Reactions under acidic conditions Ian Suckling APPI 2012.
-
Upload
alfredo-hilditch -
Category
Documents
-
view
224 -
download
1
Transcript of Reactions under acidic conditions Ian Suckling APPI 2012.

Reactions under acidic conditions
Ian Suckling APPI 2012

Polysaccharides
• Polysaccharides hydrolysed to constituent sugars under acidic conditions
• Depends on:- physical structure and accessibility (esp. for cellulose)- conformation of sugar constituents- sidechains for hemicelluloses- hydrolysis medium & conditions
• Resulting sugars more stable than under alkaline conditions, but condensation and dehydration can occur
• Acetyl groups hydrolysed under acidic conditions, esp. at elevated temperatures- Released acetic acid lowers pH and can catalyse hydrolysis

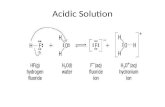
Hydrolysis mechanism
O
OR
CH2OH
OH
HO
OH
O
CH2OH
OH
HO
OH
O
OHCH2OH
HOHO
O
OR
CH2OH
OH
HO
OH
H
O
CH2OH
OH
HO
OH
Glucose+
Other products
H
- ROH H2O
- H
- H

Rates of acid hydrolysis
• In practice, heterogeneous hydrolysis rate differences are even greater:- Cellulose 1- Mannan 60- Xylan 60-80- Galactan300

Dehydration and condensation
• Acid hydrolysis can lead to formation of dehydration and condensation products, depending on hydrolysis conditions
OOHOH
HOOH
OOH
CH2OH
OH
HOOH
O CHO
furfural
condensation products
hydroxymethyl furfural
O CHOHOH2CHCOOH +
HOOC
O
levulinic acid
H+, -3H2O
H+, -3H2O

Hydrolysis of xylans
• Arabinofuranose units readily cleaved off softwood xylans • Glycosidic linkages of uronic acid group only partially hydrolysed so
get biuronic acids after hydrolysis• Uronic acid substituents also slow xylan hydrolysis
O
O
O
OHO
O
OH
O
O
O
O
OO
OH OH
OH OH
O
OHHOH2C
OHCOOH
OH
MeOOH
O
O
O
O
OHOH
COOH
OH
MeOOH
HO
Biuronic acid

Reactions of lignin
• Reactions under acidic conditions• Cleavage of - and -ether linkages• Cleavage of lignin-carbohydrate bonds• Release of formaldehyde due to sidechain cleavage• Condensation reactions
OR1
OR
OCH3
O
HOH3CO
Relative rates for hydrolysis of - and -ether lignin models in 0.2M HCl aqueous dioxane at 50ºC
Linkage Relative rate Type R =
-Aryl ether, R1 = H Nonphenolic CH3 1 Phenolic H 12 -Aryl ether, R1 = Aryl Nonphenolic CH3 65 Phenolic H 305

Acidic cleavage of -ether linkages
HC OR1
OCH3
OR
HC OR1
OCH3
OR
CH
OCH3
OR
HC OH
OCH3
OR
CH
OR
OCH3
-R1OH H2O
slow
Condensationproducts
R = HR1 = aryl, alkyl

Acidic cleavage of -ether linkages
HC OH
OR
OCH3
O
HOH3CO
CH
OR
OCH3
O
HOH3CO
HC
OR
OCH3
O
HOH3CO
HC OH
OR
OCH3
O
H3CO
CH2
CHO
OR
OCH3
OR
OCH3
CH2
O
HO
Guaiacyl
HO O
Guaiacyl
O
Guaiacyl
O O
GuaiacylH2C
O
HCHO+H
Hibbert's ketones
H
- H2O

Other reactions under acidic conditions
• Formaldehyde elimination from sidechain
• Condensation
- species that can trap the intermediate carbonium ion (bisulfite, phenols, thioglycolic acid, chloride) reduce condensation
- liberated formaldehyde can also participate in condensations
HO
H3CO
H2COH
OCH3OH
O H
HO
H3CO
OH
OCH3
+
HCHO
HO
H3CO
H3CO
CH3
OCH3
OH
OHHO
H3CO
H3CO
CH3
OCH3
OH
H2C+
1% HCl

Suppressing condensation reactions
• Addition of 2% of phenol and other additives can suppress lignin condensation- 2-naphthol most effective
CH
OH/Lignin
OMe
O
HOH2C
RO
Lignin
MeO
HC
OH/Lignin
OMe
O
HOH2C
Lignin
MeO
+ CH
OH/Lignin
OMe
O
HOH2C
Lignin
MeOOH
MeO
Lignin
Lignin
MeO
HO
H2C
OH/Lignin
OMe
O
CH2OH
CH
OH/Lignin
OMe
O
HOH2C
Lignin
MeO
OH
OH
H+
-ROH
+
30
A
B
C
OH
Lignin
MeO

Solvent pulping
• Separation of woods by treatment with organic solvents- Range of different solvents- Many include acid or alkali to
enhance pulping rates• Examples:
- Allcel – EtOH/water (1:1), ~190ºC, ~60 min
- Acetic acid/water pulping- Organocell – NaOH,
methanol, catalytic AQ• Requires v. efficient solvent
recovery

Vertichem process – an example