. K. Hoffpauir, M. C. Hoyson, D. R. Jackson, T. J. Deerinck, G. S.
Marrs, et al., "Synaptic Inputs Compete during Rapid Formation of the Calyx of
Held: A New Model System for Neural Development," The Journal of Neuroscience,
vol. 33, pp. 12954-12969, August 7, 2013 2013.
[11] L. Xiao, N. Michalski, E. Kronander, E. Gjoni, C. Genoud, G. Knott, et al., "BMP
signaling specifies the development of a large and fast CNS synapse," Nat Neurosci,
vol. 16, pp. 856-864, 07//print 2013.
[12] B. Andres, U. Koethe, T. Kroeger, M. Helmstaedter, K. L. Briggman, W. Denk, et
al., "3D segmentation of SBFSEM images of neuropil by a graphical model over
supervoxel boundaries," Medical Image Analysis, vol. 16, pp. 796-805, 5// 2012.

54
[13] A. Kreshuk, R. Walecki, U. Koethe, M. Gierthmuehlen, D. Plachta, C. Genoud, et
al., "Automated tracing of myelinated axons and detection of the nodes of Ranvier
in serial images of peripheral nerves," Journal of Microscopy, vol. 259, pp. 143-154,
2015.
[14] K. E. Froud, A. C. Y. Wong, J. M. E. Cederholm, M. Klugmann, S. L. Sandow, J.-P.
Julien, et al., "Type II spiral ganglion afferent neurons drive medial olivocochlear
reflex suppression of the cochlear amplifier," Nature Communications, vol. 6, p.
7115, 05/12/online 2015.
[15] P. J. Kempen, M. F. Kircher, A. de la Zerda, C. L. Zavaleta, J. V. Jokerst, I. K.
Mellinghoff, et al., "A correlative optical microscopy and scanning electron
microscopy approach to locating nanoparticles in brain tumors," Micron, vol. 68,
pp. 70-76, 1// 2015.
[16] N. Ohno, H. Chiang, D. J. Mahad, G. J. Kidd, L. Liu, R. M. Ransohoff, et al.,
"Mitochondrial immobilization mediated by syntaphilin facilitates survival of
demyelinated axons," Proceedings of the National Academy of Sciences, vol. 111, pp.
9953-9958, 2014.
[17] R. Yamasaki, H. Lu, O. Butovsky, N. Ohno, A. M. Rietsch, R. Cialic, et al.,
"Differential roles of microglia and monocytes in the inflamed central nervous
system," The Journal of Experimental Medicine, vol. 211, pp. 1533-1549, 2014.
[18] J. Cooperrider, H. Furmaga, E. Plow, H.-J. Park, Z. Chen, G. Kidd, et al., "Chronic
Deep Cerebellar Stimulation Promotes Long-Term Potentiation, Microstructural
Plasticity, and Reorganization of Perilesional Cortical Representation in a Rodent
Model," The Journal of Neuroscience, vol. 34, pp. 9040-9050, 2014.
[19] T. Starborg, N. S. Kalson, Y. Lu, A. Mironov, T. F. Cootes, D. F. Holmes, et al.,
"Using transmission electron microscopy and 3View to determine collagen fibril
size and three-dimensional organization," Nat. Protocols, vol. 8, pp. 1433-1448,
07//print 2013.
[20] C. Pinali and A. Kitmitto, "Serial block face scanning electron microscopy for the
study of cardiac muscle ultrastructure at nanoscale resolutions," Journal of
Molecular and Cellular Cardiology, vol. 76, pp. 1-11.
[21] M. K. O’Connell, S. Murthy, S. Phan, C. Xu, J. Buchanan, R. Spilker, et al., "The
Three-Dimensional Micro- and Nanostructure of the Aortic Medial Lamellar Unit
Measured Using 3D Confocal & Electron Microscopy Imaging," Matrix biology :
journal of the International Society for Matrix Biology, vol. 27, pp. 171-181, 11/13 2008.

55
[22] K. Ichimura, N. Miyazaki, S. Sadayama, K. Murata, M. Koike, K.-i. Nakamura, et
al., "Three-dimensional architecture of podocytes revealed by block-face scanning
electron microscopy," Scientific Reports, vol. 5, p. 8993, 03/11/online 2015.
[23] N. Hondow, M. R. Brown, T. Starborg, A. G. Monteith, R. I. K. Brydson, H. D.
Summers, et al., "Quantifying the cellular uptake of semiconductor quantum dot
nanoparticles by analytical electron microscopy," Journal of Microscopy, vol. 261,
pp. 167-176, 2016.
[24] W.-K. Ju, K.-Y. Kim, Y. H. Noh, M. Hoshijima, T. J. Lukas, M. H. Ellisman, et al.,
"Increased mitochondrial fission and volume density by blocking glutamate
excitotoxicity protect glaucomatous optic nerve head astrocytes," Glia, vol. 63, pp.
736-753, 2015.
[25] K. Mukherjee, H. R. Clark, V. Chavan, E. K. Benson, G. J. Kidd, and S. Srivastava,
"Analysis of Brain Mitochondria Using Serial Block-Face Scanning Electron
Microscopy," p. e54214, 2016/07/09/ 2016.
[26] C.-h. O. Davis, K.-Y. Kim, E. A. Bushong, E. A. Mills, D. Boassa, T. Shih, et al.,
"Transcellular degradation of axonal mitochondria," Proceedings of the National
Academy of Sciences, vol. 111, pp. 9633-9638, 2014.
[27] E. Lipke, T. Hörnschemeyer, A. Pakzad, C. R. Booth, and P. Michalik, "Serial Block-
Face Imaging and its Potential for Reconstructing Diminutive Cell Systems: A Case
Study from Arthropods," Microscopy and Microanalysis, vol. 20, pp. 946-955,
2014/002/20 2014.
[28] C. Pinali, H. J. Bennett, J. B. Davenport, J. L. Caldwell, A. W. Trafford, and A.
Kitmitto, "185 Remodelling of Specialised Domains of the Sarcolemma in Heart
Failure; Reorganisation of the Intercalted Disc Revealed by Nano-scale Imaging,"
Heart, vol. 100, pp. A103-A104, 2014.
[29] A. Zankel, B. Kraus, P. Poelt, M. Schaffer, and E. Ingolic, "Ultramicrotomy in the
ESEM, a versatile method for materials and life sciences," Journal of Microscopy, vol.
233, pp. 140-148, 2009.
[30] T. Hashimoto, G. E. Thompson, X. Zhou, and P. J. Withers, "3D imaging by serial
block face scanning electron microscopy for materials science using
ultramicrotomy," Ultramicroscopy, vol. 163, pp. 6-18, 4// 2016.
[31] R. Schurch, S. M. Rowland, R. S. Bradley, and P. J. Withers, "Comparison and
combination of imaging techniques for three dimensional analysis of electrical

56
trees," IEEE Transactions on Dielectrics and Electrical Insulation, vol. 22, pp. 709-719,
2015.
[32] T. J. Marrow, M. Mostafavi, T. Hashimoto, and G. E. Thompson, "A quantitative
three-dimensional in situ study of a short fatigue crack in a magnesium alloy,"
International Journal of Fatigue, vol. 66, pp. 183-193, 9// 2014.
[33] T. Müllner, A. Zankel, F. Svec, and U. Tallarek, "Finite-size effects in the 3D
reconstruction and morphological analysis of porous polymers," Materials Today,
vol. 17, pp. 404-411, 10// 2014.
[34] A. Zankel, J. Wagner, and P. Poelt, "Serial sectioning methods for 3D investigations
in materials science," Micron, vol. 62, pp. 66-78, 7// 2014.
[35] B. Chen, T. Hashimoto, F. Vergeer, A. Burgess, G. Thompson, and I. Robinson,
"Three-dimensional analysis of the spatial distribution of iron oxide particles in a
decorative coating by electron microscopic imaging," Progress in Organic Coatings,
vol. 77, pp. 1069-1072, 6// 2014.
[36] A. E. Hughes, A. Trinchi, F. F. Chen, Y. S. Yang, I. S. Cole, S. Sellaiyan, et al.,
"Revelation of Intertwining Organic and Inorganic Fractal Structures in Polymer
Coatings," Advanced Materials, vol. 26, pp. 4504-4508, 2014.
[37] (2016). 3View System for Image Capture of 3D Ultrastructures. Available:
http://www.gatan.com/products/sem-imaging-spectroscopy/3view-system
[38] (2016). Teneo VS SEM for Life Sciences. Available:
https://www.fei.com/products/sem/teneo-vs-sem-for-life-sciences/
[39] C. Shi, D. K. Luu, Q. Yang, J. Liu, J. Chen, C. Ru, et al., "Recent advances in
nanorobotic manipulation inside scanning electron microscopes," Microsystems &
Nanoengineering, vol. 2, p. 16024, 06/20/online 2016.
[40] Y. K. Yong, S. O. R. Moheimani, B. J. Kenton, and K. K. Leang, "Invited Review
Article: High-speed flexure-guided nanopositioning: Mechanical design and
control issues," Review of Scientific Instruments, vol. 83, p. 121101, 2012.
[41] Y. L. Zhang, Y. Zhang, C. Ru, B. K. Chen, and Y. Sun, "A Load-Lock-Compatible
Nanomanipulation System for Scanning Electron Microscope," IEEE/ASME
Transactions on Mechatronics, vol. 18, pp. 230-237, 2013.

57
[42] T. Fukuda, F. Arai, and L. Dong, "Assembly of nanodevices with carbon nanotubes
through nanorobotic manipulations," Proceedings of the IEEE, vol. 91, pp. 1803-1818,
2003.
[43] C. Zhou, Z. Gong, B. K. Chen, Z. Cao, J. Yu, C. Ru, et al., "A Closed-Loop Controlled
Nanomanipulation System for Probing Nanostructures Inside Scanning Electron
Microscopes," IEEE/ASME Transactions on Mechatronics, vol. 21, pp. 1233-1241,
2016.
[44] I. Utke, S. Moshkalev, and P. E. Russell, Nanofabrication Using Focused Ion and
Electron Beams: Principles and Applications, 1 ed.: Oxford University Press, 2012.
[45] C. Soong, P. Woo, and D. Hoyle. (2012) Contamination Cleaning of TEM/SEM
Samples with the ZONE Cleaner. Microscopy Today.
[46] J. Goldstein, D. E. Newbury, D. C. Joy, C. E. Lyman, P. Echlin, E. Lifshin, et al.,
Scanning Electron Microscopy and X-ray Microanalysis, 3 ed.: Springer US, 2003.
[47] C. E. Powers. (2016). Outgassing Data for Selecting Spacecraft Materials. Available:
https://outgassing.nasa.gov/
[48] M. J. Dykstra and L. E. Reuss, Biological Electron Microscopy: Theory, Techniques, and
Troubleshooting, 2 ed.: Springer US, 2003.
[49] S. Claesson and A. Å. Svensson, "A new ultramicrotome for electron microscopy,"
Experimental Cell Research, vol. 11, pp. 105-114, 1956/08/01 1956.
[50] J. Stirling, A. Curry, and B. Eyden, Diagnostic Electron Microscopy: A Practical Guide
to Interpretation and Technique, 1 ed.: Wiley, 2013.
[51] Studer and Gnaegi, "Minimal compression of ultrathin sections with use of an
oscillating diamond knife," Journal of Microscopy, vol. 197, pp. 94-100, 2000.
[52] (2016). Diatome Diamonds Knives. Available: http://www.diatome.ch/
[53] G. A. Kim, "Ultramicrotome Diamond Knife," United States of America Patent
US4581969 A, 1984.
[54] E. Oberg, F. Jones, H. Horton, H. Ryffel, and C. McCauley, Machinery's Handbook,
30 ed.: Industrial Press, 216.
[55] M. E. Merchant, "Mechanics of the Metal Cutting Process. I. Orthogonal Cutting
and a Type 2 Chip," Journal of Applied Physics, vol. 16, pp. 267-275, 1945.

58
[56] (2016). Delaware Diamond Knives, Inc. Available:
http://www.ddk.com/store/home.php
[57] Metal Cutting Physics. Available:
http://web.mit.edu/2.670/www/Tutorials/Machining/physics/Description.html
[58] D. H. Hall, E. Hartwieg, and K. C. Q. Nguyen. (2012). Serial Thin Sectioning - the
Hartwieg Method. Available:
http://www.wormatlas.org/EMmethods/STSHartwieg.htm#Hall2012
[59] B. Lich, X. Zhuge, P. Potocek, F. Boughorbel, and C. Mathisen, "Bringing
Deconvolution Algorithmic Techniques to the Electron Microscope," Biophysical
Journal, vol. 104, p. 500a, 2013.
[60] H. F. Helander, "Some observations on knife properties and sectioning mechanics
during ultramicrotomy of plastic embedding media," Journal of Microscopy, vol.
101, pp. 81-93, 1974.
[61] (2016). Actuators / Adjusters - Thorlabs. Available:
https://www.thorlabs.com/navigation.cfm?guide_id=5
[62] (2016). Piezoelectric Actuators and Drivers. Available: http://www.piezodrive.com/
[63] (2016). Piezoelectric Tutorial. Available:
https://www.thorlabs.com/newgrouppage9.cfm?objectgroup_id=5030
[64] N. Lobontiu and E. Garcia, Mechanics of Microelectromechanical Systems, 1 ed.:
Springer US, 2005.
[65] Y. Qin, Y. Tian, D. Zhang, B. Shirinzadeh, and S. Fatikow, "A Novel Direct Inverse
Modeling Approach for Hysteresis Compensation of Piezoelectric Actuator in
Feedforward Applications," IEEE/ASME Transactions on Mechatronics, vol. 18, pp.
981-989, 2013.
[66] C. Ru and L. Sun, "Hysteresis and creep compensation for piezoelectric actuator in
open-loop operation," Sensors and Actuators A: Physical, vol. 122, pp. 124-130, 7/29/
2005.
[67] S. A. Rios and A. J. Fleming, "Design of a Charge Drive for Reducing Hysteresis in
a Piezoelectric Bimorph Actuator," IEEE/ASME Transactions on Mechatronics, vol.
21, pp. 51-54, 2016.

59
[68] A. J. Fleming, "Charge drive with active DC stabilization for linearization of
piezoelectric hysteresis," IEEE Transactions on Ultrasonics, Ferroelectrics, and
Frequency Control, vol. 60, pp. 1630-1637, 2013.
[69] C. Lee and C.-Y. Wu, "Study of electrothermal V-beam actuators and latched
mechanism for optical switch," Journal of Micromechanics and Microengineering, vol.
15, p. 11, 2005.
[70] Y. Zhu, A. Corigliano, and H. D. Espinosa, "A thermal actuator for nanoscale in
situ microscopy testing: design and characterization," Journal of Micromechanics and
Microengineering, vol. 16, p. 242, 2006.
[71] M. L. Ericson and H. Lindberg, "A method of measuring energy dissipation during
crack propagation in polymers with an instrumented ultramicrotome," Journal of
Materials Science, vol. 31, pp. 655-662, 1996.



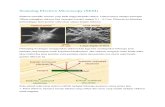















![Knife-Edge Scanning Microscopy: High-throughput Imaging and …jkwon/publications/files/choe.hpc08... · more advanced schemes such as multi-photon microscopy [3], optical sectioning](https://static.fdocuments.net/doc/165x107/5f787d3f59b36f6e7179727c/knife-edge-scanning-microscopy-high-throughput-imaging-and-jkwonpublicationsfileschoehpc08.jpg)