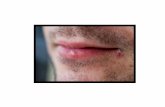
Fotos de lesões herpéticas Herpes simples I Herpes simples II Herpes zoster.
Herpes v
description
Transcript of Herpes v

HERPESVIRIDAEherpein: Greek: to creep

Herpes simplex 1 and 2 (To “The sound of silence” Simon and Garfunkle)
Hello, herpes, our old friend
Will be with us to the end.
‘Cause the virus softly creeping
Left its genes while we were sleeping
Not integrated into our genome,
It will roam that episome.
The DNA of herpes.

Herpesviruses
• Ubiquitous, all vertebrate species
• ds DNA, approximately 100 genes
• Co-evolved with their hosts, well adjusted
• Enveloped
• Latent infectionsThe case of the elephantherpesviruses
T.rex atexam time

The case of the elephant herpesviruses (Richman et al. 1999. Science 283:1171)
Asian elephant herpesvirus(“cold sores”)
Fatal hemorrhagicDisease in African elephants
African elephant herpesvirus(“cold sores”)
Fatal hemorrhagicDisease in Asian elephants

The elephant herpesvirus Richman et al. 1999. Science 283:1171

Viruses with ds DNA genomes
Poxviridae
African swine fever virus
Herpesviridae
Adenoviridae
Papovaviridaebovine herpesvirus-1,2
porcine cytomegalovirus
equine herpesviruses -1,4
malignant catarrhalfever virus

CLASSIFICATION
1. Virus FAMILY: Herpesviruses
2. Virus SUBFAMILY: Alphaherpesvirinae
a. The member viruses are characterized by:
1) A short (24 hr) replication cycle;
2) Propensity to establish latent infections in sensory ganglia, and
3) They generally are highly cytopathogenic in cell culturecausing rapid lysis of susceptible cells.
Prototype viruses in subfamily are herpes simplex and varicellazoster. Members of this subfamily include the viruses which causeinfectious bovine rhinotracheitis (Bovine herpesvirus I), equinerhinpopneumonitis, infectious laryngotracheitis of chickens, bovinemammillitis, Pseudorabies (Suid Herpesvirus I) and herpes simplexinfections of humans.

2. Virus SUBFAMILY: Alphaherpesvirinae, Cont. b. Subfamily Betaherpesvirinae
1) Individual cytomegaloviruses
2) Have restricted host range
3) Replicative cycle is slow and cell-associated.
4) Virus may be latent in lymph tissue, secretory glands.
5) Members include Human cytomegalovirus (HCMV), porcine cytomegalovirus (PCMV), BHV-4.
c) Gammaherpesvirinae
1) Defined in terms of Epstein-Barr virus (EBV).
2) Members have narrow host range and replicate in lymph cells
3) Latent virus frequently in lymphoid tissues
4) Member include EB, Karposi Sarcoma virus (HHV-8), malignant catarrhal fever
CLASSIFICATION

GENERAL CHARACTERISTICS
1. One molecule of double stranded DNA per virion.
2. The nucleocapsid measures 100-110 nm in diameter, as middle to largesized virus.
3. The virion is 120-150 nm in diameter.
4. The molecular wt is about 92 x 10-6 daltons. It takes about 1,000 basepairs per gene, so herpesvirus has a potential for abut 144 genes, butthere are open reading frames which reduce that potential. A gene isrequired for each protein of the virion. Codons, 3 adjacent nucleotides,code for amino acids of the proteins specified by the gene.
5. Genome: Linear nonsegmental ds DNA, (120-240 Kd)

Herpesvirus Family Tree (Genetic)
CLASSIFICATION
GAMMAHERPESVIRUSES
EHV2 HVS
HHV8EBV
HSV1
HSV2
EHV1
PRV
VZV
ALPHAHERPESVIRUSESBETAHERPESVIRUSES
HHV7
HHV6
HCMV

Structure
nucleocapsid
envelope
tegument glycoproteins(gB, gC…gL)

6. Polypeptides
a. Glycosylated and nonglycosylated proteins.
The nonglycosylated proteins are, by and large structural proteins.There are at least 20 nucleocapsid polypeptides (proteins) of which 3-5 are considered to be major proteins) of which 3-5 are considered to bemajor proteins, based on the number of replicates (amount of each).Viz., one is 146K daltons and in total constitutes about 40% of the capsidprotein.
The envelope contains mostly glycosylated proteins and these are theproteins against which the immune system of the host reacts. There arerelatively few major glycoproteins present. The lipoproteins are alsoderived from the virus modified nuclear membrance of the host cellduring the assembly process. These structural lipoproteins render thevirus susceptible to inactivation by lipolytic agents. About 50% of allthe viral proteins are envelope proteins.
GENERAL CHARACTERISTICS

6. Polypeptides, Cont.
b. Major envelope proteins.
There are 4. Three of the four are linked covalently by disulfide bonds.These comprise glycoprotein (gB, gC and gD) and a) at 125k; b) at 74k,and c) at 58k.
c. Minor envelope proteins.
Three Gp’s, GpI at 130k; Gp IV at 98k and Gp V at 62k form anoncovalently linked complex with a 115k nonglycosylated protein.
GENERAL CHARACTERISTICS

BIOLOGICAL PROPERTIES
1. The virion is sensitive to lipolytic agents such as ether andchloroform.
2. Eosinophilic intranuclear inclusions develop in infected cells. Theseare called Type A or Cowdry inclusions.

REPLICATION
The genome structure of herpesviruses: (A) Alphaherpesvirus genomes comprise two regions designated long (L) and short (S). Terminal repeat (TR) and internal repeat (IR) sequences may bracket unique sequences (UL, US) of both L and S or only S. Repeat sequences are shown as boxes and are encoded as indicated by the direction of the arrows. Repeat sequences allow the DNA they bracket to invert relative to the rest of the genome such that where both UL and US are bracketed by repeat sequences, four isomers are made and packaged in equimolar amounts into virions. Where only S is bracketed by repeat sequences, two equimolar isomers are made. (B) The genome of equine herpesvirus 2, a betaherpesvirus, contains terminal direct repeat structures. (C) The genome of bovine herpesvirus 4, a gammaherpesvirus, contains multiple direct terminal repeat sequences (small boxes) in a nonequal, variable number of copies.

Replicative cycle
IE genesE genes
L genesDNA replication
viral DNA
gB
gC
gD
vhs
tif
inclusion body cell-cellspread

REPLICATION
Diagram representing transcription, translation, and DNA replication of a typical herpesvirus. Transcription and posttranscriptional processing occur in the nucleus, translation in the cytoplasm, and some of the and proteins are involved in further transcription and some proteins in DNA replication.

Productive and latent infections

Viral DNA
Latency and Reactivation
LATIE genes(regulatory)
E genes (enzymes)
L genes (structural)
stressimmunosuppression
corticosteroidscAMP
reactivation

Herpesviruses That Cause Diseases in Domestic Animals
Herpesviruses That Cause Diseases in Domestic Animals
Virus Disease
Subfamily AlphaherpesvirinaeBovine herpesvirus 1
Bovine herpesvirus 2Bovine herpesvirus 5Caprine herpesvirus 1Porcine herpesvirus 1Equine herpesvirus 1Equine herpesvirus 3Equine herpesvirus 4Canine herpesvirus 1Feline herpesvirus 1Avian herpesvirus 1Avian herpesvirus 2Duck herpesvirus 1Cercopithecine herpesvirus 1
Subfamily BetaherpesvirinaeEquine herpesvirus 2Equine herpesvirus 5Porcine herpesvirus 2
Subfamily GammaherpesvirinaeAlcelaphine herpesvirus 1Bovine herpesvirus 4
Infectious bovine rhinotracheitis, infectious pustularvulvovaginitis,infectious balanoposthitis, abortionBovine mammillitis, pseudo-lumpyskin diseaseEncephalitisConjunctivitis, respiratory diseasePseudorabies, Aujeszky’sAbortionCoital exanthemaRhinopneumonitisHemorrhagic disease of pupsFeline viral rhinotracheitisInfectious laryngotracheitisMarek’s diseaseDuck plagueHerpes simplex-like disease in macaques, paralysis in man(also called B virus)
Equine cytomegalovirus infectiousUnknownInclusion body rhinitis, generalized cytomegalovirus infection
Bovine malignant catarrhal feverUnknown

1. Diseases Caused by Alphaherpesvirinae
a. BHV-1 (Infectious bovine rhintracheitis – IBR)
1) Causes a variety of diseases in cattle:- Rhinotracheitis- Pustular vaginitis- Conjunctivitis- Abortion- Enteritis- Encephalitis
2) Rapid expansion of feedlots in 1950’s led to recognition of newdisease syndromes
3) Immunization/Control- BHV-1 vaccines
MLV, DNA vaccine- Eradication by test/removal
DISEASES CAUSED BY HERPESVIRUSES

Bovine herpesviruses
Type Subfamily Clinical entity
BHV-1.1 alpha IBR
BHV-1.2 alpha IPV
BHV-1.3 alpha encephalitis
BHV-2 alpha mammalitis
BHV-3 gamma Malignant
catarrhal fever
BHV-4 beta ?

BHV-1(IBR virus)
• Infectious bovine rhinotracheitis
• Disseminated fatal disease of newborns
• Abortions
• Genital lesions
• Encephalitis

Infection in the animal
reactivationfromlatency
replication in epithelial cells(rhinotracheitis)
Infection of susceptible animal
establishment oflatency(infection for the first time)
systemiccell-associatedspread
infection of the fetus-> abortion
encephalitis
replication inepithelial cells

Primary lesion
dermisepidermis
virus, infected cells debris
eroded lesionpustule
cytokines
fever, dyspnea, coughcell associated viremia

Clinical and immunological parameters
Days after infection
5 10 15 20
temperature+
++
+
+
+
+
5 10 15 20
interferon
IgM
IgG
CMI
virus shedding

Complications of BHV-1 infection
• abortions
• encephalitis
• fatal disease in newborn calves
• Shipping fever (P. hemolytica, P. multocida)– Bronchopneumonia -> fibrinous
pleuropneumonia

Factors contributing to shipping fever
• Environmental– Crowding animals from different sources– Stress
• Host-virus– Paralysis of mucociliary escalator– Release of iron and nutrients– Increased colonization– immunosuppression

Diagnosis of BHV
• Virus isolation
• Immunofluorescence
• Immunohistochemistry
• PCR
• Serology– Virus neutralization– ELISA

Vaccination against BHV-1
• Modified-live (attenuated)– Intra nasal– Intra muscular
• Inactivated– Intra muscular

Intranasal vs intramuscular vaccines
Route of Inoculation Advantages Disadvantages
Intra nasal Stimulates mucosal and systemic immunity. Stimulates interferon. Less affected by maternal antibody.
Immunity not as long-lasting as IM.
Serum antibody titres lower.
Difficult to administer.
Intra muscular Longer lasting, higher levels of immunity.
Susceptible to maternal antibody.

Considerations for vaccination
• Economics
• New born calves

Vaccines for eradication
gB
gBgB
gB
gene for gEdeleted
gE gB
infected
vaccinated
ELISA for antibodies
infected
vaccinatedgD subunits
gE gD
gD gD

1. Diseased Caused by Alphaherpesvirinae, Cont.
b. Pseudorabies Virus (Suid Herpesvirus I - Aujeszky’s Disease [maditch])
1) First described late 1890 in cattle, in swine due to intensification ofswine operations
2) Clinical features – Swine
- Reproductive: abortions
- CNS – mortality
- Respiratory
- Age-related signs
DISEASES CAUSED BY HERPESVIRUSES

PRV - Aborted Fetuses
DISEASES CAUSED BY HERPESVIRUSES

Neonatal: Infection
DISEASES CAUSED BY HERPESVIRUSES

CNS Signs
DISEASES CAUSED BY HERPESVIRUSES

PRV Encephalitis - in situ
DISEASES CAUSED BY HERPESVIRUSES

PRV - Pneumonia
DISEASES CAUSED BY HERPESVIRUSES

PRV - Infected tonsils - in situ
DISEASES CAUSED BY HERPESVIRUSES

DISEASES CAUSED BY HERPESVIRUSES
Suid Herpesvirus Infection
Neurologic Stage Respiratory Stage
1. Acute (28d post-infection) 1. Follow neurology (>7d PI)
2. Clinical signs evidenced by: Ataxia Lateral recumbence Fatigue
2. Clinical signs evidenced by: Coughing, sneezing Labored breathing
3. High mortalityHigh morbidity
3. Low mortalityHigh morbidity
4. Encephalitis Destruction of neurons Glial cell involvement
4. Interstitial Pneumonia Due to primary virus infection Secondary bacteria
5. Due to virus replication in neurons,inflammation
5. Due to virus replication in AM,functional impairment

1. Diseased Caused by Alphaherpesvirinae, Cont.
b. Pseudorabies Virus (Suid Herpesvirus I - Aujeszky’s Disease [mad itch]),Cont.
3) Clinical features – other animals
- Primarily neurological “mad itch”
- Pruritis, paralyses (dogs, cats, mice, rabbits)
DISEASES CAUSED BY HERPESVIRUSES

Reported Host Spectrum of PrV
Naturally Susceptible Species
Farm Animals Companion Animals Feral Animals
CattleGoatSheepSwine
CatDog
BadgerCoatimundiCoyoteDeerMouseRabbitRaccoonRat
Experimentally Infectible Species
Laboratory Animals Feral Animals Birds Companion Animals
MouseRatGuinea PigFerretMarmoset MonkeyRhesus Monkey
DeerGroundhog or WoodchuckHedgehogJackalMuskratOpossumPorcupineBat, brown
BuzzardChickenDuck, mallardDuck, unspecified domestic (Taiwan)GooseHawk, sparrowPigeonTurkey
Naturally Refractive Species
Mammals Poikilotherms Insects
Ape (Barbary)Chimpanzee
FrogSnakeToadTortoise
Lice (hog)Tick (hog)

Pruritis in Mice
DISEASES CAUSED BY HERPESVIRUSES

1. Diseased Caused by Alphaherpesvirinae, Cont.
b. Pseudorabies Virus (Suid Herpesvirus I - Aujeszky’s Disease [maditch]), Cont.
4) Diagnosis
- Serology – ELISA, differential, SN
- Virus – VI, IFA, PCR
5) Immunization / Control
- Vaccine – MLV, differental
- Control – Clean up program
DISEASES CAUSED BY HERPESVIRUSES

1. Diseased Caused by Alphaherpesvirinae, Cont.
c. Equine Herpesvirus I1) Abortions as early as 4 months of gestation2) Major cause of equine abortion3) Occasionally respiratory disease
d. Equine Herpesvirus 3 (Equine coital examthema)
e. Equine Herpesvirus 4 (Equine rhinopneumonitis)1) Foals over 2 months (weaning and yearling)2) Clinical signs: Fever anorexia, serous nasal discharge,
occasionally bronchopneumonia and death3) Source older horses4) Immunization – MLV and killed vaccines for both EHV-1 and
EHV-4
DISEASES CAUSED BY HERPESVIRUSES

Equine herpesviruses• 9 types, 1,3 and 4 are of veterinary importance
Equine herpestype
Commonname
Clinical entity
EHV-1 equineabortion virus
respiratory,encephalitis,abortions
EHV- 4 equine rhino-
pneumonitisvirus
respiratory
EHV-3 coitalexanthema
virus
genital lesions

EHV-1 and EHV-4
• Immunologically related
• Respiratory disease
• EHV-1causes abortions, neurological disease
• Immunity short lived
• Reinfection or reactivation

EHV-1 and EHV-4 (respiratory disease)
• Incubation period (2-10 days)
• Primary infection in young horses
• Biphasic fever
• Nasal discharge, cough
• Uncomplicated cases -> complete recovery 1-2 weeks

Pathogenesis of EHV-1 abortions:
bronchioles
EHV infects endothelialcells and leukocytes
cell-associatedviremia
IL2 inducesAdhesion molecules
inflammation
Thromboticocclusion
vasculitis
Virus infects fetus
Endometrial vessel

• Respiratory epithelium
• Lamina propria
• Drainage lymph nodes
• Circulation
• Dissemination of infection
Key stages in pathogenesis
(Allen et al., 1998)

efferentlymphatic
EHV-1 epithelial invasion and generation of viremia
Infected endothelial cells in epitheliallamina propria
Infected T lymphocytes in drainage lymph nodes
ViremiaViremia
Infection of epithelium and lamina propria
(Kydd et al., 1994)

Viremia
• central to pathogenesis
• strictly cell associated
• EHV-1 infects mainly CD5+/CD8+ T lymphocytes (Scott et al., 1983; Slater et al., 1994; Meulen et al., 2000)
• associated with neutropaenia and lymphopaenia termination of viraemia requires generation of class I restricted, CD8+ cytotoxic T lymphocytes (CTL) (Allen et al., 1995; O’Neill et al., 1999)
• viremic lymphocytes express virus antigens briefly and are only transient targets for CTL
• after this virus persists in lymphocytes in a latent form

Circulatory immune responses: summary
• Strong humoral response to infection with transient CF and longer lived VN antibody responses (Thompson et al. 1976)
• Protection from reinfection is short lived (Allen and Bryans 1986)
• No correlation between circulating antibody levels and protection from re-infection (Hannant et al. 1993; Mumford et al. 1994)
• MHC I restricted CD8+ CTL responses are generated in response to infection (Allen et al. 1995)

EHV-1 abortions
• Late in gestation (7th to 11th month)
• Few weeks to several months after respiratory outbreak
• Abortion storms

Pathogenesis of abortion
viremia
endometrial endothelial cell infection
endometrial vasculitis and thrombosis

Pathogenesis of abortion
viremia
endometrial endothelial cell infection
endometrial vasculitis and thrombosis
extensive infarction: virus negative fetus

Pathogenesis of abortion
viremia
endometrial endothelial cell infection
endometrial vasculitis and thrombosis
extensive infarction: virus negative fetus
limited infarction: virus positive fetus

Myeloencephalopathy by EHV-1
• Often but not always associated with respiratory disease
• Often several horses
• Sudden onset rapid progression, early stabilization
• Ataxia, paresis, urinary incontinence, cystitis
• Little evidence of viral replication in neural tissues (immune mediated?)
• Vasculitis, thrombosis, hemorrhages
• (See Wilson, Vet Clin N. Am - equine pract. 1997 13:53)

Prevention of EHV-1 disease
• Management
• Vaccines
– Short-lived immunity (mares - 5,7,9 months of gestation
– Reduce severity but do not prevent
– Exacerbate myeloencephalitis??

Other herpesviruses
• alpha herpesviruses– Canine herpesvirus– Feline herpesvirus– Marek’s disease
• beta herpesviruses– porcine cytomegalovirus
• gamma herpesviruses– malignant catarrhal fever

1. Diseased Caused by Alphaherpesvirinae, Cont.
f. Canine Herpesvirus 1
1) Recognized 1965, cause of highly fatal, generalized hemorragicdisease of pups < 4 weeks of age
2) Syndrome rare, antibodies <20%
g. Feline Herpesvirus 1
1) Respiratory disease in first year of life
2) Incidence ~ 70% for colonies, 50% household
3) Clinical: Sneezing, coughing, profuse serous nasal and oculardischarge, anorexia, weight loss and fever. Corneal ulcerscommon.- Bronchopneumonia – may be fatal- Disease similar to feline calcivirus- Abortion in pregnant queens- Immunization – Killed and MLV vaccine
DISEASES CAUSED BY HERPESVIRUSES

canine herpesvirus
• wide-spread, usually asymptomatic
• generalized fatal disease in puppies (fading puppy syndrome)
• hypothermia
• no vaccine

feline herpesvirus
• feline rhinotracheitis
• wide spread in catteries
• respiratory infections
• abortions - no direct viral involvement
• vaccines

1. Diseased Caused by Alphaherpesvirinae, Cont.
h. Avian Herpesvirus I (Infectious larygotracheitis)
1) First identified in chickens – U.S. 1926
2) Clinical signs
- Chickens of all ages susceptable
- Most common 4 – 18 months
- Mild coughing, hard sneezing
DISEASES CAUSED BY HERPESVIRUSES

1. Diseased Caused by Alphaherpesvirinae, Cont.
i. Avian Herpesvirus 2 (Marek Disease)1) Specific herpesvirus etiology 1967.
2) Disease worldwide
3) Prior to introduction of vaccine most widespread lymphoproliferative disease
4) Clinical signs (Progressive disease – 4 syndromes)- Neurolymphatosis (Classical Marek” – incoordination)- Acute Marek’s: Neurological ataxia, paralysis, significant
mortality- Ocular lymphomatoses – greying of iris- Cutaneous Marek’s – nodular lesions
5) Diagnosis- History, clinical signs, age and postmortem findings, histopathology- Virus isolation, IFA
6) Immunization- MLV- 1 day old chicks
DISEASES CAUSED BY HERPESVIRUSES

Marek’s disease
• lymphoproliferative, neurological disease of young chickens
• unilateral paralysis, ataxia due to infiltration of spinal nerves
• involvement of iris, skin
• stable in feather follicle dander

2. Diseases Caused by Betaherpesviruses
a. Porcine Cytomegolovirus (Porcine herpesvirus 2)1) First recognized in 1955 – endemic in swine population
2) 1% feature in rhinitis in swine up to 10 weeks of age, “inclusionbody rhinitis”
3) May cause fetal death or generalized disease in first 2 weeks of age
4) Diagnosis- Primariliy histopathology
- basophilic inclusion bodies- virus isolation difficult
- Antibodies NO
- Immunization NO
b. Equine cytomegolovirus (EHV-2)
1) Respiratory disease?
DISEASES CAUSED BY HERPESVIRUSES

beta herpesvirus - porcine cytomegalovirus
• asymptomatic if endemic in herds
• in susceptible herds– inclusion body rhinitis– fetal death– runting, poor growth in young piglets

3. Diseases Caused by Gammaherpesvirus
a. EB - human
b. KSV – HHV - 8
c. Bovine Malignant Catarrhal Fever
d. Bovine Herpesvirus 5
1) Conjunctivitis
2) Respiratory disease
3) Vaginitis
4) Metritis
5) Lymphosarcoma
DISEASES CAUSED BY HERPESVIRUSES

malignant catarrhal fever (gamma herpesvirus)
other ruminants ?
other species
malignant catarrhal fever

clinical signs in susceptible species
• peracute– high fever, diarrhoea, death in 1-3 days
• acute– fever, depression, enlarged lymph nodes, serous
nasal discharge, erosive lesions, corneal opacity, high mortality
• chronic form

acute MCF

IMMUNE RESPONSES TO HERPESVIRUSES
1. Nonspecific
a. Interferons
b. TNF, IL-1
c. Macrophages
d. NK cells
2. Specific
a. Humoral1) ADCC2) Neutralization3) Antibody-complement lysis
b. Cell-mediated1) T-helper cell-blastogenesis, cytokine (CD4
+)
2) CTL – (either CD4+ or CD8
+)

Involvement of Glycoproteins in the Induction of Immunity
Property gE gB gC gG gD gl
MAbs neuralize - + + - + -
MAbs neutralize + C + + + = + -
Neutralizing activity inpig serum
- -/+ + - + -
MAbs mouseprotection
+ + + - + -
MAbs pig protection - + + - + np
Induction CTL in mice - np + - np -
Protected by glyco-protein
np np + - + np
IMMUNE RESPONSES TO HERPESVIRUSES

Herpesviruses in which Vaccines are of Significance
Herpesvirus Abbr Host SpeciesCommercial
Vaccine
Marek disease virus MDV Chicken MLV, killed
Bovine herpesvirus 1 BHV-1 Cattle MLV, killed
Suid herpesvirus 1(pseudorabies virus)
SHV-1(PRV)
Swine MLV, killed
Equine herpesvirus 1 EHV-1 Horse MLV, killed
Feline herpesvirus FHV-1 Cat MLV, killed
IMMUNE RESPONSES TO HERPESVIRUSES

Evolution of Herpesvirus Vaccines
IMMUNE RESPONSES TO HERPESVIRUSES
1st Generation
- Conventional killed- Modified-live- e.g. PR-Vac, Pseudo vax
2nd Generation
- Gene-deleted, i.e. virulence genes- e.g. Omni Vac-1
3rd Generation - Gene-deleted, i.e. differential marker- e.g. PRV-marker, Tolvid Omnivac II
4th Generation
- Multiple marker genes- e.g. PRV-Gold
??

Goal of Vaccine to HerpesvirusIdeal objective of any vaccine against herpesvirus. To preclude thedevelopment of latent infection.
To do so:
Vaccine must prevent multiplication portal of entry
Secondary Goal:
Preclude virus that was reactivated from reaching peripheral site
Nonetheless to date:
No vaccine has protected against viral infection.
IMMUNE RESPONSES TO HERPESVIRUSES