Intermolecular Forces
description
Transcript of Intermolecular Forces

Courtesy: labinitio.com

A phase is a homogeneous part of a system in contact with other parts of the system but separated from them by a well-defined boundary.
2 Phases
Solid phase - ice
Liquid phase - water

Gases:•Assumes the volume and shape of its container.•Low Density•Very Compressible•Very Free Motion

Liquids:•Has a definite volume but assumes the shape of its container.•High Density•Only Slightly Compressible•Molecules slide past one another easily.

Solids:•Has definite volume and shape.•High Density•Virtually Incompressible•Molecules vibrate about a fixed position.

•Intermolecular forces are attractive forces between molecules.•Intramolecular forces hold atoms together in a molecule.Intermolecular vs Intramolecular
• 41 kJ to vaporize 1 mole of water (inter)
• 930 kJ to break all O-H bonds in 1 mole of water (intra)
Generally, intermolecular forces are much weaker than intramolecular forces.
“Measure” of intermolecular force
boiling point
melting point
Hvap
Hfus
Hsub

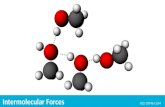
Types of Intermolecular Forces1. Hydrogen Bond (strongest)The hydrogen bond is a special dipole-dipole interaction between the hydrogen atom in a polar N-H, O-H, or F-H bond and an electronegative O, N, or F atom. IT IS NOT A BOND.
A H … B A H … Aor
A & B are N, O, or F

Figure (a): The polar water molecule.Figure (b): Hydrogen bonding among water molecules. Note that the small size of the hydrogen atom allows for close interactions.

Hydrogen Bonding
Electrostatic potential diagrams – red indicates the most electron-rich region and blue indicates the most electron-poor region.

•Hydrogen bonding has very important effect on physical properties.•Note that the nonpolar tetrahedral hydrides of Group 4A (14) show a steady increase in boiling point with molar mass (i.e., going down the group).•But in the other groups the lightest member has the highest boiling point.

Why do H2O, HF, and NH3 break the trend of decreasing molar mass = decreasing boiling point?
Unusually strong hydrogen bonding forces due to two factors: 1.relatively large electronegativity values which leads to especially polar X-H bonds 2.small size of the element, which allows for the close approach of the dipoles, strengthening the intermolecular forces.
Hydrogen bonding is so strong it takes a great amount of energy to overcome attraction and separate the molecules to produce the gaseous state.

Types of Intermolecular Forces
2. Dipole-Dipole Forces
Attractive forces between polar molecules

Types of Intermolecular Forces3. London Dispersion Forces – (weakest)
Attractive forces that arise as a result of temporary dipoles induced in atoms or molecules
ion-induced dipole interaction
dipole-induced dipole interaction

Intermolecular Forces3. London Dispersion Forces Continued
Polarizability is the ease with which the electron distribution in the atom or molecule can be distorted.
Polarizability increases with:
• greater number of electrons
Dispersion forces usually increase with molar mass.