Helicobacter pylori and risk of gastric cancer and ... fileHypothesis Hp Gastritis risk of cancer...
Transcript of Helicobacter pylori and risk of gastric cancer and ... fileHypothesis Hp Gastritis risk of cancer...

Falk Symposium 149Highlights in
Gastrointestinal OncologyBerlin, October 1-2, 2005
Helicobacter pylori and risk ofgastric cancer and lymphoma
W. FischbachAschaffenburg, Germany

Helicobacter pylori
Peptic ulcer
MALT lymphoma gastric cancer
dyspepsiaExtragastricdiseases

Helicobacter pyloriis a crucial factor in the pathogenesis
of gastric cancer
Helicobacter pyloriis a group I carcinogene
WHO 1994

Helicobacter pylori and gastric cancerEvidence for an association
• Epidemiology• Induction of gastritis, atrophy, IM• Ascorbin acid• Hyperproliferation• O2-radicals, NO• Hypochlorhydria, hypergastrinemia• Animal studies• Observational studies• Interventional studies

Hp and risk of gastric cancerMetaanalysis of prospective case-control-studies
Danesh, Aliment Pharmacol Ther
Further increase of risk by 1.6 in case of cagA +Huang, Gastroenterology 2003

Population based case-control study
More than 2/3 of non-cardia cancerare attributable to Helicobacter pylori
Ekstrom, Gastroenterology 2001
Hp and risk of gastric cancer
case-control study with rigorous exclusion criteria
OR of non-cardia cancer:3.7 18.3 for any Hp5.7 28.4 for CagA +
Brenner, Am J Epidemiol 2004

Hp and risk of gastric cancerFamilial cancer risk and Hp
German Health and Nutrition Survey1351 male and female, age 30-74 years
0
10
20
30
40
50
60
70
%
familial cancer no cancer
Hp prevalence
Brenner et al., Gastroenterology 2000
Hp is an independent risik factor(RR 2.7)
Familial risk of gastric cancer:due to intrafamilial clustering
of Hp infection ?

Familial cancer risk and Hp
Hp cagA positive individuals with a family historyof gastric cancer have
8-fold increased risk for gastric cancer16-fold increased risk for non-cardia cancer
Independent risk factors
BackgroundFamilial history of gastric cancer:
Is it a consequence of a clustering of Hp infectionor are Hp and familial history independent risk factors ?
Brenner, Cancer 2000

Gastric cancerGenetic disposition and Hp
Hypothesis
Hp Gastritis
risk gastritisrisk of cancer
Genetic disposition

Cytokine genetic polymorphismsare associated with an increased risik for
gastric cancer(Machado, Gastroenterology 2003)
• Proinflammatory IL-1 polymorphisms are associated withmore severe inflammation and increased prevalence of IM andatrophic gastritis. ATA haplotype carriers of IL-10 werecolonized by more virulent Hp strains(Rad et al., Gut 2004)
• Genetic polymorphisms in IL-8, IL-10, and TNF-alphaplay a role in gastric carcinogenesis in the Chinese population(Lu et al., Carcinogenesis 2005)
• IL-1B polymorphisms may predispose to gastric cancer in theChinese pand Korean population(Yang et al., Cancer Lett 2004; Chang et al., Int J Cancer 2005)

Host genetics Hp strains
risk gastritis phenotype
gastric cancer
Environmental and dietary factors
Hp and risk of gastric cancer

Helicobacter pylori and gastric cancerEvidence for an association
• Epidemiology• Induction of gastritis, atrophy, IM• Ascorbin acid• Hyperproliferation• O2-radicals, NO• Hypochlorhydria, hypergastrinemia• Animal studies• Observational studies• Interventional studies

Hp and gastric cancerUemura et al., N Engl J Med 2001
patients with gastric/duodenal ulcer, gastral hyperplasia, NUDn = 1526
endoscopc-bioptic survey over 7.8 years (1-10,6)
Hp +n=1246
Hp -n=280
cancer in 2,9% cancer in 0%Hp -Hp +
p < 0,001

Hp and gastric cancerUemura et al., N Engl J Med 2001
Cancer risk was associated with:
severe atrophyCorpus dominant gastritis
intestinal metaplasia
Hp positive patients
2,9 %NUD4,7%
Hplasia2,2%
Uv3,4%
Ud0 %
cancer risk

Hp and gastric cancerChemopreventive study
Correa et al., J Natl Cancer Inst 2000
Columbia: high prevalence of cancer and of multifocal atrophy/IM
randomisation
Hp eradication Beta-Caroten Ascorbin acid
Comparison of histologies before and after 6 years:significant reduction of atrophy/IM
combination not superior to monotherapy

Hp eradication for prevention ofgastric cancer in a high risk area
Wong BCY et al., JAMA 2004
GASTRO UPDATE 2005 Oberer GI-Trakt 145/1
prospective randomised placebo controlledpopulation-based interventional study in
1630 healthy Hp infected individuals from aChinese region with high cancer incidence
n = 988initially no precancerous lesions
(atrophy, IM, dysplasia)

Hp eradication for prevention ofgastric cancer in a high risk area
Wong BCY et al., JAMA 2004
GASTRO UPDATE 2005 Oberer GI-Trakt 145/2
primary study aim:Is there a difference in the incidence of
gastric cancer between eradication and placeboduring a 7.5-years follow-up ?
Gastric carcinomaeradication n = 7placebo n = 11p 0,33

Hp eradication for prevention ofgastric cancer in a high risk area
Wong BCY et al., JAMA 2004
GASTRO UPDATE 2005 Oberer GI-Trakt 145/3
secondary study aim:Is there a difference between individuals
with / without precancerous lesionsin the eradication and control group ?
Gastric carcinoma
eradication n = 0placebo n = 6p 0,02
no precancerous lesions

Gastric carcinogenesisnormal gastric mucosa
chronic-active gastritis
atrophic gastritis
intestinal metaplasia
dysplasia
carcinoma
Hp Hp
? point of no return
?

Prevalence of Hp and gastric cancerin long-living elderly in JapanMasaya Kawade et al., J Gastroenterol Hepatol 2004
0
10
20
30
40
50
60
%
65-74 75-84 > 85
Hp seropositivity
02468
1012
%
65-74 75-84 > 85
Prevalence
gastric cancer colon cancer
* **
The lower prevalence of gastric cancer in the elderlymay be due to disappearance of Hp colonization

Host genetics Hp strains
risk gastritis phenotype
gastric cancer
Environmental and dietary factors
Hp and risk of gastric cancer
Hp genotypinghost genotyping
selective early prophylactic Hp eradication

Gastric cancer originating frombone marrow-derived cells and Hp
Houghton J, et al., Science 2004
• Bone marrow-derived cells (BMDCs) are frequentlyrecruited to sites of inflammation
• Hp infection of mice induced population of the stomachwith BMDCs.
• Subsequently, these cells progressed through metaplasiaand dysplasia to cancer
• Epithelial cancers can originate from BMDCs after Hpinduced gastritis.New aspects of host and exogene factors interactions withpotential implications on carcinogenesis

Helicobacter pylori
Peptic ulcer
MALT lymphoma gastric cancer
dyspepsiaExtragastricdiseases

Hp eradication forgastric cancer prophylaxis ?
Hp eradication should be performedin cases of• EMR or subtotal surgical resection ofhigh grade dysplasia or gastric cancer
• familial clustering of gastric cancer• risk gastritis
(predominant corpus gastritis, atrophy, IM)• young patient (< 40 years ?)
• patient´s desire

Helicobacter pylori
Peptic ulcer
MALT lymphoma gastric cancer
dyspepsiaExtragastricdiseases

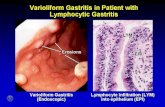
Helicobacter pylori and MALT-lymphoma
aetiopathogenetic roleof Hp for MALT-lymphoma
molecularbiologicalfindings
epidemiologicaldata
animalstudies
morphologicaldata

Helicobacter pylori and gastric MALT lymphoma
Morphological data
Lymphoid follicles in gastric mucosa represent animmune response to Campylobacter pylori. Thisacquired lymphoid tissue has characteristics of MALT.(Wyatt 1988; Stolte 1989)
Successful treatment of Hp is followed by regressionof MALT(Stolte 1992)
Hp infection is present in 92% of 110 patients withgastric MALT lymphoma(Wotherspoon 1991)

Helicobacter pylori and gastric MALT lymphoma
Epidemiological data
The incidence of gastric MALT lymphoma depends onthe rate of Hp infection(Doglioni, Lancet 1992)
Case-control-study:Hp leads to a 6-fold risk for gastric MALT lymphoma.Hp infection preceeds the development for gastriclymphoma.(Parsonnet, NEJM 1994)

Helicobacter pylori and gastric MALT lymphoma
Molecularbiological data
The growth of tumor cells from low grade gastricMALT lymphoma can be stimulated by Hp
The neoplastic B cells are not immuneresponsive to Hpbut lymphoma proliferation is mediated bycontact-dependent Hp specific T cells
Hp dependent T cell activation B cell proliferationmonoclonal selection
(Hussel, Lancet 1993, J Pathol 1996)

Helicobacter pylori and gastric MALT lymphoma
Hp induces acquisition of lymphoid tisuein the gastric mucosa:Condition for the developmentof gastric MALT lymphoma
Hp represents an antigenic stimulus forthe lymphoma growth:Progression of gastric MALT lymphoma

HELICOBACTER PYLORI ERADICATION INLOW GRADE GASTRIC MALT - LYMPHOMA
Wotherspoon et al., Lancet 1993
“Hp eradicationshould be the first choice of treatment “
6 patients with low grade gastric B-cell-lymphomaof MALT-type
Hp eradication
regression of lymphoma: 5/6 cases

Helicobacter pylori eradicationin low grade gastric MALT-lymphoma
of stage I
lymphoma regression in 70 - 80%
(literature)

What is the long-term outcome afterexclusive Hp eradication therapy ?
Are lymphoma regressions long lasting ?
Does it offer a real chance of cure ?
How often do relapse, progressionor high grade transformation occur ?

CR62 (65%)
MRD15 (16%)
PR9 (10%)
NC7 (7%)
PD1 (1%)
?1 (1%)
Hp eradication
n = 95
Follow-up49,5 (12-89 mo)
Fischbach et al., Gut 2004;53:34-37

Long-term outcomeafter Helicobacter pylori eradication therapy
Conclusions
The majority of patients havea favourable long-term outcome.
Hp eradication offers a real chance of cure

Are there any predictors forlymphoma regression
after successful eradication of Hp ?

Hp associated gastritisreactive benign disease
malignancy
Hp
dependent
geneticchanges
Marginal zone B celllymphoma of MALTt(1;14) < 5% Apoptose regulierendes BCL10
t(11;18) -t(11;18) +
DLBCLt(11;18) -
numerous genetic changes
high gradetransformation
de novo
mostly singleclonal abnormality
Hp eradicationfailure
(definition?)
Hp eradicationresponsive
Hp induces andSustains an acttive
ProliferatingB-cell population
Hp attracts andactivates neutrophils
Release

Translocation t(11;18) and regression ofgastric MALT-lymphoma after Hp eradication
Liu et al., Gastroenterology 2002
46
1 0 1
17
26
4
16
05
101520253035404550
I >II I >II
t(11;18) -t(11;18) +
completeregression
noregression
t(11;18) positive MALT-lymphoma do not/rarelyrespond to Hp eradication therapy:
predictor of treatment failure ?

Minimal residual low-gradegastric MALT-type lymphoma aftereradication of Helicobacter pylori
Fischbach W, Goebeler-Kolve ME, Starostik P,Greiner A, Müller-Hermelink HK
Lancet 2002; 360:547-548

Fischbach et al., Lancet 2002
No relapseNo lymphoma dissemination
No high grade transformation

MRD after successful eradication of H. pyloriExperience from a clinical series
of the EGILS group (Fischbach W, DDW 2005, submiited)
Follow-up42,2 (2–144 months)
PD n = 6 (6%)
MRD n = 67 (62%)
Hp eradicationafter 12 months:
MRD
Low grade gastricMALT lymphoma
stage In = 108
CR n = 35 (32%)
local progression: n=5high grade lymphoma n=1
Favourable naturalcourse of disease
in 95%

It is justifiable topostpone oncological treatment
in patients with MRDafter successful eradication of Hp
A watch-and-wait strategy isa valid approach to manage MRD

Therapy of early gastric lymphoma
MALT
Hp eradicationif Hp-, progression
or relapse:RTx (surgery)
MRD:watch-and-wait
RTx (surgery)
CTx
large B-cell-ly
CTx + RTx( surgery + CTx)
((Hp eradication))
CTx + RTx(surgery + CTx)
CTx + RTx
stage
I 1/2
II 1/2
III / IV