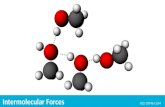
Intermolecular Forces (Gaya Antarmolekul)
-
Upload
state-university-of-yogyakarta -
Category
Education
-
view
1.491 -
download
6
description
Transcript of Intermolecular Forces (Gaya Antarmolekul)

1
Intermolecular forces
Apr 10, 2023

Apr 10, 2023 2
Overview
attraction in molecules
intramolecular bonds
intermolecular forces

Apr 10, 2023 KARBOHIDRAT 3
Intermolecular forces is much weaker than intramolecular forces

4
INTERMOLECULAR FORCES ---- attraction that exist between molecule
1. GAYA VAN DER WAALS
• GAYA DIPOLE-DIPOLE• GAYA LONDON (DISPERSI)
2. IKATAN HIDROGEN
Apr 10, 2023

Apr 10, 2023 5
1. Gaya dipole-dipole....dipole-dipole force2. Gaya london (dispersi)....dispersion force4. Ikatan hidrogen......hydrogen bonding5. Ikatan ionik....ionic bonding6. Jaringan ikatan kovalen.....covalent network

6
What should be discussed :1. What ?2. When?3. Example?4. Power?
Apr 10, 2023

Apr 10, 2023 7
London dispersion force
•The attraction between atoms and molecules caused by the formation of instantaneous atoms in the atoms and molecules
•Two non polar molecule
•Caused by momentary dipole

Apr 10, 2023 KARBOHIDRAT 8

Apr 10, 2023 KARBOHIDRAT 9

Apr 10, 2023 KARBOHIDRAT 10
polarizability
• the ease with which an atom or molecule can
be distorted to have an instantaneous dipole.

Apr 10, 2023 KARBOHIDRAT 11
Polarizability increases with: greater number of electrons
Dispersion forces usually increase with molar mass.

Apr 10, 2023 12
Effects on boiling points

Apr 10, 2023 13
Dipole-dipole force
• The force that exist between negative and positive ends of a polar molecule.
• Between two polar molecule
+ +- -
Opposite attraction

Apr 10, 2023 KARBOHIDRAT 14

Apr 10, 2023 KARBOHIDRAT 15
Hydrogen bonding
• A form of dipole attraction in which hydrogen atom bonded to a strongly electronegative atom
• H + F, O, N .... HF, NH3, H2O• Works like dipole-dipole but 5 times stronger

Apr 10, 2023 KARBOHIDRAT 16
Ionic bonding
• The forces holding ions together in ionic solids are electrostatic forces

Apr 10, 2023 KARBOHIDRAT 17
•100 % covalent bond
•There are no intermolecular force
•Only compounds of C only (diamond, graphite)
and Si (usually with Oxygen)
• very strong attraction

Apr 10, 2023 KARBOHIDRAT 18

Apr 10, 2023 KARBOHIDRAT 19

20
power
Jaringan ikatan kovalen > Ikatan ionik > Ikatan hidrogen > dipol-dipol > gaya london
Apr 10, 2023