Intermolecular Forces and Solubility
Transcript of Intermolecular Forces and Solubility

TUESDAY REVIEW SESSIONOCTOBER 4 , 2010
Intermolecular Forces and Solubility

Intermolecular Forces
Definition: Forces between moleculesIntermolecular forces are much
weaker than intramolecular forces
There are three different typesLondon Dispersion Forces/ Van der
WaalsDipole-Dipole InteractionsHydrogen Bonds

London Dispersion Forces
Also referred to as Van der Waals forces
Occurs because of momentary dipole (uneven distributions of electrons in a molecule causing a partially negative end and a partially positive end)
These are the weakest intermolecular forces

Dipole-Dipole Interactions
Occurs when polar molecules align so that their oppositely charged ends are attract each other
This bond is stronger than Van der Waals forces

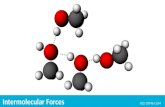
Hydrogen Bonding
A type of dipole-dipole bond that occurs when hydrogen is bonded to a highly electronegative atom (O, F, N)
This is the strongest intermolecular force

Intermolecular Forces Trends
Compounds with stronger intermolecular forces will be harder to separate therefore they will have larger melting points and boiling pointsThis means compounds with weak
intermolecular forces are more likely to be found in gaseous form, while compounds with stronger molecular forces are more likely to be found as liquids or solids