Oocyte Recovery Camel
-
Upload
govind-narayan-purohit -
Category
Documents
-
view
237 -
download
0
Transcript of Oocyte Recovery Camel
8/10/2019 Oocyte Recovery Camel
http://slidepdf.com/reader/full/oocyte-recovery-camel 1/3
f'
[niicn iaurncl
af
Aninc
S:ience:;
69
i?.t
:
iiS-90.
February
l999
Sfficiene;r
ci
three
methods
of
oocyte
recovery
in the
ci
rcrneelarl' camel iCamelus
dromedoriws)
C N i'ilROHITi.
IvIAHESH
DAIT: and S S
SHARMA3
isiesihan-
Agriculture
Llniversitl;,
Bikaner, Rajasthan
334
001
FLccrjved:
27 Februa-1'
1998;
Accepted:
26 August
1998
.{BSTRACT
(-)vari,:s
col tc..c<j
jr+rrr
,.riarl
fertale camels
of
Bikaner
and
surrounding
areas during
the
breeding
season
were usecl
for
{roc:icc..il,iriion.Fr:-rnitite
leftorei
1Ifollicleswerecolleciedcomparedtoi9folliclesfromrightovary'.Ontheovarian
surfae
e
ifl
ot
;ire 62
ibliicles i.,
ere haling
erther
blood ciots
or haemorrhagrc
margins.
Follicles
were well
prcjecting on the
c..,3ilan
si,.t'ace
*:i lilir
c-,ciallmean
diameter
*
as
9.63t3.50
mrn.
36i 5b
recovered oocytes were
not having
any curnulus
mass:ltr-arit:ij
rc
it. Arnr.'rist
ih: 3
irieihods
used
for oocyte
collection,
aspiration
was most
efficient
(32
ooc-\tes
y'ielded
framu\oyaiies)
cr.;lr':il:edtcf,rliicle
di:,section(i-loccytesfrom5ovaries)orslicing(l0oocytesfromT
ovaries).Slicing
was con-.rd*rerl
i+
b,e
non-suitable
irr
ccilectioa
of camel ooc]'tes
because of
the
presence
of haemorrhagic
folliclss.
Key
words: Camel.
Follicles,
Oocyte
in.;irr',
l'li'tilizatiln
is
sttil
in
the
experiinental
stage
in
the
camelid.ie.
l,in:bivcs
rver*
pr"Cu:ed
iit v'itt'c
irorn
llama ooc;vtes,
hcr\y.ivei'.
ilter€
are stili
no reilort?d
llve +ffspring
from
e:nbr-yos
proci,:ceC
ii,"
vttt,s,
T' re
foilieular
acti'/it)'
is a
contiiluous
ptuctsss
i.n il;,:
aniieal
r-lilring
the
breeding
season
anei
when nct
n;aied.
ii3
i6'ilicle:;
become
attretic.
rvhile,
continuousi-y
olh.,::'
foliicies
matuie and
gii,rw
even in
ihe
presence
cfair
aetive
ccrpus
luteutii
(llravo
1994.
Tibary and
Aiouas-si
i996
).
Bou
etc;/.
(199j)
i:r
China
described
srnall-
scale
studies
o;t
invltra
ienilizalicn
in rhe
Arabian
camel
on
ovaries
trom :laugi-:iereC
aniin;,is.
v.heieas Dei Campo
er
ai.(1994a)
d*scr.ii,ed
lhe
same
in iiamas.
in','ivrt
collection
of
oocytes
far inviiit
ierliiizl:iion-*'as
described
b1'Brogliatti el
al.{1996}.
Tiirary ar:'i
,1-rr{)i.ia:r;
(
i
t97). An
aftempt
ri'as made
to coliect
the camel o-.'alies
from
recenill
clied animals
and
asses
the
e fficiency-
of
iiifferent
medrcds
cf
ooclte
retrieval,
MATERIAI-S
AND TVIETHODS
'
Ovarie-s
were
cr,llected
from
dead
f-emale
dromedaries
witiiin 6
to 121rr *f-death
from
Bikaner
ruea
and its
sunoundings.
The
cornplete
gt'nitalia *'ere ccllected
wasi:ed
and
brought
to
the
laborar.;ry
in
werm nor:nal
sallne.
fort;fred
ri
ith antibiotics.
The
ovaries
$
ere sepaiatecl
rn
the laboratcry
and
the number
of
follicles
present on
the respeciive
cvaries.
their
position
and
diameters
were
tLoted down.
The
oo.\'tes
were collected
Present
address:
jr
Assrstant
I-'ioiesir,'r.
r
Professor,
Departmenl
crf
Obstetril.r
att,j G)'naecology.
College
of
Veterinarv
and
Animal
Science.
using
TCM-199
fortified
with
antibiotics
and
bovine
seru
albumin,
by
aspiration,
follicular
dissection
and
slici
methods
used
for cattle
(Katska
and
Smorag
1 984)
arid buffa
(Das
er
al.
1996).
The morphology
of
oocy'tes
was observ
under
inverted
phase
contrast
micrcscope.
Only
good
siz
follicles
(>8mm
diameter) were
used
for
follicular
aspirati
and dissection.
RESULTS
AND
DISCUSSION
Follicles,
28
out
of 62
that
were
seen
on
the ovaries
we
having
either blood
clots or
haemorrhagic
margins
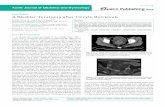
(Fig'
This
peculiar
feature
posed
a
great
difficulry
in collection
a
assessment
of the oocytes.
In the
absence
of
mating
ovulation
inciucing treatment,
the fate
of the maiure
follic
follows
one of the
two
possible
courses;
atresia
or cys
degeneration
(Tibary
and
Anouassi
i996).
The incidence
anovulatory
follicles
in
non-bred
dromedaries
may range
fro
35
to
50
%
(Skidmore
et
al.1995).
On the basis
ofpalpabie
a
Table
l.
E,fficiency
of three
methods of oocyte
recovery in
cam
Ovaries/ooc1'tes
Method
of collection
Total folli
presen
Aspiration
Slicing
Puncture
-r.*----J"
r
ovary
ov
No.
of ovaries
taken
8
Recovery ofoocytes
32
7643
10
14
8/10/2019 Oocyte Recovery Camel
http://slidepdf.com/reader/full/oocyte-recovery-camel 2/3
st
I
OOCYTE
RECOVERY IN THE DROMEDARY
CAMEL
89
ebruary
19991
Figs 1-3.
1
Camel
ovaries
with
haemorrhagic
follicles
and
mii.rgins.
2.
Photo-micrograph
shorving
camel oocy'tes
*'ithout
cumulus
mass. 3.
Photo-micrograph
shorving
camel oocl'tes
rl
ith
cumulus
mass.
ultrasono
graphic
characteri
stics,
Tibary
and
Anouassi
(1
997
)
defined
5
categories
of
anolulatory
follicles
in
camelids,
which
included
thick-walled
strucfure
with blood clot
and
fibrin
sftands
within
the
cavity
(haemorrhagic
foilicle).
This explains
that the
follicles
with blood
clots or haemorrhagic
margins
observed
during
this study
were
anovulatory
regressing
follicles,
however,
it
is
not known
for certain
why
some
of
the
follicles
develop
into
these large anolulatory,&aemonhagic
follicles
and others
do
not.
The
overall
mean
diameter of
the
follicles
on the ovarian
surface was 9.63*3.50
(range
2-16 mm) ttre mean diameters ar
lower than those reported by Homeida
et
al.
(1988)
by recta
palpation
and
by Tibary
et
al.
(1996)
by
ultrasonography in
the
dromedary
camels,
moreover,
the
upper
and
lower limits
o
the
follicular diameters found during this sfudy were very low
compared
to
reports
of
Homeida et
a/.(
1988) andTibary et al
(1996),
this
could be due
to
difference
in
the
method o
assessment of follicular diametrics
and the
fact
that the
ovarie
were
collected
from
dead camels
whose cause
of
death
wa
not
investigated during this study. Skidm
ore et
al.
(
1
996)
have
however, reported the
follicular
diameter
of 5-9 mm in dromedary
camels.
The
follicles
were
well
projecting
on
the
ovarian
surfac
except
3 thick follicles
which were noted to be deepiy embed
ded, and,
not
taken into account during
this study. The recov
ered oocy'tes
(6a%;
35156)
were denuded and
did not hav
any cumulus
mass attached
to it
(Fig.
2). The shrunken natur
of the cytoplasm
and the
dark
inner cellular
mass in
Fig.
3
(an
a
common
finding)
could
be because, although,
due care
ha
been taken
in
collecting
the
genitalia
from
dead camels
in
time
but
the
time
lost from death of
the camels
to
intimation fo
collection
was
beyond
control
and
probably
the oocyes
rver
dead
by that
time.
Del Campo et
al.
(1994b)
reported that
17
52oh
ofthe oocytes
are
degenerated
after
recovery from th
follicles.
By careful aspiration
4.0 oocytes
per
ovary
(3214
could
be
recovered
whereas by
follicular dis;ection
only 2.3
(i4l6)
oocytes could
be recovered
and by slicing
only 0.
ooc)'tes
could
be recovered
per
ovary
(Table
1).
3licing
metho
was
all
the more unsuitable
because
plenty
of blood
rva
extravasated into the
medium of collection
by the
haemonhagi
follicles.
Primarily follicular
aspiration can
be
considered a
the
most suitable
method for retrieval
of
camel oocytes
however, more sfudies
need to be done.
Methods of oocy
retrieval have been sparsely
reported
in
camelids
except
th
in vivo methods
like transvaginal ultrasounil
guided
method
and
hence comparison of the
present
study
was not
possible
Horvever, our results
are
at
variance
with
those
of
Del
Camp
et al.
(1994
a)
who reported an
average
recovery
of6.4
oocyte
per
llama
with
the
slicing method.
REFERENCES
Bravo
P W.
1994.
Reproductive endocrinology
of liamas an
alpacas.
Veterinary
Clinics
of
North
America. Food Anima
Practice
l0
265-79.
Das G K. Jain G C. Solanki
V S and Tripathi
V
N.
i996. Efficienc
of
various collection method
for oocyte retrieval in buffalo
TheriogenologY
,16(8):
1403-l
1.
Del
Campo
M
R, Del
Campo
C
H,
Donso
M X
and Berland
M
1994a.
In
t;itro fertilization
of
llama
(Llama
glama)
follicula
oocytes.
Theriogenology
4l: 18'7.
Del
Campo
M R. Del Campo C
H,
Donoso
M X,
Berland
l\{
an
Mapletoft
R
J.
1994b.1n
vitro
fertilizatron and development
llamas
(Llanta
glama)
oocytes
using epididymal
spermatozo
and oviduct
cell-culture.
Zlr eriogenology
4l:1219- 29
.
Homeida
AM.
Khalil
M.R
and Taha
A A M.1988.
Plasm
8/10/2019 Oocyte Recovery Camel
http://slidepdf.com/reader/full/oocyte-recovery-camel 3/3
90
PUROHIT
ETII.
[Vol.
69, No.
concentrations
of
progesterone. estrogens,
testosterone
and
LH like
activity
during the
estrus c1'cle
of the
camel
(Camelus
dromedarius).
Journal
of
Reproduction
and Fertilit.v
83:593-98.
Katska
L
and Smorag
Z.
1984.
The
possibilities of
recovering
cattle
ooc),
tes.
LI
oz
l iw o s c
i L z
"v
s
kiw ani a
O
oc.r
t
ov B
"v"
d
I e c.," c
h
M e
d1,cy
na
LI/e tery naryi
na 40(8)
:
499-5 02.
Skidmore
J
A, Billah
M and
Allen
W
R. 1995, The
ovarian
follicular
wave
pattern
in
the
mated
and
non-mated
dromedary
camel
(Canrelus
dromedarius).
Journal
of Reproduction
and
Fertility
(Suppl)
49:545-48.
Skidmore
J A, Billah
M and Allen
W R. 1996. The ovarian
follicu
wave
pattern
and
induction of ovulation
in the
mated and no
mated one-humped
camel
(.Came
lus dr ome
dar
ius).Journal
Reproduction and
Fertility
106:
I
85-92.
Tibary A
and
Anouassi
A. 1996. Ultrasonographic
changes of
t
reproductive tract
in
the
female camel
(Camelus
dromedariu
during
the
follicular
phase
and
pregnancy.Journal of
Cam
P ractice and
Re
se arch
3 : 7 l-90.
Tibary A and
Anouassi
A. 1997.
Theriogenology
in Camelida
pp.l75-79.
Abu
Dhabi
Printing
and
Publishing
Company.
Mi
Abu
Dhabi. United
Arab Emirates.