Lecture 2: Matter, Elements and Compounds, States of Matter, Physical and Chemical Changes
-
Upload
nsanusi -
Category
Technology
-
view
6.050 -
download
4
Transcript of Lecture 2: Matter, Elements and Compounds, States of Matter, Physical and Chemical Changes

ELEMENTS AND COMPOUNDS STATES OF MATTERPHYSICAL AND CHEMICAL PROPERTIES AND CHANGES
Lecture for 9/11/2007

What is matter?
Anything that has mass and volume
Ancient Greece and India – intrigued by the origin of matter and the makeup of the universe
Is there anywhere that doesn’t contain matter?

States of matter
Solid – holds a particular shape and has definite volume
Liquid – doesn’t hold its own shape but it does occupy a definite volume
Gas – has no definite shape or volume Plasma – exists inside stars and forms
briefly on Earth when electrical sparks separate atoms into their component parts.
Changes in state - Heating or cooling a substances

Properties of matter
Physical properties Characteristics of a substance that can be
observed without altering the identity of the substance
Chemical properties Characteristics of a substance that cannot be
observed without altering the substance

Physical and Chemical Changes
Physical changes – changes that don’t alter the identity of a substance
Chemical changes – alter the identity of the substance
Identify the following as physical or a chemical change:
Sugar ferments to form alcohol. Gallium metal melts in your hand. Platinum reacts with hydrogen peroxide
oxygen at room temperature. Leaves turn color in the Fall.

Identify the following as physical or a chemical change CON’T:
Milk turns sour. Wax is melted over a flame and then
catches fire and burns. You make scrambled eggs. You step on a piece of chalk and it
becomes powdered. You light a candle when the electricity
goes out. Steam from your hot shower condenses
on a cold mirror.

Law of conservation of matter
Antoine Lavoisier (1743 – 1794)
Matter, like energy, is neither created nor destroyed in any process

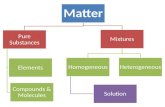
Elements and Compounds

Distinguishing Between Elements and Compounds
Both elements and compounds are pure substances
Difficult to determine when a more “fundamental” element was reached
Tear apart substances to see if a more simpler element can be reached