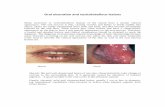
GRAFTJACKET - wmtemedia.com 3 GRAFTJACKET ® Matrix ... painful plantar weight bearing surface that...
-
Upload
trinhkhuong -
Category
Documents
-
view
223 -
download
5
Transcript of GRAFTJACKET - wmtemedia.com 3 GRAFTJACKET ® Matrix ... painful plantar weight bearing surface that...

P L A N T A R S O F T - T I S S U E A U G M E N T A T I O N S U R G I C A L
T E C H N I Q U E
GRAFTJACKETRegenerative Tissue Matrix
™

GRAFTJACKET®
Regenerative Tissue Matrix
SURGICAL TECHNIQUE
Surgical Technique as described by:
Thomas Rocchio, DPMwith Comments byDavid G. Armstrong, DPM, PhD

Wright recognizes that proper surgical procedures and techniques are the responsibility of the medical professional. The following guidelines are furnished for information purposes only. Each surgeon must evaluate the appropriateness of the procedures based on his or her personal medical training, experience, and patient condition. Prior to use of the system, the surgeon should refer to the product Instructions For Use package insert for additional warnings, precautions, indications, contraindications and adverse effects. Instructions For Use package inserts are also available by contacting the manufacturer. Contact information can be found on the back of this surgical technique and the Instructions For Use package inserts are available on wmt.com under the link for Prescribing Information.
Please contact your local Wright representative for product availability.
Contents
Chapter 1 4 Introduction
Chapter 2 5 Product Information
Chapter 3 6 Surgical Technique
Positioning and Set-up
Explant Information
Appendix A 10 Sizing and Ordering Information
Appendix B 11 Technical Comments

Chapter 1 Introduction
Introduction 1chap
ter
4
The plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using GRAFTJACKET™ Matrix covers and protects the underlying soft tissues and bone.

Information 2chapter
Chapter 2 Information 5
The GRAFTJACKET Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body.
The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The GRAFTJACKET Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. Coupled with excellent tensile and suture retention strength, the biological characteristics of the GRAFTJACKET Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
The GRAFTJACKET Matrix provides a scaffold for host cell repopulation and revascularization.

Surgical TechniquePlantar Augmentation Applied to Either Heel Pad or Metatarsal Pad
Presented by: Thomas Rocchio, DPM
3chap
ter
Chapter 3 Surgical Technique6
Surgical TechniquePlantar Augmentation Applied to Either Heel Pad or Metatarsal Pad
Presented by: Thomas Rocchio, DPM
3chap
ter
Chapter 3 Surgical Technique6
Step 1
Pre-Operative MeasurementPerform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
Step 2
Surgical ProcedureUsing blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to FIGURE 1
Step 3
GRAFTJACKET® Matrix PreparartionGRAFTJACKET® Matrix Preparatio Rehydrate the GRAFTJACKET® Matrix allowing sufficient time according to the instructions for use. The author prefers the GRAFTJACKET® MAXFORCE EXTREME, which is an average thickness of 2.0mm. After rehydration, cannula in the same orientation outside the shoulder as they are in the subacromial space. The grasper should pass through the opposite side of the cannula to avoid suture entanglement.
IntroductionThe plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using gRAFTJAcKeT® Matrix covers and protects the underlying soft tissues and bone.
Product InformationThe gRAFTJAcKeT® Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body. The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The gRAFTJAcKeT® Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. coupled with excellent tensile and suture retention strength, the biological characteristics of the gRAFTJAcKeT® Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
STEP 1
Pre-Operative Measurement
Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
STEP 2
Surgical Procedure
using blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to Figure 1.
STEP 3
GRAFTJACKET® Matrix Preparation
Rehydrate the gRAFTJAcKeT® Matrix allowing sufficient time according to the instructions for use. The author prefers the gRAFTJAcKeT® MAXFORce EXTREME, which is an average thickness of 2.0mm. After rehydration, the gRAFTJAcKeT® Matrix is cut to the desired shape using the pre-operative measurements.
Figure 1
GRAFTJACKET® Regenerative Tissue Matrix
Figure 1
Figure 2 Figure 2
Heel Pad Augmentation
Metatarsal Pad Augmentation
Surgical Technique Plantar Augmentation Applied to Either Heel Pad or Metatarsal PadPresented By Thomas Rocchio, DPM
IntroductionThe plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using gRAFTJAcKeT® Matrix covers and protects the underlying soft tissues and bone.
Product InformationThe gRAFTJAcKeT® Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body. The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The gRAFTJAcKeT® Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. coupled with excellent tensile and suture retention strength, the biological characteristics of the gRAFTJAcKeT® Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
STEP 1
Pre-Operative Measurement
Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
STEP 2
Surgical Procedure
using blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to Figure 1.
STEP 3
GRAFTJACKET® Matrix Preparation
Rehydrate the gRAFTJAcKeT® Matrix allowing sufficient time according to the instructions for use. The author prefers the gRAFTJAcKeT® MAXFORce EXTREME, which is an average thickness of 2.0mm. After rehydration, the gRAFTJAcKeT® Matrix is cut to the desired shape using the pre-operative measurements.
Figure 1
GRAFTJACKET® Regenerative Tissue Matrix
Figure 1
Figure 2 Figure 2
Heel Pad Augmentation
Metatarsal Pad Augmentation
Surgical Technique Plantar Augmentation Applied to Either Heel Pad or Metatarsal PadPresented By Thomas Rocchio, DPM
FIGURE 1 FIGURE 1
Heel Pad Augmentation Metatarsal Pad Augmentation
GRAFTJACKET Matrix PreparationRehydrate the GRAFTJACKET Matrix allowing sufficient time according to the instructions for use. The author prefers the GRAFTJACKET MAXFORCE EXTREME, which is an average thickness of 2.0mm. After rehydration, the GRAFTJACKET Matrix is cut to the desired shape using the pre-operative measurements
Step 1
Pre-Operative Measurement Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
Step 2
Surgical ProcedureUsing blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to FIGURE 1
FIGURE 1A FIGURE 1B

Chapter 3 Surgical Technique 7Chapter 3 Surgical Technique 7
Step 4
Traction SuturesAttach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the GRAFTJACKET® Matrix. Refer to FIGURE 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other
Repeat with second suture and needles to the other deep corner. Refer to FIGURE 3. Ensure the sutures do not become knotted.
IntroductionThe plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using gRAFTJAcKeT® Matrix covers and protects the underlying soft tissues and bone.
Product InformationThe gRAFTJAcKeT® Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body. The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The gRAFTJAcKeT® Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. coupled with excellent tensile and suture retention strength, the biological characteristics of the gRAFTJAcKeT® Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
STEP 1
Pre-Operative Measurement
Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
STEP 2
Surgical Procedure
using blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to Figure 1.
STEP 3
GRAFTJACKET® Matrix Preparation
Rehydrate the gRAFTJAcKeT® Matrix allowing sufficient time according to the instructions for use. The author prefers the gRAFTJAcKeT® MAXFORce EXTREME, which is an average thickness of 2.0mm. After rehydration, the gRAFTJAcKeT® Matrix is cut to the desired shape using the pre-operative measurements.
Figure 1
GRAFTJACKET® Regenerative Tissue Matrix
Figure 1
Figure 2 Figure 2
Heel Pad Augmentation
Metatarsal Pad Augmentation
Surgical Technique Plantar Augmentation Applied to Either Heel Pad or Metatarsal PadPresented By Thomas Rocchio, DPM
IntroductionThe plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using gRAFTJAcKeT® Matrix covers and protects the underlying soft tissues and bone.
Product InformationThe gRAFTJAcKeT® Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body. The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The gRAFTJAcKeT® Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. coupled with excellent tensile and suture retention strength, the biological characteristics of the gRAFTJAcKeT® Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
STEP 1
Pre-Operative Measurement
Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
STEP 2
Surgical Procedure
using blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to Figure 1.
STEP 3
GRAFTJACKET® Matrix Preparation
Rehydrate the gRAFTJAcKeT® Matrix allowing sufficient time according to the instructions for use. The author prefers the gRAFTJAcKeT® MAXFORce EXTREME, which is an average thickness of 2.0mm. After rehydration, the gRAFTJAcKeT® Matrix is cut to the desired shape using the pre-operative measurements.
Figure 1
GRAFTJACKET® Regenerative Tissue Matrix
Figure 1
Figure 2 Figure 2
Heel Pad Augmentation
Metatarsal Pad Augmentation
Surgical Technique Plantar Augmentation Applied to Either Heel Pad or Metatarsal PadPresented By Thomas Rocchio, DPM
FIGURE 2
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 2STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
FIGURE 3
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 3
Step 4
Traction SuturesAttach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the GRAFTJACKET Matrix. Refer to FIGURE 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other
Repeat with second suture and needles to the other deep corner. Refer to FIGURE 3. Ensure the sutures do not become knotted.
Chapter 3 Surgical Technique 7
Step 4
Traction SuturesAttach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the GRAFTJACKET® Matrix. Refer to FIGURE 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other
Repeat with second suture and needles to the other deep corner. Refer to FIGURE 3. Ensure the sutures do not become knotted.
IntroductionThe plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using gRAFTJAcKeT® Matrix covers and protects the underlying soft tissues and bone.
Product InformationThe gRAFTJAcKeT® Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body. The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The gRAFTJAcKeT® Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. coupled with excellent tensile and suture retention strength, the biological characteristics of the gRAFTJAcKeT® Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
STEP 1
Pre-Operative Measurement
Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
STEP 2
Surgical Procedure
using blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to Figure 1.
STEP 3
GRAFTJACKET® Matrix Preparation
Rehydrate the gRAFTJAcKeT® Matrix allowing sufficient time according to the instructions for use. The author prefers the gRAFTJAcKeT® MAXFORce EXTREME, which is an average thickness of 2.0mm. After rehydration, the gRAFTJAcKeT® Matrix is cut to the desired shape using the pre-operative measurements.
Figure 1
GRAFTJACKET® Regenerative Tissue Matrix
Figure 1
Figure 2 Figure 2
Heel Pad Augmentation
Metatarsal Pad Augmentation
Surgical Technique Plantar Augmentation Applied to Either Heel Pad or Metatarsal PadPresented By Thomas Rocchio, DPM
IntroductionThe plantar fat pad, made up of mooring ligaments and columnar fat at the sub-metatarsal pads and/or heel pads, protects and cushions the underlying bone, tendons and neurovascular structures of the foot. With any number of traumatic events, including multiple surgical incisions, fractures, or chronic metatarsal plantar flexion, this plantar fat pad can become displaced or atrophied. This often results in a painful plantar weight bearing surface that can lead to callus or ulceration. Plantar soft-tissue augmentation using gRAFTJAcKeT® Matrix covers and protects the underlying soft tissues and bone.
Product InformationThe gRAFTJAcKeT® Regenerative Tissue Matrix is a human dermal collagen template that is readily incorporated into the body. The Matrix undergoes a patented process that renders the material essentially acellular and is freeze-dried with a proprietary process that prevents the formation of ice crystals to preserve the intact matrix including vascular channels.
The gRAFTJAcKeT® Matrix provides a scaffold for host cell repopulation, revascularization and, ultimately, conversion to host tissue. coupled with excellent tensile and suture retention strength, the biological characteristics of the gRAFTJAcKeT® Matrix make it an excellent scaffold to reinforce primary repairs in the foot and ankle while eliminating morbidity associated with harvesting autograft.
STEP 1
Pre-Operative Measurement
Perform clinical measurement using diagnostic ultrasound to establish the graft thickness and area needed. Mark the desired position of the graft on the foot.
STEP 2
Surgical Procedure
using blunt and sharp dissection, create the desired sized pocket in the deep subcutaneous tissue. Refer to Figure 1.
STEP 3
GRAFTJACKET® Matrix Preparation
Rehydrate the gRAFTJAcKeT® Matrix allowing sufficient time according to the instructions for use. The author prefers the gRAFTJAcKeT® MAXFORce EXTREME, which is an average thickness of 2.0mm. After rehydration, the gRAFTJAcKeT® Matrix is cut to the desired shape using the pre-operative measurements.
Figure 1
GRAFTJACKET® Regenerative Tissue Matrix
Figure 1
Figure 2 Figure 2
Heel Pad Augmentation
Metatarsal Pad Augmentation
Surgical Technique Plantar Augmentation Applied to Either Heel Pad or Metatarsal PadPresented By Thomas Rocchio, DPM
FIGURE 2
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 2STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
FIGURE 3
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 3
FIGURE 2A FIGURE 2B
FIGURE 3A FIGURE 3B

Chapter 3 Surgical Technique8 Chapter 3 Surgical Technique8
STEP 5
Graft ImplantationPull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in FIGURE 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to FIGURE 5.
STEP 6
ClosureClose the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to FIGURE 5.
STEP 7
Post-Operative ProtocolThe patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
FIGURE 4
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 4
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
FIGURE 5
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 5
STEP 5
Graft ImplantationPull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in FIGURE 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to FIGURE 5.
Chapter 3 Surgical Technique8
STEP 5
Graft ImplantationPull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in FIGURE 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to FIGURE 5.
STEP 6
ClosureClose the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to FIGURE 5.
STEP 7
Post-Operative ProtocolThe patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
FIGURE 4
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 4
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
STEP 4
Traction Sutures
Attach (2) double needle, non-absorbable sutures to what will be the two deepest corners of the gRAFTJAcKeT® Matrix. Refer to Figure 2. Drive both needles from one suture inside the subcutaneous pocket and through skin at one of the far corners of the defect. Place the two sutures ends approximately 3-4mm apart from each other.
Repeat with second suture and needles to the other deep corner. Refer to Figure 3. ensure the sutures do not become knotted.
STEP 5
Graft Implantation
Pull the sutures from the far end of the pocket to draw the graft into the body and into the predetermined position. The arrow in Figure 4 indicates the direction to pull. Tie the suture ends into a knot. Refer to Figure 5.
STEP 6
Closure
close the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to Figure 5.
STEP 7
Post-Operative Protocol
The patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.
Figure 4
Figure 3
Figure 4
Figure 5 Figure 5
Figure 3
Heel Pad Augmentation
Metatarsal Pad Augmentation
“Plantar soft-tissue augmentation may be valuable for a broad patient population but current modalities
and techniques have had, to date, limited success. Dr. Rocchio’s technique using the GRAFTJACKET® Matrix
via a minimal incision may prove an excellent new alternative to address this unmet clinical need.”
- David G Armstrong, DPM, PhD
FIGURE 5
Heel Pad Augmentation Metatarsal Pad Augmentation
FIGURE 5
FIGURE 4A FIGURE 4B
FIGURE 5A FIGURE 5B
STEP 6
ClosureClose the subcutaneous tissue and repair the skin using your preferred method. Wrap the patient’s foot in a soft compressive dressing. Refer to FIGURE 5.
STEP 7
Post-Operative ProtocolThe patient should remain non-weight bearing for 2 weeks, while also maintaining standard wound care at the incision site. At two weeks, the sutures are removed and patient may begin weight bearing.

Chapter 3 Surgical Technique 9
Explant InformationIf the removal of the implant is required due to revision or failure of the device, the surgeon should contact the manufacturer using the contact information located on the back cover of this surgical technique to receive instructions for returning the explanted device to the manufacturer for investigation.
Postoperative ManagementPostoperative care is the responsibility of the medical professional.

Chapter 2 Surgical Technique10
Headline Headline A
pp
end
ix
PROFEMUR® Xm Total Hip System10 Appendix A Ordering Information
GRAFTJACKET® MAXIMUM FORCE PART NUMBER QUANTITY
4X7CM REGENERATIVE TISSUE MATRIX - MAXIMUM FORCE 8600-4X07 1EA Non-Meshed, Thick (Average Thickness = 1.4mm)
5X5CM REGENERATIVE TISSUE MATRIX - MAXIMUM FORCE 86MX-5X05 1EA Non-Meshed, Thick (Average Thickness = 1.5mm)
INDICATIONS |Augmentation, Rotator cuff, Achilles tendon, Quadriceps, Patellar tendon, Capsular reinforcement, and Extrasynovial ligament augmentation
GRAFTJACKET® MAXIMUM FORCE EXTREME PART NUMBER QUANTITY
4X7CM REGENERATIVE TISSUE MATRIX - MAXFORCE EXTREME 86UM-4X07 1EA Non-Meshed, Thick (Average Thickness = 2.0mm)
INDICATIONS |Augmentation, Rotator cuff, Achilles tendon, Quadriceps, Patellar tendon, Capsular reinforcement, and Extrasynovial ligament augmentation
Additional Products
GRAFTJACKET® - HAND SURGERY PART NUMBER QUANTITY
2X4CM REGENERATIVE TISSUE MATRIX - HAND SURGERY 8600-2X04 1EA Non-Meshed, Thin (Average Thickness = 0.5mm)
INDICATIONS |Flexor/Extensor Tendon Reinforcement, Tendon sheath augmentation of the hand and foot
GRAFTJACKET® REGENERATIVE TISSUE MATRIX PART NUMBER QUANTITY
5X5CM REGENERATIVE TISSUE MATRIX 8600-5X05 1EA Non-Meshed, Standard (Average Thickness = 1.0mm)
INDICATIONS |General tendon augmentation, Rotator cuff augmentation, Periosteal covering - small defects
GRAFTJACKET® REGENERATIVE TISSUE MATRIX PART NUMBER QUANTITY
5X10CM REGENERATIVE TISSUE MATRIX 8600-5X10 1EA Non-Meshed, Standard (Average Thickness = 1.0mm)
INDICATIONS |General tendon augmentation Periosteal covering - large defects
Sizing and Ordering Information A
pp
end
ix A
8600-2X04GRAFTJACKET® REGENERATIVE TISSUE MATRIX- HAND SURGERYDimensions 2 x 4cm 1eA.Average Thickness 0.5mm
8600-0530GRAFTJACKET® SLR- REGENERATIVE TISSUE MATRIXLIGAMENT REINFORCEMENTDimensions 5 x 30Mm 1eA.Average Thickness 1.0mm
876P-0400MINI IGNITE® Power Mix- INJECTABLE STIMULUS FOR SMALL BONEFRACTURE CALLOUS FORMATION 4CC
86DR-0300ALLOMATRIX® DR GRAFT- GENERAL FOOT AND ANKLE BONE GRAFTING 3CC
8400-0511OSTEOSET® RESORBABLE MINI BEAD KIT- FINDS USE IN BONE VOIDS SECONDARY TO OSTEOMYELITIS IN DIABETIC FEET 5CC
This GRAFTJACKET® Matrix is intended to be used as a scaffold for repair of damaged or inadequate, integumental tissue and does not contain wound healing agents.
Many variables including patient pathology, anatomy, and surgical techniques may influence procedural outcomes. Before use, physicians should review all risk information, which can be found in the ‘Directions for Use’ attached to the packaging of each GRAFTJACKET® Graft.
™Trademarks and ®Registered marks of Wright Medical Technology, inc.
Products processed by lifecell corporation for Wright Medical Technology, inc.
©2008 Wright Medical Technology, inc. All Rights Reserved. SK 429-1107 R311
Wright Medical Technology, Inc.5677 Airline RoadArlington, TN USA 38002901.867.9971 phone800.238.7188 toll-freewww.wmt.com
Wright Medical EMEAKrijgsman 11 1186 DM Amstelveen The Netherlands 011.31.20.545.0100 www.wmt-emea.com
8600-5X05GRAFTJACKET® REGENERATIVE TISSUE MATRIXDimensions 5 x 5cm 1eA.Average Thickness 1.0mm
8600-5X10GRAFTJACKET® REGENERATIVE TISSUE MATRIXDimensions 5 x 10cm 1eA.Average Thickness 1.0mm
8600-4X07GRAFTJACKET® REGENERATIVE TISSUE MATRIX- MAXIMUM FORCEDimensions 4 x 7cm 1eA.Average Thickness 1.5mm
86MX-5X05GRAFTJACKET® REGENERATIVE TISSUE MATRIX- MAXIMUM FORCEDimensions 5 x 5cm 1eA.Average Thickness 1.5mm
86TM-4X04GRAFTJACKET® REGENERATIVE TISSUE MATRIX- ULCER REPAIRDimensions 4 x 4cm 1eA.Average Thickness 0.6mm
86TM-4X08GRAFTJACKET® REGENERATIVE TISSUE MATRIX- ULCER REPAIRDimensions 4 x 8cm 1eA.Average Thickness 0.6mm
86GL-0200GRAFTJACKET® XPRESS- FLOWABLE SOFT-TISSUE SCAFFOLD 2CC
ADDITIONAL BIOLOGICS FOR FOOT AND ANKLE SURGERY
TECHNICAL COMMENT REGARDING GRAFT SELECTIONThe gRAFTJAcKeT® Regenerative Tissue Matrix is available in multiple dimensions and thicknesses (from 0.5mm to 2.0mm average thickness). Selection of the appropriate graft is the responsibility of the operating surgeon based on the individual patient needs and circumstances. graft thickness and graft strength requirements are considrations in appropriate selection. generally, thicker grafts have increased strength.
Ordering information
86UM-4X07GRAFTJACKET® REGENERATIVE TISSUE MATRIX - MAXFORCE - EXTREMEDimensions 4 x 7cm 1eA.Average Thickness 2.0mm
Proper surgical procedures and techniques are the reponsibility of the medical professional. The above guidelines are furnished for information purposes only. Each surgeon must evaluate the appropriateness of the procedures based on his or her personal medical training and experience. Prior to use of the system, the surgeon should refer to the product package insert for complete warnings, precautions, indications, contraindications, and adverse effects. Package inserts are also available by contacting Wright Medical Technologies, Inc.
GRAFTJACKET® - HAND SURGERY PART NUMBER QUANTITY
0.5x3CM REGENERATIVE TISSUE MATRIX - HAND SURGERY 8600-0530 1EA Non-Meshed, Thin (Average Thickness = 1.4mm)
INDICATIONS |Flexor/Extensor Tendon Reinforcement, Tendon sheath augmentation of the hand and foot

Headline Headline A
pp
end
ix
PROFEMUR® Xm Total Hip System 11Appendix A Ordering Information
GRAFTJACKET® MAXIMUM FORCE PART NUMBER QUANTITY
4X7CM REGENERATIVE TISSUE MATRIX - MAXIMUM FORCE 8600-4X07 1EA Non-Meshed, Thick (Average Thickness = 1.4mm)
5X5CM REGENERATIVE TISSUE MATRIX - MAXIMUM FORCE 86MX-5X05 1EA Non-Meshed, Thick (Average Thickness = 1.5mm)
INDICATIONS |Augmentation, Rotator cuff, Achilles tendon, Quadriceps, Patellar tendon, Capsular reinforcement, and Extrasynovial ligament augmentation
GRAFTJACKET® MAXIMUM FORCE EXTREME PART NUMBER QUANTITY
4X7CM REGENERATIVE TISSUE MATRIX - MAXFORCE EXTREME 86UM-4X07 1EA Non-Meshed, Thick (Average Thickness = 2.0mm)
INDICATIONS |Augmentation, Rotator cuff, Achilles tendon, Quadriceps, Patellar tendon, Capsular reinforcement, and Extrasynovial ligament augmentation
Additional Products
GRAFTJACKET® - HAND SURGERY PART NUMBER QUANTITY
2X4CM REGENERATIVE TISSUE MATRIX - HAND SURGERY 8600-2X04 1EA Non-Meshed, Thin (Average Thickness = 0.5mm)
INDICATIONS |Flexor/Extensor Tendon Reinforcement, Tendon sheath augmentation of the hand and foot
GRAFTJACKET® REGENERATIVE TISSUE MATRIX PART NUMBER QUANTITY
5X5CM REGENERATIVE TISSUE MATRIX 8600-5X05 1EA Non-Meshed, Standard (Average Thickness = 1.0mm)
INDICATIONS |General tendon augmentation, Rotator cuff augmentation, Periosteal covering - small defects
GRAFTJACKET® REGENERATIVE TISSUE MATRIX PART NUMBER QUANTITY
5X10CM REGENERATIVE TISSUE MATRIX 8600-5X10 1EA Non-Meshed, Standard (Average Thickness = 1.0mm)
INDICATIONS |General tendon augmentation Periosteal covering - large defects
Sizing and Ordering Information A
pp
end
ix A
Headline Headline A
pp
end
ix
PROFEMUR® Xm Total Hip System10 Appendix A Ordering Information
GRAFTJACKET® MAXIMUM FORCE PART NUMBER QUANTITY
4X7CM REGENERATIVE TISSUE MATRIX - MAXIMUM FORCE 8600-4X07 1EA Non-Meshed, Thick (Average Thickness = 1.4mm)
5X5CM REGENERATIVE TISSUE MATRIX - MAXIMUM FORCE 86MX-5X05 1EA Non-Meshed, Thick (Average Thickness = 1.5mm)
INDICATIONS |Augmentation, Rotator cuff, Achilles tendon, Quadriceps, Patellar tendon, Capsular reinforcement, and Extrasynovial ligament augmentation
GRAFTJACKET® MAXIMUM FORCE EXTREME PART NUMBER QUANTITY
4X7CM REGENERATIVE TISSUE MATRIX - MAXFORCE EXTREME 86UM-4X07 1EA Non-Meshed, Thick (Average Thickness = 2.0mm)
INDICATIONS |Augmentation, Rotator cuff, Achilles tendon, Quadriceps, Patellar tendon, Capsular reinforcement, and Extrasynovial ligament augmentation
Additional Products
GRAFTJACKET® - HAND SURGERY PART NUMBER QUANTITY
2X4CM REGENERATIVE TISSUE MATRIX - HAND SURGERY 8600-2X04 1EA Non-Meshed, Thin (Average Thickness = 0.5mm)
INDICATIONS |Flexor/Extensor Tendon Reinforcement, Tendon sheath augmentation of the hand and foot
GRAFTJACKET® REGENERATIVE TISSUE MATRIX PART NUMBER QUANTITY
5X5CM REGENERATIVE TISSUE MATRIX 8600-5X05 1EA Non-Meshed, Standard (Average Thickness = 1.0mm)
INDICATIONS |General tendon augmentation, Rotator cuff augmentation, Periosteal covering - small defects
GRAFTJACKET® REGENERATIVE TISSUE MATRIX PART NUMBER QUANTITY
5X10CM REGENERATIVE TISSUE MATRIX 8600-5X10 1EA Non-Meshed, Standard (Average Thickness = 1.0mm)
INDICATIONS |General tendon augmentation Periosteal covering - large defects
Sizing and Ordering Information A
pp
end
ix A
8600-2X04GRAFTJACKET® REGENERATIVE TISSUE MATRIX- HAND SURGERYDimensions 2 x 4cm 1eA.Average Thickness 0.5mm
8600-0530GRAFTJACKET® SLR- REGENERATIVE TISSUE MATRIXLIGAMENT REINFORCEMENTDimensions 5 x 30Mm 1eA.Average Thickness 1.0mm
876P-0400MINI IGNITE® Power Mix- INJECTABLE STIMULUS FOR SMALL BONEFRACTURE CALLOUS FORMATION 4CC
86DR-0300ALLOMATRIX® DR GRAFT- GENERAL FOOT AND ANKLE BONE GRAFTING 3CC
8400-0511OSTEOSET® RESORBABLE MINI BEAD KIT- FINDS USE IN BONE VOIDS SECONDARY TO OSTEOMYELITIS IN DIABETIC FEET 5CC
This GRAFTJACKET® Matrix is intended to be used as a scaffold for repair of damaged or inadequate, integumental tissue and does not contain wound healing agents.
Many variables including patient pathology, anatomy, and surgical techniques may influence procedural outcomes. Before use, physicians should review all risk information, which can be found in the ‘Directions for Use’ attached to the packaging of each GRAFTJACKET® Graft.
™Trademarks and ®Registered marks of Wright Medical Technology, inc.
Products processed by lifecell corporation for Wright Medical Technology, inc.
©2008 Wright Medical Technology, inc. All Rights Reserved. SK 429-1107 R311
Wright Medical Technology, Inc.5677 Airline RoadArlington, TN USA 38002901.867.9971 phone800.238.7188 toll-freewww.wmt.com
Wright Medical EMEAKrijgsman 11 1186 DM Amstelveen The Netherlands 011.31.20.545.0100 www.wmt-emea.com
8600-5X05GRAFTJACKET® REGENERATIVE TISSUE MATRIXDimensions 5 x 5cm 1eA.Average Thickness 1.0mm
8600-5X10GRAFTJACKET® REGENERATIVE TISSUE MATRIXDimensions 5 x 10cm 1eA.Average Thickness 1.0mm
8600-4X07GRAFTJACKET® REGENERATIVE TISSUE MATRIX- MAXIMUM FORCEDimensions 4 x 7cm 1eA.Average Thickness 1.5mm
86MX-5X05GRAFTJACKET® REGENERATIVE TISSUE MATRIX- MAXIMUM FORCEDimensions 5 x 5cm 1eA.Average Thickness 1.5mm
86TM-4X04GRAFTJACKET® REGENERATIVE TISSUE MATRIX- ULCER REPAIRDimensions 4 x 4cm 1eA.Average Thickness 0.6mm
86TM-4X08GRAFTJACKET® REGENERATIVE TISSUE MATRIX- ULCER REPAIRDimensions 4 x 8cm 1eA.Average Thickness 0.6mm
86GL-0200GRAFTJACKET® XPRESS- FLOWABLE SOFT-TISSUE SCAFFOLD 2CC
ADDITIONAL BIOLOGICS FOR FOOT AND ANKLE SURGERY
TECHNICAL COMMENT REGARDING GRAFT SELECTIONThe gRAFTJAcKeT® Regenerative Tissue Matrix is available in multiple dimensions and thicknesses (from 0.5mm to 2.0mm average thickness). Selection of the appropriate graft is the responsibility of the operating surgeon based on the individual patient needs and circumstances. graft thickness and graft strength requirements are considrations in appropriate selection. generally, thicker grafts have increased strength.
Ordering information
86UM-4X07GRAFTJACKET® REGENERATIVE TISSUE MATRIX - MAXFORCE - EXTREMEDimensions 4 x 7cm 1eA.Average Thickness 2.0mm
Proper surgical procedures and techniques are the reponsibility of the medical professional. The above guidelines are furnished for information purposes only. Each surgeon must evaluate the appropriateness of the procedures based on his or her personal medical training and experience. Prior to use of the system, the surgeon should refer to the product package insert for complete warnings, precautions, indications, contraindications, and adverse effects. Package inserts are also available by contacting Wright Medical Technologies, Inc.
GRAFTJACKET® - HAND SURGERY PART NUMBER QUANTITY
0.5x3CM REGENERATIVE TISSUE MATRIX - HAND SURGERY 8600-0530 1EA Non-Meshed, Thin (Average Thickness = 1.4mm)
INDICATIONS |Flexor/Extensor Tendon Reinforcement, Tendon sheath augmentation of the hand and foot

™ and ® denote Trademarks and Registered Trademarks of Wright Medical Group N.V. or its affiliates.©2016 Wright Medical Group N.V. or its affiliates. All Rights Reserved.
010661B_25-Jul-2015
1023 Cherry Road Memphis, TN 38117 800 238 7117 901 867 9971 www.wright.com
56 Kingston RoadStaines-upon-ThamesSurrey TW18 4NLUnited Kingdom+44 (0)845 833 4435
161 Rue Lavoisier38330 Montbonnot Saint MartinFrance+33 (0)4 76 61 35 00