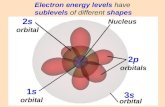
S- orbitals (l=0) p- orbital (l=1) d-orbital (l=2)
13
-
date post
22-Dec-2015 -
Category
Documents
-
view
242 -
download
0
Transcript of S- orbitals (l=0) p- orbital (l=1) d-orbital (l=2)
Electronic Configuration
Pauli exclusion principle – no two electrons in an atom can have the same four quantum numbers.
Electronic Configuration
Pauli exclusion principle – no two electrons in an atom can have the same four quantum numbers.
Shielding may cause some orbitals to over lap.
Electronic Configuration Pauli exclusion principle – no two
electrons in an atom can have the same four quantum numbers.
Shielding may cause some orbitals to over lap.
Hunds rule – the most stable arrangement of electrons in subshells is the one with greatest number of parallel spins