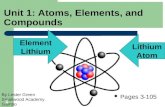
Joseph Kings Lithium Atom Please press the space bar and hold It down to view the atom.
-
Upload
leslie-craig -
Category
Documents
-
view
223 -
download
1
Transcript of Joseph Kings Lithium Atom Please press the space bar and hold It down to view the atom.

Joseph King’s Lithium Atom
Please press the space bar and hold It down to view the atom.





Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium

Protons the number is 3 to make up Lithium






















Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4
Protons the number is 3 to make up Lithium

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

Neutrons the number is 4 NO CHARGE
Protons the number is 3 to make up LithiumPOSITIVE CHARGE4 Neutrons + 3 Protons = 7
THIS IS THE ATOMIC WEIGHT

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.

And now for the ELECTRONS!
The Electrons have very little weight and a negative charge. They equal the number of Protons.