Esters Team
-
Upload
gem-toledo -
Category
Documents
-
view
241 -
download
0
Transcript of Esters Team

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 1/17
Esters

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 2/17
Defnition:
Esters are chemical compounds derived rom acid (organic orinorganic) in which at least one -OH (hydroxyl) group is replacedy an -O-al!yl (al!oxy) group" #sually$ esters are derived rom acaroxylic acid and an alcohol"

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 3/17
&pplications:
Esters that are have ragrant odours are used as a constituent operumes$ essential oils$ ood 'avourings$ cosmetics$ etc
Esters are used as an organic solvent
atural esters are ound in pheromones
aturally occurring ats and oils are atty acid esters o glycerol
hospoesters orm the ac!one o D& molecules itrate esters$ such as nitroglycerin$ are !nown or their
explosive properties
olyesters are used to ma!e plastics
Esters are used to ma!e suractants E"g" soap$ detergents

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 4/17
hysical properties:
*oiling points:
- +mall esters have oiling points which are similar to those oaldehydes and !etones with the same numer o caron atoms"Esters$ li!e aldehydes and !etones$ are polar molecules and sohave dipole-dipole interactions as well as van der ,aals dispersionorces" However$ they do not orm ester-ester hydrogen onds$ sotheir oiling points are signifcantly lower than those o an acid witthe same numer o caron atoms"

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 5/17
hysical properties:
elting point:
- elting points determine whether the sustance is a at (a
solid at room temperature) or an oil (a li.uid at room temperature)& greater numer o doule onds$ or degree o unsaturation$ in thmolecules results in a lower melting point$ ecause the van der,aals orces are less e/ective"

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 6/17
hysical properties:
+oluility:- +mall esters are airly solule in water ut soluility decreases
with increasing chain length$ as shown elow:

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 7/17
hysical properties:
+oluility:- 0he reason or this trend in soluility is that although esters
cannot hydrogen ond with each other$ they can hydrogen ondwith water molecules" &s chain length increases$ the hydrocaronportion orces itsel etween water molecules$ rea!ing therelatively strong hydrogen onds etween water molecules withouo/ering an energetic compensation1 urthermore$ the water
molecules are orced into an ordered alignment along the chain$decreasing the entropy in the system"

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 8/17
hysical properties:

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 9/17
*asic reaction:
Esters can e converted into other esters (transesterifcation)$ the parentcaroxylic acid (hydrolysis) or amides (see aove)"
Transesterifcation : heat with alcohol and acid catalyst
Hydrolysis : heat with a." acid or ase (e.g. a." H2+O3 or a." aOH)
Amide preparation : heat with the amine$ methyl or ethyl esters are themost reactive

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 10/17
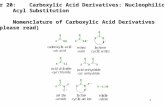
omenclature:
4irst$ identiy the oxygen that is part o the continuous chain andonded to caron on oth sides" (On one side o this oxygenthere will e a caronyl present ut on the other side there wone")
+econd$ egin numering the caron chains on either side o theoxygen identifed in step 6"
ext$ use this ormat: 7al!yl on side urther rom the caronyl8(space) 7al!ane on the side with the caronyl8 - (9n this case:7methyl8 7methane8)
4inally$ change the ending o the al!ane on the same side as thecaronyl rom -e to oate"

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 11/17

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 12/17
omenclature:
Benzenecarboxylic acid (Benzoic acid)
- Other sustituents that exist on either side o the ester arenamed in the same way as they are on regular al!ane chains" 0heonly thing you must ma!e sure o is placing the sustituent nameon the part o the name that corresponds to the side o the esterthat it is on"

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 13/17

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 14/17
octyl ethanoate
methyl propanoate

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 15/17
methyl 1-chloroet1-chloromethyl ethanoate

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 16/17

7/25/2019 Esters Team
http://slidepdf.com/reader/full/esters-team 17/17