Humidification Performance of 48 Passive Airway … de Evaluation 48...manufacturers (indication for...
Transcript of Humidification Performance of 48 Passive Airway … de Evaluation 48...manufacturers (indication for...

DOI 10.1378/chest.08-0679 2009;135;276-286Chest
Bruno Fumagalli and Laurent BrochardSalvatore Maurizio Maggiore, Philippe Jouvet, Jean-Damien Ricard, François Lellouche, Solenne Taillé, Frédéric Lefrançois, Nicolas Deye, Airway HumidifiersHumidification Performance of 48 Passive
http://www.chestjournal.org/content/135/2/276.full.html
and services can be found online on the World Wide Web at: The online version of this article, along with updated information
) ISSN:0012-3692http://www.chestjournal.org/misc/reprints.shtml(of the copyright holder.may be reproduced or distributed without the prior written permission Northbrook IL 60062. All rights reserved. No part of this article or PDFby the American College of Chest Physicians, 3300 Dundee Road,
2007Physicians. It has been published monthly since 1935. Copyright CHEST is the official journal of the American College of Chest
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

Humidification Performance of 48Passive Airway Humidifiers*Comparison With Manufacturer Data
Francois Lellouche, MD, PhD; Solenne Taille, Eng; Frederic Lefrancois, Eng;Nicolas Deye, MD, MSc; Salvatore Maurizio Maggiore, MD, PhD;Philippe Jouvet, MD, PhD; Jean-Damien Ricard, MD, PhD; Bruno Fumagalli, Eng;and Laurent Brochard, MD; and Groupe de travail sur les Respirateurs de l’AP-HP
Introduction: Heat and moisture exchangers (HMEs) are increasingly used in the ICU for gasconditioning during mechanical ventilation. Independent assessments of the humidificationperformance of HMEs are scarce. The aim of the present study was thus to assess thehumidification performance of a large number of adult HMEs.Method: We assessed 48 devices using a bench test apparatus that simulated real-life physiologicventilation conditions. Thirty-two devices were described by the manufacturers as HMEs, and 16were described as antibacterial filters. The test apparatus provided expiratory gases with anabsolute humidity (AH) of 35 mg H2O/L. The AH of inspired gases was measured after steadystate using the psychrometric method. We performed three hygrometric measurements for eachdevice, measured their resistance, and compared our results with the manufacturer data.Results: Of the 32 HMEs tested, only 37.5% performed well (> 30 mg H2O/L), while 25%performed poorly (< 25 mg H2O/L). The mean difference (� SD) between our measurementsand the manufacturer data was 3.0 � 2.7 mg H2O/L for devices described as HMEs (maximum,8.9 mg H2O/L) [p � 0.0001], while the mean difference for 36% of the HMEs was > 4 mg H2O/L.The mean difference for the antibacterial filters was 0.2 � 1.4 mg H2O/L. The mean resistance ofall the tested devices was 2.17 � 0.70 cm H2O/L/s.Conclusions: Several HMEs performed poorly and should not be used as HMEs. The valuesdetermined by independent assessments may be lower than the manufacturer data. Describing adevice as an HME does not guarantee that it provides adequate humidification. The performanceof HMEs must be verified by independent assessment. (CHEST 2009; 135:276–286)
Key words: absolute humidity; endotracheal tube; heat and moisture exchangers; mechanical ventilation; occlusions;psychrometry
Abbreviations: AH � absolute humidity; HME � heat and moisture exchanger; HMEF � heat and moisture ex-changer with an antibacterial filter; ISO � International Standard Organization
D uring invasive mechanical ventilation, gases de-livered to the patient must be heated and
humidified.1,2 Heat and moisture exchangers(HMEs) are the most commonly used humidificationdevices in Europe3 and are being increasingly usedin North America.3,4 While some hygrometric stud-ies of passive humidification systems have beenconducted, few have included a large number ofdevices, and clinicians largely rely on data from themanufacturer. Given this conflict of interest, inde-pendent performance assessments are needed.5
In the past, strictly hydrophobic HMEs often ledto endotracheal tube occlusions.6–10 Nowadays, mostHMEs have good hydrophobic and hygroscopicproperties, and many devices have both humidifica-tion and antibacterial-filtering properties. Some,however, should only be used for short-term venti-lation, such as during anesthesia, because they aremainly antibacterial filters with poor humidificationperformance. Despite this, some manufacturers de-scribe them as humidification devices that can beused for prolonged mechanical ventilation. Also, very
Original ResearchCRITICAL CARE MEDICINE
276 Original Research
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

similar aspects of devices with and without humidi-fication properties can be a source of confusion forclinicians.
There are many commercially available devices,but few large-scale, independent studies havebeen conducted to assess their humidificationperformance.11,12 The goal of the present studywas to assess a large number of such devices withand without humidification proprieties. An invita-tion to tender by the Assistance Publique-Hopi-taux de Paris was used to collect the devices. Wedeveloped a bench test apparatus that reproducedreal-life saturated expired gas conditions in orderto assess the hygrometric performance of thesedevices. We also measured the resistance of thedevices, classified into two categories: HMEs withan antibacterial filter (HMEF) or without anantibacterial filter (HME), and those described asantibacterial filters. The measurements obtainedwith the bench test apparatus were compared tomanufacturer data.
Materials and Methods
We received 47 devices for adults from the manufacturersfollowing the invitation to tender for the HMEs and antibacterialfilters. Five of each type of device were provided by themanufacturer: three were assessed for their humidification per-formances, one was used for resistance measurement, and oneadditional device was available if required. Also, the hydrophobicPall BB2215 filter (Pall Biomedical; Saint-Germaine-en-Laye,
France), which has a poor humidification performance and hasbeen associated with a high rate of endotracheal intubations, wasused as a reference device.6–10 The goal of the present study wasto assess the performance of the devices using a bench testapparatus and to select the most efficient ones for a subsequentclinical trial.
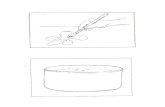
The bench test apparatus was a stable, reproducible model andused the same ventilation and physiologic conditions (wet expi-ratory gases, dry motor gases, and constant ambient temperature)for assessing the HMEs and antibacterial filters. The apparatus isshown in Figure 1. The relative humidity of real-life expired gasesis 100% while the temperature at the T-piece is 32 to 33°C,13–15
which corresponds to an absolute humidity (AH) of 34 to 36 mgH2O/L. We generated expiratory gases with an AH of 35 mgH2O/L using a heated humidifier (MR 730; Fisher&Paykel;Auckland, New Zealand, with a heated wire circuit) connected tothe expiratory limb of a ventilator to simulate real-life expiratoryparameters. The humidity of simulated expired gas was measuredwith the psychrometric method (see below) until the target valuewas reached and then regularly during the study days to ensurethe maintain of a stable value. A Servo 900C (Maquet; Lund,Sweden) ventilator was used in assist-control mode with arespiratory rate of 20 breaths/min, a tidal volume of 500 mL, apositive expiratory pressure at 5 cm H2O, and an fraction ofinspired oxygen of 21%. With this ventilator, motor gases usedare dry medical gases (� 5 mg H2O/L). The room temperaturewas kept constant between 24.5°C and 25.5°C.
Hygrometric Measurements
The parameters of the test apparatus were stabilized during 3 hfor each device on the bench, and then the hygrometry of theinspiratory gases was measured using the psychrometric method.We used a variant of the previously described method: weconsidered the maximal temperature value and not the meantemperature.16,17 This method was used in previous publishedstudies15,18 with hygrometric measurements. Although, this mayslightly overestimate the values of hygrometric measurements,comparison with published data shows very good agreement forsimilar devices tested.17,19 Three measurements for each devicewere recorded on 3 separate days in a randomized order.Forty-eight devices (HMEs and filters) for adult patients wereassessed (47 from the tender and the Pall BB2215). We alsocalculated the humidity efficiency of the devices using thefollowing formula: humidity efficiency � (inspired AH/expiredAH) � 100. This provided information on the intrinsic perfor-mances of the devices. Also we displayed another index ofefficiency: AH delivered/dead space (volume of the device) aspreviously described.11
Resistance Measurements
The resistance of 34 devices was also measured on aseparate bench. Resistance measurements were performed byplacing a pressure sensor (MP45 � 100 cm H2O; Validyne;Northridge, CA) before the filter and by progressively increas-ing the gas flow to 1 L/s. Flow was measured using a Fleisch#1 pneumotachograph (Fleisch; Zurich, Switzerland) con-nected to a differential pressure transducer (MP45 � 2 cmH2O; Validyne) placed before the pressure sensor and thetested device. The pressure after the filter was consideredequal to 0 (atmospheric pressure). The differential pressurethus corresponded to the pressure measured before the filter.The flow and pressure signals were recorded using software(Acknowledge; Biopac Systems; Santa Barbara, CA) with anacquisition frequency of 200 Hz. The analog signals wereconverted into digital signals (MP100WS; Biopac Systems).
*From the Service de Reanimation Medicale (Mrs. Taille,Mr. Lefrancois, and Dr. Brochard), Assistance Publique-Hopitaux de Paris, Centre Hospitalier Albert Chenevier-Henri-Mondor, Creteil, France; Service de Soins Intensifs de ChirurgieCardiaque (Dr. Lellouche), Centre de Recherche Hopital Laval,Universite Laval, Quebec City, QC, Canada; Service de Reani-mation Medicale (Dr. Deye), Hopital Lariboisiere, Paris, France;Istituto di Anestesiologia e Rianimazione-Universita CattolicaPoliclinico A. Gemelli (Dr. Maggiore), Rome, Italy; Service deReanimation Medicale Pediatrique (Dr. Jouvet), Hopital Necker-Enfants Malades, Paris, France; Service de Reanimation Medi-cale (Dr. Ricard), Hopital Louis Mourier, Hopitaux de Paris,Colombes, Paris, France; and Pharmacie Centrale des Hopitaux-Assistance Publique-Hopitaux de Paris (Mr. Fumagalli), Paris,France.Presented in part at the 14th Annual Congress of the EuropeanSociety of Intensive Care Medicine, Geneva, Switzerland, Sep-tember 30 to October 3, 2001.Dr. Brochard, as director of the research laboratory, received agrant from the Hudson Company of $10,000 in 2002, and a grantfrom Fisher & Paykel Company of $8,700 in 2001, with $10,000in 2002 to the same laboratory. The other authors have noconflict of interest to declare.Manuscript received March 11, 2008; revision accepted August13, 2008.Reproduction of this article is prohibited without written permissionfrom the American College of Chest Physicians (www.chestjournal.org/misc/reprints.shtml).Correspondence to: Francois Lellouche, Unite de soins intensifsde chirurgie cardiaque, Centre de Recherche Hopital Laval,Universite Laval, 2725, Chemin Sainte-Foy, QC, Canada, G1V4G5; e-mail: [email protected]: 10.1378/chest.08-0679
www.chestjournal.org CHEST / 135 / 2 / FEBRUARY, 2009 277
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

Dead space values (devices volume) as well as bacterial andviral filtration efficiency provided by manufacturers are also pre-sented. Lastly, we compared our hygrometric and resistance mea-surements with the manufacturer data when they were available.
Statistical Analysis
Results are expressed as mean � SD. Comparisons betweenour measured data and manufacturer data for AH and resistancewere obtained by Wilcoxon signed-rank test; p � 0.05 was con-sidered significant.
Results
Hygrometric and resistance values obtained on thebench are presented in Table 1. Data coming frommanufacturers (indication for use, hygrometry, resis-tance, dead space, antimicrobial filtration efficiency)are presented in Table 2.
Hygrometry
We assessed 48 devices (47 from tender and theBB 2215) using the bench test apparatus and per-formed three measurements for each device. Thirty-two devices were described as HME or HMEF bythe manufacturers, but some were proposed forhumidification during anesthesia only. The average
AH of the devices was 27.5 � 4.4 mg H2O/L. Twelvedevices performed well (� 30 mg H2O/L), 12 wereintermediate (� 30 to � 25 mg H2O/L), and 8performed poorly (� 25 mg H2O/L). Sixteen deviceswere described as antibacterial filters only by themanufacturers. The average AH of these devices was17.3 � 3.6 mg H2O/L. The hygrometric measure-ments are presented in Table 1 and Figure 2.Humidity efficiency of the devices ranged from 91.1to 37.8% (Fig 3).
We could compare our results with manufac-turer data for 29 devices, 25 of the HMEs and 4 ofthe antibacterial filters. There were no hygromet-ric measurements provided by the manufacturerfor four HMEs, while for two others the hygro-metric values were obtained with tidal volumes of250 mL and 1,500 mL and were thus not used.Among the devices presented as HME or HMEF,55% were evaluated by the manufacturers accord-ing to the International Standards Organization(ISO) 9360 standard (gravimetric method). Themethod was not specified for the other devices.
Of the 29 devices that could be compared,manufacturer data were higher than the valuesdetermined using the test apparatus for 23 devices(79%) [Fig 2]. The mean difference between ourvalues and those of the manufacturers was
One-way valves
Tdry 32.1°C
Hygrometer(PSYCHROMETRIC METHOD)
Bacterial filter
Simulated lung
Device being tested
Heated humidifier (MR 730) simulating expiratory gases
Ventilator
Twet 30.9°C
Figure 1. Hygrometric bench test apparatus used to measure the humidification performances ofHMEs and antibacterial filters. A ventilator delivered controlled cycles (respiratory rate of 20breaths/min with a tidal volume of 500 mL and a positive end-expiratory pressure of 5 cm H2O).A heated humidifier (MR 730) was connected to the expiratory limb of the model and was set todeliver gases with a water content of 35 mg H2O/L at the Y-piece. A circuit with heated wire wasused after the humidification chamber. The room temperature was kept constant between 24.5°Cand 25.5°C.
278 Original Research
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

Table 1—Measured AH Obtained Using the Bench Test Apparatus for All Devices and Measured Resistances for 34Devices*
DeviceNo. Device Make†
Manufacturer(Location)
MeasuredAH, mgH2O/L
� AHManufacturer-Measured
Values, mg H2O/LAH/mL of
Dead Space
MeasuredResistance
at 60 L/min,cm H2O
� ResistancesManufacturer-Measured
Values, cm H2O/L/s
1 Hygrovent Peters 31.9 � 0.6 2.7 0.34 1.8 �0.72 Hygrobac Mallinckrodt 31.7 � 0.7 1.0 0.33 2.1 0.03 Hygrovent S Peters 31.7 � 0.5 2.8 0.58 2.8 �1.24 Hygrobac S Mallinckrodt 31.2 � 0.2 �0.5 0.69 2.3 0.25 HMEF 1000 Datex 31.2 � 0.7 1.8 0.41 NA NA6 9000/100 Allégiance 31.2 � 1.4 2.6 0.35 2.7 �0.77 Servo Humidifier 172
(173)Siemens 30.9 � 0.3 NA 0.56 NA NA
8 Humid Vent FilterCompact
Hudson 30.8 � 0.3 0.2 0.88 2.3 �0.5
9 All round Filter MAP05 Peters 30.7 � 0.2 �0.7 0.34 NA NA10 Hygroster Mallinckrodt 30.7 � 0.6 2.1 0.32 2.3 �0.411 Slimeline HMEF
9040/01Sodis 30.3 � 0.3 2.7 0.67 NA NA
12 BACT HME Ansell 30.1 � 0.5 4.2 0.43 2.4 �1.013 Filtraflux Vygon 29.9 � 0.9 1.1 0.54 NA NA14 HME 12 BASIC Ansell 29.8 � 0.4 3.2 0.37 0.4 0.115 Humid Vent 2 Hudson 29.7 � 0.4 �1.7 1.03 NA NA16 Servo Humidifier 162
(163)Siemens 29.7 � 0.8 NA 0.78 NA NA
17 FE52A Rüsch 29.3 � 0.4 NA 0.35 2.4 NA18 Humid Vent 2S Hudson 29.2 � 0.4 �1.2 1.01 NA NA19 9040/01 Allégiance 28.6 � 1.1 NA 0.61 2.4 NA20 9000/01 Allégiance 28.5 � 0.8 2.9 0.32 3.9 �1.921 Thermovent 1200 Sims 27.8 � 0.0 NA 0.86 NA NA22 BB100E Pall 27.2 � 0.7 3.8 0.32 1.4 0.623 BB100 Pall 26.8 � 0.5 4.2 0.30 2.0 0.524 Clear Therm � Intersurgical 26.2 � 2.1 5.8 0.50 2.1 0.325 Filta Therm Intersurgical 24.9 � 1.1 5.1 0.51 2.9 �0.426 Stérivent Mallinckrodt 23.8 � 0.9 �0.7 0.26 1.9 �0.127 Iso Gard Hepa Light Hudson 23.6 � 0.3 2.2 0.47 2.4 �0.428 Filtraflux Vygon 22.4 � 0.4 NA 0.41 NA NA29 Stérivent S Mallinckrodt 22.2 � 0.2 �1.0 0.36 1.7 0.230 Pall BB 2,215‡ Pall 21.8 � 1.5 NA NA NA NA31 8222/01 Allégiance 20.5 � 0.2 NA 0.28 NA NA32 Maxipleat Sodis 20.1 � 0.6 4.8 0.27 NA NA33 HME 10 Ansell 20.0 � 0.6 8.9 0.74 NA NA34 BB25 Pall 19.6 � 1.4 6.4 0.56 2.6 0.935 All Round Filter
MAP01Peters 19.0 � 0.3 NA 0.27 1.7 �0.1
36 BB2000AP Pall 18.9 � 0.4 7.1 0.54 3.1 0.437 BACT TRAP HEPA Ansell 18.4 � 0.7 NA NA 1.3 NA38 Clear Therm Midi Intersurgical 17.3 � 0.3 5.7 0.72 2.1 0.439 Barr Vent Peters 16.8 � 1.1 NA 0.17 1.4 0.140 Stérivent Mini Mallinckrodt 16.6 � 1.0 0.4 0.47 2.2 0.841 4444/66 Allégiance 16.4 � 0.6 NA 0.35 3.4 NA42 Filta Guard Intersurgical 16.2 � 1.0 NA 0.29 2.3 0.043 4000/01 Allégiance 15.1 � 0.9 NA 0.40 2.2 NA44 Clear Guard II Intersurgical 14.9 � 0.6 NA 0.35 2.1 0.245 Clear Guard Midi Intersurgical 13.7 � 0.8 NA 0.57 1.6 0.546 ISO GUARD
FILTER SHudson 13.4 � 0.6 NA 0.52 2.1 �0.5
47 Barr Vent S Peters 13.3 � 0.8 NA 0.36 1.4 0.348 Barrierbac S Mallinckrodt 13.2 � 0.2 NA 0.38 2.1 �0.3
*The difference between our hygrometric data and those of the manufacturers was 3.0 � 2.7 mg H2O/L for the HMEs (p � 0.0001) and 0.2 � 1.4mgH2O/L for the antibacterial filters (p � 0.99). Mean difference between our measured resistances and those of the manufacturers was– 0.1 � 1.6 cm H2O/L (p � 0.63). NA � not available.
†The numbering is based on the measured hygrometric results of the tested devices.‡Filter BB2215 (not part of the tender) was also assessed.
www.chestjournal.org CHEST / 135 / 2 / FEBRUARY, 2009 279
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

Tab
le2—
Man
ufa
ctu
rer
Dat
aC
once
rnin
gth
eIn
dica
tion
and
Mai
nC
hara
cter
isti
csfo
rA
llth
eT
este
dD
evic
es*
Dev
ice
Indi
catio
nfo
rU
se(M
anuf
actu
rer
Dat
a)M
ain
Cha
ract
eris
tics
(Man
ufac
ture
rD
ata)
No.
Mak
eM
anuf
actu
rer
Ref
eren
ceA
naes
thes
iaIC
UA
dult
Pedi
atri
cF
ilter
(Ven
tilat
or)
Filt
er(P
atie
nt)
HM
EH
ME
F
Bac
teri
alan
dV
iral
Filt
ratio
nE
ffic
ienc
y,%
Vol
ume,
mL
Res
ista
nce
toF
low
at60
L/m
in,c
mH
2O
/L/s
AH
,mg
H2O
/L
1H
ygro
vent
Pete
rsM
F6
Yes
Yes
Yes
No
No
Yes
Yes
Yes
99.9
9995
1.1
34.6
†2
Hyg
roba
cM
allin
ckro
dtD
AR
352/
5805
Yes
Yes
Yes
Yes
�99
.99
952.
132
.73
Hyg
rove
ntS
Pete
rsM
F11
Yes
Yes
Yes
No
No
Yes
Yes
Yes
99.9
9955
1.6
34.5
†4
Hyg
roba
cS
Mal
linck
rodt
DA
R35
2/58
77Ye
sYe
sYe
sYe
s�
99.9
945
2.5
30.7
5H
ME
F10
00D
atex
HM
EF
1000
Yes
Yes
Yes
Yes
99.9
9999
9–99
.99
772.
333
†6
9000
/100
Allé
gian
ce90
00/1
01Ye
sYe
sYe
sYe
sYe
sYe
s99
.999
901.
9533
.8†
7Se
rvo
Hum
idifi
er17
2(1
73)
Siem
ens
6419
373
Yes
Yes
Yes
Yes
99.9
97–9
9.94
55N
A33
.1–2
7.6†
‡
8H
umid
Ven
tF
ilter
Com
pact
Hud
son
1840
1Ye
sYe
sYe
sYe
sYe
sYe
sN
A35
1.8
31
9A
llR
ound
Filt
erM
AP0
5Pe
ters
MA
05Ye
sN
oYe
sN
oYe
sYe
sYe
sYe
s99
.989
.52.
130
†
10H
ygro
ster
Mal
linck
rodt
DA
R35
4/58
76Ye
sYe
sYe
sYe
s�
99.9
998
951.
932
.811
Slim
elin
eH
ME
F90
40/0
1So
dis
9040
/01
Yes
Yes
Yes
Yes
Yes
99.9
9945
NA
33
12B
AC
TH
ME
Ans
ell
602
0F
Yes
Yes
Yes
�99
.99
701.
434
.313
Filt
raflu
xV
ygon
551,
01Ye
sYe
sYe
sYe
s(�
25kg
)Ye
sYe
sYe
sYe
s99
.997
–99.
9455
2.1
31†
14H
ME
12B
ASI
CA
nsel
l6
072
FYe
sYe
sYe
sYe
sN
A81
0.5
�33
15H
umid
Ven
t2
Hud
son
1331
1F
Yes
Yes
Yes
NA
29N
A28
16Se
rvo
Hum
idifi
er16
2(1
63)
Siem
ens
6419
357
Yes
Yes
Yes
Yes
NA
38N
A33
–25.
6†‡
17F
E52
AR
üsch
FE
52A
Yes
Yes
Yes
Yes
Yes
�99
.99
84N
AN
A18
Hum
idV
ent
2SH
udso
n14
411
FYe
sYe
sYe
sN
A29
NA
2819
9040
/01
Allé
gian
ce90
40/0
1Ye
sYe
sYe
sYe
sYe
sYe
s99
.999
47N
AN
A20
9000
/01
Allé
gian
ce90
00/0
1Ye
sYe
sYe
sYe
sYe
sYe
s99
.999
901.
9531
.4†
21T
herm
oven
t12
00Si
ms
100
582
000
Yes
Yes
Yes
Yes
NA
32.4
2023
22B
B10
0EPa
llB
B10
0EYe
sYe
sYe
s(�
23kg
)Ye
sYe
sYe
s�
99.9
999
852
31†
23B
B10
0Pa
llB
B10
0Ye
sYe
sYe
s(�
23kg
)Ye
sYe
sYe
s�
99.9
999
902.
531
†24
Cle
arT
herm
�In
ters
urgi
cal
1841
001
Yes
Yes
Yes
Yes
99.9
9952
2.4
32†
25F
ilta
The
rmIn
ters
urgi
cal
1941
Yes
Yes
Yes
Yes
99.9
9999
492.
530
†26
Stér
iven
tM
allin
ckro
dtD
AR
351/
5856
Yes
Yes
Yes
�99
.999
992
1.8
23.1
27Is
oG
ard
Hep
aL
ight
Hud
son
2801
2Ye
sYe
sYe
sYe
sN
A50
1.1
25.8
28F
iltra
flux
Vyg
on55
3,01
Yes
Yes
Yes
Yes
Yes
No
No
99.9
97–9
9.94
551.
7N
A29
Stér
iven
tS
Mal
linck
rodt
DA
R35
1/58
78Ye
sYe
sYe
s99
.999
962
1.9
21.2
30B
B22
15Pa
ll31
8222
/01
Allé
gian
ce82
22/0
1Ye
sYe
sYe
sYe
sYe
s99
.999
974
NA
NA
32M
axip
leat
Sodi
s82
22/0
1Ye
sYe
sYe
sYe
sYe
sYe
sYe
s99
.999
974
2–6
24.9
280 Original Research
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

Tab
le2—
Con
tinu
ed
Dev
ice
Indi
catio
nfo
rU
se(M
anuf
actu
rer
Dat
a)M
ain
Cha
ract
eris
tics
(Man
ufac
ture
rD
ata)
No.
Mak
eM
anuf
actu
rer
Ref
eren
ceA
naes
thes
iaIC
UA
dult
Pedi
atri
cF
ilter
(Ven
tilat
or)
Filt
er(P
atie
nt)
HM
EH
ME
F
Bac
teri
alan
dV
iral
Filt
ratio
nE
ffic
ienc
y,%
Vol
ume,
mL
Res
ista
nce
toF
low
at60
L/m
in,c
mH
2O
/L/s
AH
,mg
H2O
/L
33H
ME
10A
nsel
l60
60PF
Yes
Yes
Yes
Yes
NA
270.
928
.934
BB
25Pa
llB
B25
Yes
Yes
Yes
(�10
kg)
Yes
Yes
Yes
�99
.999
935
3.5
26†
35A
llR
ound
Filt
erM
AP0
1Pe
ters
MA
01Ye
sN
oYe
sN
oYe
sYe
sN
oN
o99
.9–9
9.99
99–9
9.99
9970
1.6
NA
36B
B20
00A
PPa
llB
B20
00A
PYe
sYe
sYe
s(�
10kg
)Ye
sYe
sYe
s�
99.9
999
353.
526
†37
BA
CT
TR
AP
HE
PAA
nsel
l70
80PF
Yes
Yes
Yes
99.9
99N
AN
AN
A
38C
lear
The
rmM
idi
Inte
rsur
gica
l16
41Ye
sYe
sYe
sYe
s99
.999
242.
523
†
39B
arr
Ven
tPe
ters
MF
1Ye
sN
oYe
sN
oN
oYe
sN
oN
o99
.999
101
1.1
NA
40St
ériv
ent
Min
iM
allin
ckro
dtD
AR
351/
5979
Yes
Yes
Yes
Yes
�99
.999
9935
317
4144
44/6
6A
llégi
ance
4444
/66
Yes
Yes
Yes
Yes
Yes
99.9
9947
NA
NA
42F
ilta
Gua
rdIn
ters
urgi
cal
1944
Yes
Yes
Yes
Yes
99.9
9999
562.
3N
A43
4000
/01
Allé
gian
ce40
00/0
1Ye
sYe
sYe
sYe
sYe
s99
.999
38N
AN
A44
Cle
arG
uard
IIIn
ters
urgi
cal
1844
Yes
Yes
Yes
Yes
99.9
999
422.
3N
A45
Cle
arG
uard
Mid
iIn
ters
urgi
cal
1644
Yes
Yes
Yes
Yes
99.9
9924
2.1
NA
46IS
OG
UA
RD
FIL
TE
RS
Hud
son
1921
1F
Yes
Yes
Yes
Yes
NA
261.
6N
A
47B
arr
Ven
tS
Pete
rsM
FP3
Yes
No
Yes
No
No
Yes
No
No
99.9
9937
1.5
NA
48B
arri
erba
cS
Mal
linck
rodt
DA
R35
0/58
79Ye
sYe
sYe
s�
99.9
935
1.8
NA
*See
Tab
le1
for
expa
nsio
nof
abbr
evia
tion.
†HM
Eor
HM
EF
eval
uate
dw
ithth
eIS
O93
60st
anda
rd.
‡For
tidal
volu
mes
of25
0to
1,50
0m
L;n
umbe
ring
isba
sed
onth
em
easu
red
hygr
omet
ric
resu
ltsof
the
test
edde
vice
s(T
able
1).
www.chestjournal.org CHEST / 135 / 2 / FEBRUARY, 2009 281
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

3.0 � 2.7 mg H2O/L for the HMEs (p � 0.0001).The maximum difference was 8.9 mg H2O/L,while for 9 of the 25 HMEs the difference was � 4mg H2O/L. The mean difference for the antibac-terial filters was 0.2 � 1.4 mg H2O/L (p � 0.99).
Resistance
We performed 34 resistance measurements of the48 tested devices. Once again, we were able tocompare the values for 29 devices. The averageresistance of these devices was 2.17 � 0.70 cm H2O/L/s at 1 L/s, with a maximum of 3.9 cm H2O/L/s anda minimum of 0.4 cm H2O/L/s. The mean resis-tances at 1 L/s were 2.32 � 0.70 cm H2O/L/s and1.98 � 0.53 cm H2O/L/s for the HMEs and antibac-terial filters, respectively. The mean difference be-tween our values and those of the manufacturers was– 0.1 � 1.6 cm H2O/L (p � 0.63). Values are pre-sented in Table 1.
Dead Space
The dead space of the HMEs ranged from 22 to 95mL, with a mean of 60 � 26 mL. The dead space ofthe antibacterial filters ranged from 24 to 101 mL,with a mean of 55 � 24 mL.
Discussion
In the present study, we assessed the humidifica-tion performances and resistance of 48 HMEs and
antibacterial filters, and compared our results tomanufacturer data. The most important result of thestudy was the heterogeneity of the humidificationperformance of the devices. This highlights that thechoice of a suitable HME used for prolonged me-chanical ventilation is very difficult and may have areal clinical impact. In addition, the humidificationperformance of a significant number of the HMEstested was much lower than recommended, whichwould put patients at high risk for endotracheal tubeocclusions.6–10 These HMEs should be avoided forpatients receiving mechanical ventilation. We ob-served a marked difference between the AH valuesof the HMEs obtained using the bench test appara-tus and manufacturer data, with a mean difference of3.0 � 2.7 mg H2O/L. The difference was � 4 mgH2O/L for over one third of the HMEs, while thelargest was 8.9 mg H2O/L. The difference was only0.2 � 1.4 mg H2O/L for the antibacterial filters. Weonly observed small differences for the resistancemeasurements. Because of potentially fatal conse-quences resulting from inadequate humidification,independent assessments of the performance of hu-midification devices should be conducted to enableclinicians to select the most suitable devices.6,7,20
The present study is the largest evaluation ofHMEs and antibacterial filters ever conducted.Branson and Davis11 compared the performances of21 devices according to the ISO 9360 standard usingthe gravimetric method, Cigada et al12 assessed 12devices, and other studies12,16,21–34 assessed smallnumbers of humidification devices using various
15
20
25
30
35
Manufacturers’ data
Measured valuesBB2215 (measured)
1 2 3 4 5 6 8 9 10 11 26 27 29 30 3220 22 23 24 2512 13 14 15 18 33 34 36 38 40* * * *
Abs
olut
eH
umid
ity(m
gH2O
/L)
Figure 2. Inspired AH values obtained using the bench test apparatus compared to manufacturer data when available (29 devices).Results of the hydrophobic HME Pall BB2215 are also displayed. The asterisks indicate the devices that are proposed as antimicrobialfilters. The numbering corresponds to Tables 1 and 2.
282 Original Research
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

hygrometric measurement methods. To our knowl-edge, only two other bench studies16,34 used thepsychrometric method.
In the present study, 12 of the 48 HMEs testeddelivered inspiratory gases with an AH exceeding 30mg H2O/L, compared to 18 according to the manu-facturer (Table 1). This level of AH is recommendedfor humidification devices used during mechanicalventilation.35,36 Twelve other devices delivered gaseswith an AH ranging from 25 to 30 mg H2O/L, whichis usually considered a “gray zone.” The remainingdevices delivered gases with an AH � 25 mg H2O/Land should, in our opinion, be avoided during me-chanical ventilation, apart from limited use for theirfiltration properties. We also assessed the PallBB2215 hydrophobic HME, which has been associ-ated with a high rate of endotracheal tube occlu-sions.6–10 In the present study, this device deliveredgases with an AH of 21.8 � 1.5 mg H2O/L (Table 1).Our results were consistent with clinical resultsobtained using the psychrometric method.19,37 Byextrapolation, our data suggest that using an HMEwith a hygrometric performance close to 20 mgH2O/L carries a high risk of endotracheal tubeocclusion (Fig 4). Clinical results indicate that de-vices that deliver gases with an AH � 30 mg H2O/Lhave a low risk of endotracheal tube occlusions,38–43
even during prolonged use.17,44 However, slightlylower levels may also be safe44–48 (Fig 4).
The humidity efficiency of the most efficientHME exceeded 90% within the experimental condi-tions of our bench test (Fig 3). The efficiency valuesmust be interpreted cautiously because changing thedenominator value (ie, AH of the expiratory gas)could improve or worsen the calculated efficiency ofa given HME. We found it useful, however, topresent these data (Fig 2) because it allows compar-ing any filter with another, and also allows compar-isons with published studies.11,15 In addition, thevalue of AH used in this study represents a veryrealistic value of the normal water content of expiredgases.14,15 This means that all devices tested receivedadequate humidification to meet or exceed theirindividual AH capacity.
Resistance measurements was not the primaryobjective of the study, and therefore we performedresistance measurements only on 34 of the 48 de-vices tested for humidification properties. However,this evaluation was large enough to demonstrate thatthere was no significant difference when comparedwith manufacturer measurements, in opposition withhygrometric comparisons. This may underline thespecific issue of the hygrometric measurements.
Given that the performance of some devices maydecrease with prolonged use, one limitation of thepresent study was that the performances of thedevices were not assessed after prolonged use.17,49
The impact of prolonged use on HME performance
Figure 3. Humidity efficiency of the 48 devices tested calculated using the following formula: humidity efficiency (%) � (inspiredAH/expired AH) � 100. Minimum and maximum efficiencies are indicated.
www.chestjournal.org CHEST / 135 / 2 / FEBRUARY, 2009 283
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

and clinical consequences remains poorly docu-mented and should be assessed, especially if passivehumidifiers are used as first-line humidification de-vices. In addition, given that the performances ofsome humidification devices are reduced by highambient air and ventilator output temperatures, asecond limitation was that we did not assess theimpact of external factors such as temperature.18
However, it is likely that ambient air and ventilatortemperature have little influence on HME perfor-mance,50 and the room temperature was kept con-stant during the study days between 24.5°C and25.5°C to reduce the influence of this parameter.
The bench test apparatus may also be considered asa limitation; however, it allowed us to compare a largenumber of devices under the same conditions. It hasbeen shown that the efficacy of HMEs depends on theAH of expired gases.15 Studies11,12,22–27,30,31 usingmodels to simulate saturated expired gases used gastemperatures ranging from 32 to 37°C, making themdifficult to compare. In the present study, however,we assessed a large number of devices using a gaswith 35 mg H2O/L (saturated gas at 32.6°C), likeprevious studies13–15 that assessed expiratory gases inpatients receiving ventilation. In clinical studies, thetemperature of expiratory gas depends on the coretemperature of patients and is difficult to control.
Minute ventilation can also have an impact onhydrophobic HMEs.37,49,51 In the present study,minute ventilation was maintained at 10 L/min for allthe devices. It is likely that humidification perfor-mances may be influenced by the tidal volume used,mainly for hydrophobic HMEs.
The psychrometric method used in the presentstudy may also be considered as a limitation sincesome researchers may consider the ISO 9360 as the“reference method.”52 The ISO 9360 using the gravi-metric method is an attempt to homogenize thepractices for HME evaluation and is used by amajority of manufacturers. However, the gravimetricmethod is difficult to set up, and weight-in as well asspirometric measurement errors make this tech-nique difficult to use.11,22 Another limitation of thistechnique is that it was not described for evaluationof heated humidifiers. In addition, the gravimetricmethod cannot be used with patients, and the resultsthus cannot be directly compared with clinical data,unlike the psychrometric method.15,17,49 To date,there are no data that allow to state that one methodis more accurate than another, and there is no “goldstandard” for hygrometric measurements. The ISO9360 standard was used by 55% of the manufacturersfor HME or HMEF evaluation, and this techniquedoes not seem to overestimate humidity measure-
0
5
10
15
20
25
30
35
40
20 25 30 35
Absolute Humidity (mgH2O/L)
% E
ndot
rach
eal t
ube
occl
usio
ns
Figure 4. Frequency of endotracheal tube occlusions reported in the literature6–10,17,38–43,45–48,53–55
compared to the AH delivered by the HMEs measured using the bench test apparatus (Table 1). Onestudy (circle) is an outlier56 with no endotracheal occlusions despite very poor humidification. However,this study56 only included 12 patients, which is not enough to reach a conclusion about the safety of thedevices tested. A hydrophobic HME (Pall BB2215), which delivered a measured AH of 21.8 � 1.5 mgH2O/L in the present study, has been associated with high rates of endotracheal tube occlusions in fivepreviously published studies6–10 (Table 1). A number of studies are represented by gray dots to makethem stand out. The sizes of the dots are proportional to the number of patients in the studies.
284 Original Research
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

ments in comparison with the psychrometricmethod. Indeed, when comparing the performancesof the same HMEs evaluated by the hygrometric orthe gravimetric methods, the differences are small.In the study by Branson and Davis,11 Hygrobac,Hygrobac S, Hygroster, and Sims Portex 1200 pro-vided 32.5, 29.6, 33.2, and 27.2 mg H2O/L, respec-tively, when tested with the gravimetric method. Inthe present study, with the psychrometric method,the same devices provided very close results: 31.7,31.2, 30.7, and 27.8 mg H2O/L (Table 1). Thus, thedifferences we found in the present study betweenour measurements and manufacturer data are likelynot related to the measurement technique.
In conclusion, we conducted a large-scale assess-ment of passive airway humidifiers and showed thatthe humidification performances of these devicesvaried widely. Independent assessments of HMEperformances are required to guide clinicians intheir choice of a suitable device. The distinctionbetween devices with adequate humidification per-formances and those with predominantly antibacte-rial filtering properties should be made muchclearer, and the external appearance of devices withand without humidification proprieties might differ.Lastly, the use of poorly performing HMEs duringinvasive mechanical ventilation can lead to poten-tially fatal complications.
References1 Burton JDK. Effect of dry anaesthetic gases on the respira-
tory mucus membrane. Lancet 1962; 1:235–2382 Ricard JD. Humidification. In: Tobin M, ed. Mechanical
ventilation. New York, NY: McGraw-Hill, 2006; 1109–11203 Ricard JD, Cook D, Griffith L, et al. Physicians’ attitude to
use heat and moisture exchangers or heated humidifiers: aFranco-Canadian survey. Intensive Care Med 2002; 28:719–725
4 Burns KEA, Lellouche F, Loisel F, et al. Weaning critically illadults from invasive mechanical ventilation: a national survey[abstract]. Am J Respir Crit Care Med 2007; 175:A963
5 Thiery G, Boyer A, Pigne E, et al. Heat and moistureexchangers in mechanically ventilated intensive care unitpatients: a plea for an independent assessment of theirperformance. Crit Care Med 2003; 31:699–704
6 Cohen IL, Weinberg PF, Fein IA, et al. Endotracheal tubeocclusion associated with the use of heat and moistureexchangers in the intensive care unit. Crit Care Med 1988;16:277–279
7 Martin C, Perrin G, Gevaudan MJ, et al. Heat and moistureexchangers and vaporizing humidifiers in the intensive careunit. Chest 1990; 97:144–149
8 Misset B, Escudier B, Rivara D, et al. Heat and moistureexchanger vs heated humidifier during long-term mechanicalventilation: a prospective randomized study. Chest 1991;100:160–163
9 Roustan JP, Kienlen J, Aubas P, et al. Comparison ofhydrophobic heat and moisture exchanger with heated hu-midifier during prolonged mechanical ventilation. IntensiveCare Med 1992; 18:97–100
10 Villafane MC, Cinnella G, Lofaso F, et al. Gradual reductionof endotracheal tube diameter during mechanical ventilationvia different humidification devices. Anesthesiology 1996;85:1341–1349
11 Branson R, Davis J. Evaluation of 21 passive humidifiersaccording to the ISO 9360 standard: moisture output, deadspace, and flow resistance. Respir Care 1996; 41:736–743
12 Cigada M, Elena A, Solca M, et al. The efficiency of twelveheat and moisture exchangers: an in vitro evaluation. Int CareWorld 1990; 7:98–101
13 Dery R. Humidity in anesthesiology: III. Heat and moisturepatterns in the respiratory tract during anesthesia with thesemi-closed system. Can Anaesth Soc J 1967; 14:287–298
14 Dery R. The evolution of heat and moisture in the respiratorytract during anesthesia with a nonrebreathing system. CanAnaesth Soc J 1973; 20:296–309
15 Lellouche F, Qader S, Taille S, et al. Under-humidificationand over-humidification during moderate induced hypother-mia with usual devices. Intensive Care Med 2006; 32:1014–1021
16 Jackson C, Webb AR. An evaluation of the heat and moistureexchange performance of four ventilator circuit filters. Inten-sive Care Med 1992; 18:264–268
17 Ricard JD, Le Miere E, Markowicz P, et al. Efficiency andsafety of mechanical ventilation with a heat and moistureexchanger changed only once a week. Am J Respir Crit CareMed 2000; 161:104–109
18 Lellouche L, Taille S, Maggiore M, et al. Influence ofambient air and ventilator output temperature on perfor-mances of heated-wire humidifiers. Am J Respir Crit CareMed 2004; 170:1073–1079
19 Ricard JD, Markowicz P, Djedaini K, et al. Bedside evalua-tion of efficient airway humidification during mechanicalventilation of the critically ill. Chest 1999; 115:1646–1652
20 Kapadia FN. Factors associated with blocked tracheal tubes.Intensive Care Med 2001; 27:1679–1681
21 Chalon J, Markham JP, Ali MM, et al. The Pall Ultiporbreathing circuit filter: an efficient heat and moisture ex-changer. Anesth Analg 1984; 63:566–570
22 Eckerbom B, Lindholm CE. Performance evaluation of sixheat and moisture exchangers according to the Draft Inter-national Standard (ISO/DIS 9360). Acta Anaesthesiol Scand1990; 34:404–409
23 Gedeon A, Mebius C. The hygroscopic condenser humidifier:a new device for general use in anaesthesia and intensive care.Anaesthesia 1979; 34:1043–1047
24 Mebius C. A comparative evaluation of disposable humidifi-ers. Acta Anaesthesiol Scand 1983; 27:403–409
25 Mebius C. Heat and moisture exchangers with bacterialfilters: a laboratory evaluation. Acta Anaesthesiol Scand 1992;36:572–576
26 Ogino M, Kopotic R, Mannino FL. Moisture-conservingefficiency of condenser humidifiers. Anaesthesia 1985; 40:990–995
27 Shelly M, Bethune BW, Latimer RD. A comparison of fiveheat and moisture exchangers. Anaesthesia 1986; 41:527–532
28 Turtle MJ, Ilsley AH, Rutten AJ, et al. An evaluation of sixdisposable heat and moisture exchangers. Anaesth IntensiveCare 1987; 15:317–322
29 Unal N, Kanhai JK, Buijk SL, et al. A novel method ofevaluation of three heat-moisture exchangers in six differentventilator settings. Intensive Care Med 1998; 24:138–146
30 Unal N, Pompe JC, Holland WP, et al. An experimentalset-up to test heat-moisture exchangers. Intensive Care Med1995; 21:142–148
31 Walker AK, Bethune DW. A comparative study of condenserhumidifiers. Anaesthesia 1976; 31:1086–1093
www.chestjournal.org CHEST / 135 / 2 / FEBRUARY, 2009 285
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

32 Branson R, Hurst JM. Laboratory evaluation of of moistureoutput of seven airway heat and moisture exchangers. RespirCare 1987; 32:741–747
33 Weeks DB, Ramsay FM. A laboratory investigation of sixartificial nose for use during endotracheal anesthesia. AnesthAnalg 1981; 62:758–763
34 Vanderbroucke-Grauls CM, Teeuw KB, Ballemans K, et al.Bacterial and viral removal efficiency, heat and moistureexchange properties of four filtration devices. J Hosp Infect1995; 29:45–56
35 International Organization of Standards. Humidifiers formedical use. ISO 1988; 60:14
36 AARC clinical practice guideline: humidification during me-chanical ventilation; American Association for RespiratoryCare. Respir Care 1992; 37:887–890
37 Martin C, Papazian L, Perrin G, et al. Performance evaluationof three vaporizing humidifiers and two heat and moistureexchangers in patients with minute ventilation � 10 L/min.Chest 1992; 102:1347–1350
38 Boots RJ, George N, Faoagali JL, et al. Double-heater-wirecircuits and heat-and-moisture exchangers and the risk ofventilator-associated pneumonia. Crit Care Med 2006; 34:687–693
39 Boots RJ, Howe S, George N, et al. Clinical utility ofhygroscopic heat and moisture exchangers in intensive carepatients. Crit Care Med 1997; 25:1707–1712
40 Dreyfuss D, Djedaini K, Gros I, et al. Mechanical ventilationwith heated humidifiers or heat and moisture exchangers:effects on patient colonization and incidence of nosocomialpneumonia. Am J Respir Crit Care Med 1995; 151:986–992
41 Hurni JM, Feihl F, Lazor R, et al. Safety of combined heatand moisture exchanger filters in long-term mechanical ven-tilation. Chest 1997; 111:686–691
42 Lacherade JC, Auburtin M, Cerf C, et al. Impact of humid-ification systems on ventilator-associated pneumonia: a ran-domized multicenter trial. Am J Respir Crit Care Med 2005;172:1276–1282
43 Thomachot L, Vialet R, Viguier JM, et al. Efficacy of heat andmoisture exchangers after changing every 48 hours ratherthan 24 hours. Crit Care Med 1998; 26:477–481
44 Markowicz P, Ricard JD, Dreyfuss D, et al. Safety, efficacy,and cost-effectiveness of mechanical ventilation with humid-ifying filters changed every 48 hours: a prospective, random-ized study. Crit Care Med 2000; 28:665–671
45 Kirton OC, DeHaven B, Morgan J, et al. A prospective,randomized comparison of an in-line heat moisture exchange
filter and heated wire humidifiers: rates of ventilator-associatedearly-onset (community-acquired) or late-onset (hospital-acquired)pneumonia and incidence of endotracheal tube occlusion. Chest1997; 112:1055–1059
46 Thomachot L, Leone M, Razzouk K, et al. Randomizedclinical trial of extended use of a hydrophobic condenserhumidifier: 1 vs. 7 days. Crit Care Med 2002; 30:232–237
47 Thomachot L, Vialet R, Arnaud S, et al. Do the componentsof heat and moisture exchanger filters affect their humidifyingefficacy and the incidence of nosocomial pneumonia? CritCare Med 1999; 27:923–928
48 Thomachot L, Viviand X, Arnaud S, et al. Comparing twoheat and moisture exchangers, one hydrophobic and onehygroscopic, on humidifying efficacy and the rate of nosoco-mial pneumonia. Chest 1998; 114:1383–1389
49 Martin C, Thomachot L, Quinio B, et al. Comparing two heatand moisture exchangers with one vaporizing humidifier inpatients with minute ventilation greater than 10 L/min. Chest1995; 107:1411–1415
50 Lellouche F, Qader S, Taille S, et al. Impact of ambient airtemperature on a new active HME and on standard HMES:bench evaluation. Intensive Care Med 2003; 29:S169
51 Martin C, Papazian L, Perrin G, et al. Preservation ofhumidity and heat of respiratory gases in patients with aminute ventilation greater than 10 L/min. Crit Care Med1994; 22:1871–1876
52 Anesthetic and respiratory equipment: heat and moistureexchangers for use in humidifying gases in humans, 1st ed;ISO International Standard 9360. Geneva, Switzerland: In-ternational Organization for Standardization, 1992
53 Branson RD, Davis K, Campbell RS, et al. Humidification inthe intensive care unit: prospective study of a new protocolutilizing heated humidification and a hygroscopic condenserhumidifier. Chest 1993; 104:1800–1805
54 Jaber S, Pigeot J, Fodil R, et al. Long-term effects of differenthumidification systems on endotracheal tube patency: evalu-ation by the accoustic reflection method. Anesthesiology2004; 100:782–788
55 Kollef MH, Shapiro SD, Boyd V, et al. A randomized clinicaltrial comparing an extended-use hygroscopic condenser hu-midifier with heated-water humidification in mechanicallyventilated patients. Chest 1998; 113:759–767
56 Boisson C, Viviand X, Arnaud S, et al. Changing a hydropho-bic heat and moisture exchanger after 48 hours rather than 24hours: a clinical and microbiological evaluation. IntensiveCare Med 1999; 25:1237–1243
286 Original Research
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from

DOI 10.1378/chest.08-0679 2009;135; 276-286Chest
Fumagalli and Laurent BrochardSalvatore Maurizio Maggiore, Philippe Jouvet, Jean-Damien Ricard, Bruno
François Lellouche, Solenne Taillé, Frédéric Lefrançois, Nicolas Deye,Humidification Performance of 48 Passive Airway Humidifiers
February 10, 2009This information is current as of
& ServicesUpdated Information
http://www.chestjournal.org/content/135/2/276.full.htmlhigh-resolution figures, can be found at:Updated Information and services, including
References
tml#ref-list-1http://www.chestjournal.org/content/135/2/276.full.haccessed free at:This article cites 54 articles, 15 of which can be
Open AccessoptionFreely available online through CHEST open access
Permissions & Licensing
http://chestjournal.org/misc/reprints.shtml(figures, tables) or in its entirety can be found online at: Information about reproducing this article in parts
Reprints http://chestjournal.org/misc/reprints.shtml
Information about ordering reprints can be found online:
Email alerting service
online article.article. sign up in the box at the top right corner of the Receive free email alerts when new articles cit this
formatImages in PowerPoint
format. See any online article figure for directions.downloaded for teaching purposes in PowerPoint slide Figures that appear in CHEST articles can be
Copyright © 2009 American College of Chest Physicians on February 10, 2009www.chestjournal.orgDownloaded from