Lecture 5.1 – Intermolecular Forces and the Dissolving Process
description
Transcript of Lecture 5.1 – Intermolecular Forces and the Dissolving Process

Lecture 5.1 – Intermolecular Forces and the Dissolving Process

Today’s Learning Targets• LT 4.1 – I can discuss how ionic solids dissolve due to the
random molecular motion of the water molecules. • LT 4.2 – I can identify a compound as containing
hydrogen bonding, dipole – dipole forces, and Van der Waals forces.
• LT 4.3 – I can compare and contrast various intermolecular forces and relate them to the dissolving process of a solid.

What do solids, liquids, and gases look like?


I. Solids• Atoms in a solid hold a very fixed position. • They are said to be vibrating in place.

II. Liquids• Atoms in a liquid are further spread out from
one another.• They flow freely within the solution and are
moving in a random motion.

III. Gases• Atoms in a gas are in constant random
motion.

Most forces
Least forces

What are intermolecular forces?

I. Intermolecular Forces• Intermolecular forces are forces that attract molecules
to one another. • The three main types of intermolecular forces are:
(1) Hydrogen Bonding(2) Dipole-Dipole Forces(3) Dispersion Force (also known as Van der Waals
Forces)

II. Dipoles• A dipole is a separation of charge. • Molecules can have a separation of charge due to
electronegativity• This causes the electrons to be held closer to the
element with the higher electronegativity.

H F
FH
Pulls electrons towards itself because it has a higher electronegativity
Partial negative charge
δ-δ+
This is what a dipole looks like

III. Dipole-Dipole Forces• Dipole-dipole forces refer to the attraction
between molecules that have a permanent dipole.

What Dipole-Dipoles Look Like
Partial negative attracted to the partial positive

III. Dispersion Forces• Some molecules have an induced dipole. • Dispersion forces are the intermolecular
forces resulting from the uneven distribution of electrons and the creation of temporary dipoles.
• Weakest intermolecular force

There is no electronegativity difference between two hydrogens, so they are non-polar and do not have a dipole
But, when two H2 molecules are put next to one another, a dipole is induced because electrons rearrange themselves

What the Heck?

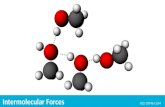
IV. Hydrogen Bonding• Hydrogen bonding is the attraction of one molecule
that contains hydrogen to another molecule. • When hydrogen is bound to N, O, or F, there is a
large difference in electronegativity • Creates a partial positive and negative charge• The partial positive charge on hydrogen is attracted
to N, O, or F of another molecule.

Hydrogen bond between H and O


V. Intermolecular Forces and Phase of Substance
• The degree of intermolecular forces determines the phase of a substance.
Least intermolecular forces
Most intermolecular forces