Ionic bonding
-
Upload
felipe-de-oca -
Category
Technology
-
view
791 -
download
2
Transcript of Ionic bonding

Ionic Bonding

Why do atoms of different elements combine to form
Ionic Compounds?
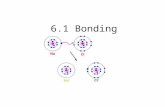
Sodium Chlorine
Sodium Chloride

Why do atoms of different elements combine to form
Ionic Compounds? • Human beings need to be complete in
order for them to be happy.
Ionic compound• Consist of a metal and non-metal element• Atoms of elements bond together in order for them
to obtain a complete set of valence electrons in their outermost energy level.– By obtaining eight valence electrons they become
stable by satisfying the Octet Rule.

Charges and Number of Valence Electrons
1A 2A 3A 4A 5A 6A 7A 8A
(1) (2) (3) (4) (5) (6) (7) (8)


- A system of representation wherein dots are used to show the number of valence electrons in atoms and molecules- Named after Gilbert Newton Lewis
Lewis Dot Electron Structure

Lewis Electron Structure

The Ionic Bond
Formation
– One atom of an element losing its electron to an atom of another element is known as the electron transfer.
– Ex. the metallic atom Na, gives one electron to a nonmetallic atom F. In the process, Na acquires a positive charge while F acquires a negative charge.
– Both achieves noble gas configuration.– Oppositely charged ions are formed whenever a metal
combines with a nonmetal.

Ionic Bonding
Example….• MgO, magnesium loses two electrons to form a +2
cation, while oxygen adds two electrons to form a -2 anion.

Chemical Formula of Ionic Compounds
• Charges are used to predict the chemical formula of Ionic Compounds.
• The charge of a metal element and the charge of a non-metal element must be equal to zero when combined.– If not trial and error is applied to come up
with the sum which is zero.• Metal elements are written before the non
metal elements.

Writing Chemical Formula
• Aluminum, the more metallic element, is written first.
• Oxygen is written last since it is less metallic
• There are 3 atoms for oxygen, thus a subscript of 3 is written after the symbol while aluminum has 2.
Aluminum Oxide
Al2O3

Writing Chemical Formula
More examples:1. Magnesium and Sulfur2. Potassium and Nitrogen

Naming Simple Ionic Compounds
• Examples:Mg and O2 MgO2
Rule:1. Name the metal (Mg is Magnesium)2. Name the nonmetal ending in –ide (O2
is Oxygen + -ide = oxide)3. MgS is Magnesium oxide

Naming Simple Ionic Compounds
1. KI -
2. CaBr2 -
3. Al2O3 -
1.Potassium iodide
2. Calcium bromide
3. Aluminum oxide