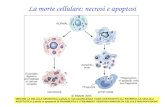
Il ruolo del PTP in apoptosi/necrosi. Some characteristics common to several diseases: Oxidative...
-
Upload
julia-flood -
Category
Documents
-
view
222 -
download
1
Transcript of Il ruolo del PTP in apoptosi/necrosi. Some characteristics common to several diseases: Oxidative...

Il ruolo del PTP in apoptosi/necrosi

Some characteristics common to several diseases:
Oxidative damageChanges in Calcium homeostasisLoss of energy production Cell death
Tissue and organ dysfunction
Disease

The Mitochondrial connection.
Mitochondria are key integrators of cellular signals and stress: make life and death decisions
Apoptosis and necrosis are facilitated at the level of the mitochondria by the opening of large pores (excluding extrinsic apoptotic pathway)
c c
cc
c c
mPTPBax/Bcl2 Swelling, rupture
Release of proapoptotic factors
IMM
OMM

• Mitochondrial Permeability transition (mPT) characterized by:
• Loss of inner membrane potential;
• Release of Cytochrome C;
• Cell death by apoptosis or necrosis;
• Cessation of mitochondrial respiration;
• Release of mitochondrial Ca 2+;
• Mitochondrial swelling (rupture)
• The mPT can be caused by several agents/mechanisms (e.g. ROS, Ca2+ overload);
• The mPT, and as a consequence, mitochondrial dysfunction, is associated with the onset and progression of several diseases.
ANT: Adenine nucleotide transporter, VDAC: voltage dependent anion channel, HK: Hexokinase; CyD: Cyclophilin D, CK: Creatine kinase, PBR: Periferal benzodiazepin receptor
The Mitochondrial Permeability Transition.

Recenti sviluppi nella identificazione delle componenti del PTP
La ATP sintetasi, maggiore produttore cellulare di ATP nella cellula, è stata recentemente proposta come l’unico componente molecolare necessario per la formazione del PTP: questo dato è ancora da confermare in maniera definitiva, ma enfatizza la funzione duale dei componenti della catena respiratoria nel controllo di apoptosi e necrosi (che ritroveremo con il citocromo C)

Consequences of mPTP opening
mitochondria swell and outer membrane rupture
Ca++ andprotein release
(eg. Cytochrome c)
mPTP openingsolutes
across mPTPenter mitochondria
mitochondria swell and outer membrane
ruptures
stimulus
loss of membranepotential/energy
production
Cell death

• Opening of mPTP has been implicated in the aetiology and progression of several diseases including :
• Neurodegenerative diseases (Parkinsons, Alzheimers, MS, Huntingtons, ALS)
• Ischemia / reperfusion injury (AMI, Stroke, organ transplantation)
• Dystrophies (Bethlem, Ullrich, Duchene)
• Diabetic Complications (retinopathy, nephropathy)
• Oxidative damage, irregular calcium signalling and mitochondrial dysfunction is common to all of these diseases
The mPTP and Disease.

Take a closer look at Ischemia / reperfusion injury
(from a Mitochondrial standpoint only)

• Heart ischemia leads to infarct size of > 70%.
• Reperfusion of tissue can reduce infarct size.
• Paradoxically reperfusion also leads to tissue death and contributes to infarct size (LRI).
• Methods to reduce lethal reperfusion injury should have clinical benefit
Lethal reperfusion Injury.

PTP in Ischemia / Reperfusion Injury
What’s happening in the the cell and mitochondria:
Ca2+
Calcium enters mitochondria via the Calcium Uniporter (not saturated at High [Ca2+])
Ca2+ Uniporter
Ca2+
Normal conditions
Calcium is pumped out of the mitochondria via the Sodium / Calcium Antiporter (saturated at High [Ca2+])
Na/Ca2+ Antiporter
Mitochondria Cell
Under conditions of high cytoplasmic [Ca2+] the mitochondria can overload with calcium and this can cause the mPT.

PTP in Ischemia / Reperfusion Injury
Ischemic conditions
Mitochondria Cell
Loss of ATP production via respiration; increase in lactic acid and a drop in cellular pH.
pH [H+]
H+
H+
H+
H+
H+
H+
Mitochondria Cell
To counter the high [H+] the cell uses the Na+/H+ Antiporter. Due to lack of ATP the Na+ can not be pumped out and the cell loads with Sodium.
Na+/H+ Antiporter
Na+
Na+
Na+Na+
Na+Na+
Na+
Na+
Na+
H+
Ischemic conditions
Mitochondria Cell
High intracellular [Na+] causes reversal of the Na+/Ca2+ Antiporter and the cell loads with Ca2+.
Na+
Na+/Ca2+ Antiporter
Ischemic conditions
Ca2+
Ca2+Ca2+
Ca2+Ca2+
Ca2+
Ca2+Ca2+
Ca2+Ca2+
Ca2+
Cell
Restart to respiration. Large burst of ROS production. Mitochondria load with calcium via uniporter and saturate the Na+/Ca2+ antiporter.
Reoxygenation conditions
Ca2+
Ca2+
Ca2+ Uniporter
Na/Ca2+ Antiporter
Ca2+
Ca2+
Ca2+Ca2+ Ca2+
Mitochondria Mitochondria Cell
Calcium overload and oxidative damage from ROS production induce opening of the mPTP and mitochondrial dysfunction.Cell death via necrosis/apoptosis depending on damage.
Reoxygenation conditions
mPTP
Ca2+
Ca2+
Ca2+
CytC
CytC
CytC
Cell DeathNecrosisApoptosis
Mitochondria

Analisi di topi KO per la ciclofilina D

I topi KO hanno un difetto nell’apertura del PTP, ma sono normali
Apertura del PTP in mitocondri isolati

Cellule PTP -/- sono resistenti alla morte cellulare indotta da ROS (H2O2)

I topi KO sono resistenti al danno cardiaco indotto da ischemia-riperfusione

New cardioprotective strategies.
From Yellon and Hausenloy (2007 NEJM)
mPTP inhibitors could be a promising strategy for lethal reperfusion injury.

Inibitori del PTP (CsA) sono anche efficaci nella riduzione del danno post-ischemico dell’Uomo
Marker sierico Area infarto (NMR)

The mPTP is involved in the pathogenesis of several aging-associated diseases
• Studies using CsA and Ppif-/- mice (cyD null) have highlighted a potential role for the mPTP in the progression of several diseases.
Ppif -/- mice and CsA
Stroke
Korde et al, 2007
Coronary Artery Bypass GraftHeart Disease
Transplantation and Surgery
Reperfusion Injury
Alzheimer’s Disease ALS
Neurodegenerative Disease
Duchenne MD
Millay et al, 2008
Dystrophies
Diabetic Retinopathy HyperglycemiaDoxorubicin toxicity Platelet activationTraumatic brain injuryNon-alcoholic fatty liver disease
Other
Myocardial infarction
Piot et al 2008
Ullrich / Belthem
Du et al, 2009 Keep et al, 2001
Palma et al, 2009

L’identificazione di nuovi farmaci inibitori dell’apertura del PTP

A large network of industry/academia interactions
Padua:P BernardiF Di Lisa
Milan:CONGENIAPG PelicciR LatiniNikemNMS
HamburgOxford:EVOTECLyon:
M Ovize

Ca2+ 150 μM
1 mM CsA
ControlAbs
orba
nce
(A 5
40 n
m)
Time (mins)0 5 10
Innovative HTS
• A high pulse of Ca2+ (ca. 150 mM) given to purified mitochondria will cause mPTP opening and induce mitochondrial swelling (DA540nm).
• A Ca2+-induced swelling assay was developed suitable for HTS• Fresh Rat liver mitochondria were prepared daily and used in this assay
to screen >300,000 compounds • Counterscreens were run to elimate compounds that interfere with mitochondrial respiration.• Several chemical classes were identified
•Organelle based•Requiring the use of animals->liver mitochondria
unprecedented at this scale

Adaptation of mitochondrial assays to 1536 well format
• Parameters to optimize:• density / fitness of mitochondria
• concentration of stimulus (Ca++)
• assay sensitivity (CysA)
• reagents volumes / order of addition
• reagents incubation times
• reagents dispensing devices
• readout kinetics / stability of assay signal
• DMSO-sensitivity
• reader settings (i.e. OD filter sets)
Results of time-course experiment in 1536 well plates:
4 Fitzones Z‘: 0,74 / 0,76 / 0,70 / 0,61
Mitochondria: 1 mg/ml
Calcium: 300 µM
Incubation: 10 min
Cyclosporin A: 1 µM

HTS-Hardware in Operations
2 EVOscreen® Mark II: biochemical assays at 1 µL level 1 EVOscreen® Mark III: biochemical & cellular assays at 1-10 µL level

Class 6 compounds inhibit the mPT induced by:- Calcium overload- Oxidative damage- Protein crosslinkers- Respiration uncouplers
300μM Diamide,
40μM Ca2+, pH 7.2,
100μM Menadione
40μM Ca2+, pH 7.4,
50nM FCCP
40μM Ca2+, pH 7.4,
A 5
40 n
m
Time (mins)
Treated
GNX-A
Treated
GNX-A
Assay performed on prepared mouse hepatic mitochondria
Ab
sorb
ance
(A
540
nm
)
Time (mins)
Ca2+ 150μM, pH 7.4
Treated
GNX-A
Untreated
A 5
40 n
m
Time (mins)
Treated
GNX-A
A 5
40 n
m
Time (mins)
UntreatedUntreated Untreated
PTPi block mPTP openingby several stimuli

State 3 inhibition (with ADP)
[GNX-C] (mM)
% m
ax in
hib
ition
-30-20-10
0102030405060708090
100
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21
State 4 inhibition
[GNX-C] (mM)
% m
ax in
hib
ition
3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21-20-10
0102030405060708090
100
0 1 2
0.0
10.0
20.0
30.0
40.0
50.0
60.0
70.0
80.0
90.0
100.0
0 5 10 15 20 25
-10.0
0.0
10.0
20.0
30.0
40.0
50.0
60.0
70.0
80.0
90.0
100.0
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21
State 3 inhibition (with ADP)
[GNX-B] (mM)
% m
ax in
hib
ition
State 4 inhibition
[GNX-B] (mM)
% m
ax in
hib
ition
PTPi do not have significant effects on mitochondrial respiration

The Calcium Retention Capacity (CRC) of purified mouse liver mitochondria is determined by measuring the point at which pulse-loaded calcium is released from the mitochondria. After the addition of a pulse of calcium (10 mM) the extramitochondrial fluorescence increases. As the mitochondria take-up the calcium the fluorescence signal decreases. Calcium is continually loaded into the mitochondria until there is a sudden, large, increase in calcium fluorescence which indicates complete release of the stored calcium due to opening of the mPTP. Inhibition of the mPTP, with Cyclosporin A or our proprietary inhibitors can increase the capacity of mitochondria to retain calcium and thus protect the mitochondria from calcium overload in stress situations. Extramitochondrial calcium is measured by the fluorescence of calcium green. Note, increasing the concentration of CsA does not increase the CRC due to saturation of its target (Cyclophilin D) and is one of its limitations.
Control CsA (1μM)GNX-B (0.1mM)
Ca2+ Pulses (10 mM)
Ext
ra-m
itoc
hond
rial
Cal
cium
flu
ores
cenc
e
Ca2+ Release mPTP open
GNX-B (1.0mM) GNX-B (5.0mM)
40 mM 120 mM 250 mM180 mM 380 mM
Time (min)
CR
C (m
M C
a2+)
GNX C
0.1mM
0.5mM
1.0mM
5.0mM
0.1mM
0.5mM
1.0mM
5.0mM
0
50
100
150
200
250
300
350
CsA
Control
The identified PTP inhibitors are >> than CsA
Class 6 compounds are able to increase the CRC (by mPTP inhibition) several fold over that of unprotected mitochondria.
Class 6 compounds increase the CRC significantly over that maximally attainable with CsA.

PTP inhibitors for Acute Myocardial Infarction
The highway to proof-of-concept:•unmet medical need•strong evidence for involvment of the mPTP•linear path for the design of a clinical study

Area at Risk (g)
Arae
Nec
rotic
(g)
vehicle A-B
GNX -B
CsA
0.0
0.1
0.2
0.3
0.4
0.5
0.6
0.2 0.4 0.6 0.8 1.0 1.2 1.4
No protection
Protection
.
. ..
. ..
. .
..
.
.. .
. ..
. ..
PTP inhibitors are cardioprotectivein animal models
New Zealand White Rabbits were subjected to Left Anterior Descending (LAD) coronary artery occlusion for 30 mins followed by 4 hrs of reperfusion. Area at Risk and infarcted area (area necrotic) was determined by Evans blue and TTC staining. GNX-B (15 mg /kg in 3 mls 40% PEG 400; 20% DMSO) and CsA (10 mg/kg in sandimune) were administered by i.v. bolus 5 mins prior to reperfusion. Note, as with CsA in the clinical proof of concept trial, there is greater protection when the area at risk is larger. (In collaboration with Prof Ovize, Ospice di Lyon)

Un ruolo inaspettato per p53 nell’induzione di necrosi
Dopo stress, p53 si trova localizzata insieme con la ciclofilina-D ai mitocondri

P53 è richiesta per l’apertura del PTP durante la necrosi da stress ossidativo
Viene misurata l’apertura del PTP

Lo stress ossidativo induce necrosi e non apoptosi che dipende da p53

Considerazioni
• P53 è un regolatore fondamentale dell’apoptosi, ma sembra giocare un ruolo altrettanto rilevante nella morte cellulare da necrosi dopo stress ossidativo– P53 è “good” e “bad”: in questo caso potrebbe essere
coinvolto nella patogenesi di molte malattie• Il coinvolgimento di p53 è un’altra dimostrazione
che anche la necrosi è sottoposta (almeno in alcuni casi) a un livello di regolazione impensabile fino a poco tempo fa