薬化学特論w3pharm.u-shizuoka-ken.ac.jp/yakka/Japanese/manabeCLASS/...Noyori, R. Asymmetric...
Transcript of 薬化学特論w3pharm.u-shizuoka-ken.ac.jp/yakka/Japanese/manabeCLASS/...Noyori, R. Asymmetric...

平成24年度
薬化学特論
●教科書の5,8,9章の中から抜粋した内容
●評価:最終試験(11月14日)
●プリントはウェブサイト(薬化HP →「眞鍋担当授業」 → 「薬化学特論」)からダウンロード可

1
Chapter 1
Principles of
Asymmetric Synthesis
chiral
achiral

2
R1
N
R2
R3R1
P
R2
R3
NMe3
34 kJ/molPMe3
133 kJ/mol
atropisomer
inversion barrier

3
P
P
Ph
Ph
MeO
OMe
DIPAMP
ZrCl
Cl
Fe
PPh2
PCy2
JOSIPHOS
C2 symmetry C1 symmetryCn : 360 / n
180O rotation
C3 symmetry
CO2HHO
HO CO2H
BH R2
R1

4
optically active
racemic
誤用例1「・・・・BINAPは、炭素‐炭素単結合
の回転が妨げられているため、光学活性な分子構造をもつ。」
誤用例2「・・・・以前に我々はラセミ体の1を合成した。今回、キラルな1の合成に成功したので報告する。」
1
HN
HO2C H
H

5
...Current usage of the term ‘chiral’ in a generalized manner such as, ‘chiral synthesis, ‘chiral approach’, etc. is misleading and incorrect....
Hanessian, S. Total Synthesis of Natural Products: The ‘Chiron’ Approach; Pergamon Press: Oxford, 1983, p 21.
► chiral pool synthesis
► resolution
► asymmetric synthesis (narrow definition)
Synthesis of Optically Active Compounds(Asymmetric Synthesis)
“classical” resolutionkinetic resolution
diastereoselectiveenantioselective stoichiometric
catalytic
absolute asymmetric synthesis

6
“classical” resolution
R CO2
NH3
R CO2
NH3
R CO2H
NH
R CO2H
NH
H
O
H
O
MeO
MeO N
N
OO
H
H
H
H
R CO2
NH
H
O
MeO
MeO N
N
OO
H
H
H
H
R CO2
NH
H
O
NaOHH2O
R CO2
NH
H
ONaOHH2O
R CO2
NH3pure salt
pH ~6
O O
R*
OLi
RX

7
Enders, D.; Eichenauer, H. Chem. Ber. 1979, 112, 2933.
ON
NH2OMe
(SAMP)
NN
OMe
LDA;Me2SO4
NN
OMe O3
O
70% overall yield99% ee
Meyers, A. I. et al. J. Am. Chem. Soc. 1981, 103, 3081.
Hashimoto, S.; Koga, K. Tetrahedron Lett. 1978, 573.
Koga, K. et al. J. Am. Chem. Soc. 1994, 116, 8829.
Koga, K. et al. J. Chem. Soc., Chem. Commun. 1990, 1657.

8
enantiomeric excess (ee)enantiomeric ratio (er)optical purity (op)optical yieldasymmetric yield
% ee ∆∆G‡ (kJ/mol)–100 oC
∆∆G‡ (kJ/mol)23 oC
ratio ratio
99.9 10.9 17.7 99.95:0.05 1999
99.5 8.63 14.7 99.75:0.25 399
99 7.63 13.0 99.5:0.5 199
98 6.62 11.3 99:1 99
95 5.28 9.05 97.5:2.5 39
90 4.23 7.25 95:5 19
80 3.18 5.41 90:10 9
70 2.51 4.27 85:15 5.7
60 2.01 3.39 80:20 4
50 1.59 2.72 75:25 3
30 0.88 1.51 65:35 1.9
10 0.29 0.50 55:45 1.2
Energy differences between two isomers as a function of ee and temp.
Cf.) Koskinen, A. AsymmetricSynthesis of Natural Products; John Wiley & Sons, 1993.

9
∆∆G‡ (kJ/mol)
% ee
23 oC
% op
% ee
Optical Purity vs. Enantiomeric Excess
CO2H
HO2C
in CHCl3
Horeau, A. Tetrahedron Lett. 1969, 3121.

10
selectivity factor (s) = kfast / kslow
Fu, G. C. et al. J. Am. Chem. Soc. 2002, 124, 10296.
0.25 0.5 0.75 1
0.25
0.5
0.75
1
Cf.) Sharpless, K. B., et al. J. Am. Chem. Soc. 1981, 103, 6237.
0 25 50 75 100
100
75
50
25
0
% ee remaining starting material
% conversion
s = 2
3
5
10
20
100
1000

11
OMe
OO
OMe
OO
OMe
OHO
OMe
OHO
+
major minor
OMe
OHO
OMe
OHO
+
Dynamic Kinetic Resolution
100% conversion, dr = 99:1, 92% ee
Noyori, R. et al. J. Am. Chem. Soc. 1989, 111, 9134.
Ph
O
H+ Et2Zn
Ph
OH
Et
96% yield82% ee
t-BuN
OH
(11% ee, 2 mol %)
Ph
O
H+ Et2Zn
Ph
OH
Et
92% yield95% ee
(15% ee, 8 mol %)
NMe2
OH
Asymmetric amplification
Oguni, N. et al. J. Am. Chem. Soc. 1988, 110, 7877.
Noyori, R. et al. J. Am. Chem. Soc. 1989, 111, 4028.
Kagan, H. B. et al. J. Am. Chem. Soc. 1986, 108, 2353. - - - nonlinear effects

12
練習問題1
柴﨑らによって開発されたヘテロバイメタリック錯体(下図)は、様々な不斉反応の触媒として用いられている(参考文献:Acc. Chem. Res. 2009, 42, 1117.)。中心のランタニドが正八面体型をとるとき、この錯体にはどのようなCn軸が存在するか? すべての軸を図に書き入れ、それぞれCnのnがいくつなのかも示せ(n > 1)。
練習問題2下の速度論的分割(Vedejs, E. et al. J. Org. Chem. 1996, 61, 430.)のselectivity factor (s)を、未反応の出発物質のeeから計算して求めよ。

13
Chapter 2
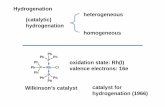
Asymmetric Hydrogenation
(catalytic)hydrogenation
Hydrogenation
homogeneous
heterogeneous
Wilkinson’s catalyst
oxidation state: Rh(I)valence electrons: 16e
catalyst for hydrogenation (1966)

14
大野桂二、佐治木弘尚, Organic Square (Wako) 2008, 22, 2.
Heterogeneous Pd Catalysts for Hydrogenation
Birch, A. J.; Williamson, D. H. Org. React. 1976, 24, 1.
Wilkinson’s catalyst RhCl(PPh3)3
RhCl(PPh3)3(cat.)
H2
R R R R
R
R
R
R
> > > > >
CO2Me CO2Me CO2Me
96%
>49%
4%
26%
PtO2 or Pd/BaSO4(cat.)
H2

15
RhL L
Cl L
RhL S
Cl L
RhL H
Cl L
H2
H
S
R
RhL H
Cl L
H
R
RhL
Cl L
H
S
H
R
H
R
H
Catalytic Cycle
oxidative addition
insertionturnover-limiting step
reductive elimination
◆dihydride◆hydrogen-first mechanism
Crabtree, R. Acc. Chem. Res. 1979, 12, 331.
Crabtree’s catalyst
TOF (h–1) of hydrogenation
TON = turnover number = moles of product / moles of catalystTOF = turnover frequency = TON / time
mechanism: Ir(III) and Ir(V) rather than Ir(I) and Ir(III) ?Hall, M. B.; Burgess, K. Chem. Eur. J. 2005, 11, 6859.

16
Crabtree, R. H.; Davis, M. W. J. Am. Chem. Soc. 1986, 51, 2655.
Stork, G.; Kahne, D. E. J. Am. Chem. Soc. 1983, 105, 1072.
Directing EffectsOH, CO2Me, C=O, or OMe
Catalytic Asymmetric Hydrogenation
PhCO2H
NHAc
H2RhBF4[((R)-camp)2(cod)] (cat.)
PhCO2H
NHAc
88% ee(R)-CAMP
P
Cy
Me
OMe
PhCO2H
NHAc
H2RhCl[(R,R)-diop] (cat.)
PhCO2H
NHAc
85% ee(R,R)-DIOP
O
OPPh2
PPh2
PhCO2H
NHBz
H2
[Rh((R)-binap)(MeOH)2]+ClO4– (cat.)
PhCO2H
NHBz
100% ee(R)-BINAP
PPh2
PPh2
AcHN CO2H
NaOAcAc2O
reflux reflux
H2O–acetone+ PhCHO
AcHN CO2H
Ph
「演習で学ぶ有機反応機構」化学同人 A019

17
CAMPKnowles, et al. (1972)
P
Cy
Me
OMe
BINAPNoyori, et al. (1980)
PPh2
PPh2
PhP
MeBu
Horner, et al. (1968)Knowles, et al. (1968)
O
OPPh2
PPh2
DIOPKagan, et al. (1971)
Fe
PPh2
PPh2
NMe2
BPPFAHayashi, Kumada, et al. (1974)
N
Boc
Ph2P
PPh2
BPPMAchiwa (1976)
PPh2
PPh2
chiraphosBosnich, et al. (1977)
P
PC6H4-2-OMe
Ph
C6H4-2-OMe
Ph
DIPAMPKnowles, et al. (1975)
Akabori, S. et al. Nature 1956, 178, 323.
Catalytic Cycle
Noyori, R. Asymmetric Catalysis in Organic Synthesis; John Wiley & Sons: New York, 1994.Cf.) For hydrogen-first mechanism in cationic Rh catalysis: Gridnev, I. D.; Imamoto, T. Acc. Chem. Res. 2004, 37, 633.
◆dihydride◆olefin-first mechanism
PRh
P
S
S
PhCO2R
NHAc PRh
P ON
Ph
CO2R
H
H2
PRh
PO N
Ph
CO2R
H
H
H
PRh
P O N
Ph
CO2R
H
H
S
PhCO2R
NHAc
AB
CD
PRh
PON
Ph
RO2C
H
B'
turnover-limiting step

18
Curtin–Hammett Principle
Seeman, J. I. et al. J. Am. Chem. Soc. 1980, 102, 7741.
Tani, K.; Yamagata, S.; Akutagawa, S.; Kumobayashi, S.; Takemoto, T.; Takaya, H.; Miyashita, A.; Noyori, R.; Otsuka, S. J. Am. Chem. Soc. 1984, 106, 5208. 芥川進 有合化 1986, 44, 513.
NEt2
NEt2
99% yield98.5% eeTON = 8000
P
P
R R
R RP
P
RR
RR
Rh ClO4
R = Me
7 tons
6.7 kg
OH (–)-menthol
Enantioselective Isomerization

19
Noyori, R. Asymmetric Catalysis in Organic Synthesis; John Wiley & Sons: New York, 1994.
Morris, R. H. In Handbook of Homogeneous Hydrogenation; de Vries, J. G.; Elsevier, C. J., Eds.; Wiley-VCH: Weinheim, 2007.
◆monohydrideCatalytic Cycle

20
Hydrogenation of Ketones
Enantioselective Hydrogenation
Kinetic Resolution
Noyori, R. Asymmetric Catalysis in Organic Synthesis; John Wiley & Sons: New York, 1994.
O
OMe
O
NHBz
H2Ru(II)-cat.
OH
OMe
O
NHBz
1) HCl2) NaOH
OH
OH
O
NH2
PPh3
NO H
OTBSTBSClimi.
RuCl3·nH2OAcO2HAcONa
NO H
OTBS
OAc
N
S
S S
N
S
Route to a Key Intermediate of Carbapenem
DKR
Shimizu, H. et al. Acc. Chem. Res. 2007, 40, 1385.

21
BINAP/1,2-Diamine–Ru(II) Complex
O
H2 (8 atm)catalyst (0.2 mol%)
OH
100% yield96% ee
catalyst =
PR2
R2P
Ru
Cl
Cl
R = 4-tolyl
NH2
H2N Ph
Ph
+ KOH
For mechanistic studies, see: Noyori, R. et al. J. Am. Chem. Soc. 2003, 125, 13490.Bergens, S. H. et al. J. Am. Chem. Soc. 2008, 130, 11979.
P
PRu
H
X NH2
H2N
OO
H H
H2
P
PRu
X NH2
HN
Asymmetric Transfer Hydrogenation
Noyori, R. et al. J. Am. Chem. Soc. 1997, 119, 8738.
Ikariya, T. et al. Org. Lett. 1999, 1, 1119.

22
練習問題3下の反応は、水素の代わりに重水素、あるいはメタノールの代わりに重メタノールを用いて行った不斉水素化の結果である。式1および2の結果は、Morrisらの反応機構から説明できる。一方、基質としてアルコール類を用いたときには、式3および4のような結果となった。この結果は、反応機構がカルボン酸基質とアルコール基質とで異なることを示している。アルコール基質のときにはどのような反応機構が働くと考えられるか? 実験結果と矛盾しない触媒サイクルを提唱せよ。(参考文献:Noyori, R. Asymmetric Catalysis in Organic Synthesis; John Wiley & Sons: New York, 1994. )

23
Chapter 3
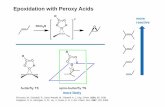
Asymmetric Epoxidation
Epoxidation with Peroxy Acids
Freccero, M.; Gandolfi, R.; Sarzi-Amadè, M.; Rastelli, A. J. Org. Chem. 2000, 65, 2030.Singleton, D. A.; Merrigan, S. R.; Liu, J.; Houk, K. N. J. Am. Chem. Soc. 1997, 119, 3385.
more likely
morereactive

24
Grieco, P. A. et al. J. Am. Chem. Soc. 1977, 99, 5773.
Epoxidation under Basic Conditions
trans-decalin half-chair
1,3-diaxial interaction
O
OO
MeO
H
t-BuOOH
Triton® B
THF
Triton® B = benzyltrimethylammonium hydroxide in H2O
O
OO
MeO
H
O
O
OO
MeO
H
O
Ot-Bu
transition metal-catalyzed epoxidation
Hoveyda, A. H.; Evans, D. A.; Fu, G. C. Chem. Rev. 1993, 93, 1307.Henbest, H. B.; Wilson, R. A. I. J. Chem. Soc. 1957, 1958.Sharpless, K. B.; Michaelson, R. C. J. Am. Chem. Soc. 1973, 95, 6136.
O
O
OH OH
relative rate
VO(acac)2t-BuOOH
1.00
>200
Henbest epoxidation
V
O
O
OO
RO
t-Bu
Epoxidation of Allyl Alcohols

25
Sharpless, K. B.; Michaelson, R. C. J. Am. Chem. Soc. 1973, 95, 6136.
Early Examples of Asymmetric Epoxidationof Allyl Alcohols
Sharpless, K. B. et al. J. Am. Chem. Soc. 1977, 99, 1990.
Yamada, S. et al. J. Am. Chem. Soc. 1977, 99, 1988.

26
R2 R1
R3 OHR
H
R2 R1
R3 OHH
R
[O]
[O]
slow
fast
Essence of Sharpless Asymmetric Epoxidation
Johnson, R. A.; Sharpless, K. B. In Catalytic Asymmetric Synthesis, 2nd ed.; Ojima, I. Ed.; Wiley-VCH: New York; 2000.
R2 R1
R3 OH
R2 R1
R3OH
O
R2 R1
R3OH
O
Ti(Oi-Pr)4t-BuOOH
CH2Cl2
CO2Et
OHEtO2C
HO
L-(+)-diethyl tartrate(natural)
CO2Et
OHEtO2C
HO
D-(–)-diethyl tartrate(unnatural)
[O]
[O]
Katsuki, T.; Sharpless, K. B. J. Am. Chem. Soc. 1980, 102, 5974.Sharpless, K. B., et al. J. Am. Chem. Soc. 1987, 109, 5765.
Katsuki, T. In Comprehensive Asymmetric Synthesis, Vol II; Jacobsen, E. N.; Pfaltz, A.; Yamamoto, H., Eds.; Springer:Berlin; 1999.
BnO OH
OBn
OBn
ClCH2SO2O
Sharplessepoxidation
L-(+)-DET
D-(–)-DET
BnO OH
OBn
OBn
ClCH2SO2O
BnO OH
OBn
OBn
ClCH2SO2O
O
O
single isomer
single isomer
Kajimoto, T.; Wong, C.-H. Tetrahedron Lett. 1995, 36, 8247.

27
Sharpless, K. B., et al. J. Am. Chem. Soc. 1981, 103, 6237.
Kinetic Resolution of Racemic Allylic Alcohols
OH
H
OH
H
OH
H
O O
+
(+)-DIPTTi(Oi-Pr)4
CH2Cl2–20 ºC
erythro threo98 : 2(S)
OH
H
OH
H
OH
H
O O
+
(+)-DIPTTi(Oi-Pr)4
CH2Cl2–20 ºC
erythro threo38 : 62(R)
fast
slow
kfast/kslow = 104
Combination of asymmetric synthesis and kinetic resolution can deliver products with very high ee.
Schreiber, S. L., et al. J. Am. Chem. Soc. 1987, 109, 1525.
OHTi(Oi-Pr)4 (1.1 equiv)t-BuOOH (2.0 equiv)(–)-DIPT (1.3 equiv)
CH2Cl2–25 ºC
80–85% yield
OH
OH
O
O
major
minor
0.5 h: 88% ee
1 h : 94% ee
1.5 h: >99.3% ee

28
OH
O
OH
O
OH
O
OH
O
OH
O
OH
O
OH
O
OH
O
OH
O
OH
O
OH
O
OH
OO O O OO O O O
k1k1 k2 k3 k4
OH
Finn, M. G.; Sharpless, K. B. J. Am. Chem. Soc. 1991, 113, 113.Katsuki, T. In Comprehensive Asymmetric Synthesis, Vol II; Jacobsen, E. N.; Pfaltz, A.; Yamamoto, H., Eds.; Springer:Berlin; 1999.
TiO
TiO
O
O
O
RO
O
t-Bu
OR
O
RO
E
E
E
O
TiO
TiO
O
O
OR
RO
O
OR
O
RO
E
EOR
OR
HO
t-BuOOH
Catalyst Structure and Proposed “Loaded” Catalyst

29
Na2(EDTA) is used to prevent trace-metal-catalyzed peroxide decomposition.
Ph Ph
O
O
O
O
OO
O
(30 mol %)
OXONE®, K2CO3
CH3CN/dimethoxymethane
0.05 M Na2B4O7 10H2O of aqueous
Na2(EDTA) (4x10-4 M) solution
OXONE® = 2KHSO5 · KHSO4 · K2SO4
94% yield95.5% ee
Shi, Y. et al. J. Am. Chem. Soc. 1997, 119, 11224.
Asymmetric Epoxidation of Unfunctionalized Olefins
dioxirane

30
Mn–Salen-Catalyzed Asymmetric Epoxidation
Jacobsen, E. N. et al. J. Am. Chem. Soc. 1991, 113, 7063. Jacobsen, E. N. et al. J. Am. Chem. Soc. 1990, 112, 2081. Katsuki, T. et al. Tetrahedron Lett. 1990, 31, 7345.
Dihydroxylation with Osmium Tetroxide
catalytic reaction
Criegee, R.; Marchand, B.; Wannowius, H. Liebigs Ann. Chem. 1942, 550, 99.
acceleration by tertiary amines
Milas, N. A.; Sussman, S. J. Am. Chem. Soc. 1936, 58, 1302.

31
PhPh
PhPh
OH
OH
N
OAc
N
OMe
(1.1 equiv)
OsO4 (1.1 equiv)
toluene
LiAlH4
90% yield83.2% ee
PhPh
PhPh
OH
OH
(1 mol%)
K2OsO2(OH)4 (0.2 mol%)K3Fe(CN)6 (3 equiv), K2CO3 (3 equiv)
CH3SO2NH2 (1 equiv)
t-BuOH/H2O
>80% yield>99.5% ee
NNO O
N N
MeO OMe
N
H
N
H
Asymmetric Dihydroxylation (AD)
Sharpless, K. B., et al. J. Am. Chem. Soc. 1980, 102, 4263.
Sharpless, K. B., et al. J. Org. Chem. 1992, 57, 2768.
.
OOs
O
O
O
R
+ L
Os O
OO
O
L
R
R
R
HO
R
OH
R
+ L
2 HO2 H2O
Os
O
O
HO
HO
OH
OH
2–
Os
O
O
HO
HO
O
O
2– 2 HO
2 HO
2 Fe(CN)63–
2 H2O
2 Fe(CN)64–
organic
aqueous
Catalytic Cycle

32
Two Proposed Mechanisms
Hoffmann, R., et al. J. Am. Chem. Soc. 1986, 108, 1867.Corey, E. J., et al. J. Am. Chem. Soc. 1996, 118, 11038.
Sharpless, K. B., et al. J. Am. Chem. Soc. 1997, 119, 1840.Houk, K. N.; Sharpless, K. B.; Singleton, D. A., et al. J. Am. Chem. Soc. 1997, 119, 9907.
練習問題4あなたは今、(–)-laulimalideの全合成の計画を立てている。その最終ステップで6個のC=C二重結合のうち1個だけを選択的に(しかも立体選択的に!)エポキシ化する計画(下式)は、無謀だろうか、それとも勝算はあるだろうか? 理由とともに述べよ。
(参考:Mulzer, J.; Öhler, E. Angew. Chem. Int. Ed. 2001, 40, 3842. Paterson, I.; De Savi, C.; Tudge, M. Org. Lett. 2001, 3, 3149. Wender, P. A.; Hegde, S. G.; Hubbard, R. D.; Zhang, L. J. Am. Chem. Soc. 2002, 124, 4956.)

33
Chapter 4
Frontier Orbitals
Atomic Orbital (AO)
E
1s
2s
3s
2p
3p

34
antibonding MO
bonding MO
Molecular Orbital (MO)
E
*
H H
H2
Molecular Orbital (MO)
E
*
*

35
Molecular Orbital (MO)
E
*
C
Cl
C Cl
C Cl

36
H
C
H
H H
H
C
H
H H
Hybridization
sp3
H
C
H
HH
H
C
H
HH
H
C
H
HH
H
C
H
HH
Siegbahn, K., et al. Chem. Phys. Lett. 1968, 1, 613.阿武聰信 ,量子化学基礎の基礎,化学同人,1996.

37
E
Butadiene

38
Staggered vs. Eclipsed Conformation of Ethane
See: Pophristic V.; Goodman, L. Nature 2001, 411, 565.See also: Bickelhaupt, M.; Baerends, E. J. Angew. Chem. Int. Ed. 2003, 42, 4183.
Weinhold, F. Angew. Chem. Int. Ed. 2003, 42, 4188.Mo, Y.; Gao, J. Acc. Chem. Res. 2007, 40, 113.
HOMO
LUMO
LUMO
HOMO

39
HOMO
LUMO
LUMO
HOMO
LUMO
HOMO
Diels–Alder Reaction
‡
H HH H
H HH H
H H
H H
H H
H H

40
LUMO
HOMO LUMO
HOMO
H HH H
H HH H
LUMO
HOMO
[4 + 2]
[2 + 2]
Salem–Klopman Equation
∆E = –∑(qa+qb)abSab + ∑ ab k<l
QkQl
Rkl
+ ∑ ∑ – ∑ ∑occ unocc occ unocc
r rs s
2(∑cracsbab)2ab
Er–Es
occ-occ Coulomb
occ-unocc

41
Hard and Soft Acids and Bases (HSAB)
Pearson, R. G. J. Am. Chem. Soc. 1963, 85, 3533.Fleming, I. (竹内敬人, 友田修司 訳) ,フロンティア軌道法入門,講談社サイエンティフィク,1978.
acid
base
hard borderline soft
(A) HO– + Br2 HOBr + Br–
(B) HO– + H+ H2O
(C) + Br2 + Br–CH2
CH2
CH2
CH2Br
(D) + H+CH2
CH2
CH2
CH3
(A) < (B)
(C) > (D)

42
Hard and Soft Acids and Bases (HSAB)
acid
base
hard soft
high positive charge
low polarizability
high-energy LUMO
high negative charge
low polarizability
low-energy HOMO
low positive charge
high polarizability
low-energy LUMO
low negative charge
high polarizability
high-energy HOMO
Jencks, W. P.; Carriuolo, J. J. Am. Chem. Soc. 1960, 82, 1778.
pKa
log k2
O2N O
O
0 2 4 6 8 10 12 14
–4
–3
–2
–1
0
1
2
3
4
5
HO
HOO
NH3
pyridine
O
O
F
N3
H2NOH
ClOH2NNH2

43
-Effect
HOMO
Fleming, I. Molecular Orbitals and Organic Chemical Reactions, John Wiley & Sons: West Sussex, 2009.
Ambident Nucleophile
charge controlfrontier orbital control

44
“From the hard and soft acids and bases (HSAB) theory,it was deduced that the substitution of hydrides in BH4
– by alkoxy groups increases the hardness of the reagent. The attack of the conjugate enone system is then enhanced at the hard site, i.e., carbon 2.”
Gemal, A. L.; Luche, J.-L. J. Am. Chem. Soc. 1981, 103, 5454.
Luche Reduction(ルーシュ還元)1,2-reduction 1,4-reduction
練習問題5下に示したエノラートアニオンのエチル化反応では、C-エチル体とO-エチル体の2つの生成物が生じうる。それぞれの生成速度定数の比(kc / ko)を測定したところ、脱離基(X)の種類によってkc / koの値が大きく異なることがわかった(下表)。この実験結果をどのように解釈したらいいだろうか?
O
OEt
O
Et X+
O
OEt
O
O
OEt
OEt
Et
Na+ kC
kO
kC / kO
X
> 100
I
60
Br
6.6
TsO
3.7
TfO

45
Chapter 5
Diels−Alder Reaction
diene
dienophile

46
Konovalov
HOMO
HOMO
LUMO
LUMO
LUMO
HOMO

47
normal electron-demand
reverse electron-demand
Fields, D. L.; Regan, T. H.; Dignan, J. C. J. Org. Chem. 1968, 33, 390.

48
Stereospecificity
+
CO2Me
CO2Me
+
CO2Me
CO2Me
CO2Me
CO2Me
CO2Me
MeO2C
OMe
CO2Me
OMe
CO2Me
OMe
CO2Me
OMe
CO2Me
OMe
CO2Me
OMe
CO2Me
EtO
O OH
HEtO
OH
HEtO
regio
endo/exo
stereo (facial)

49
Regioselectivity
Danishefsky’s diene
Danishefsky, S.; Kitahara, T. J. Am. Chem. Soc. 1974, 96, 7807.

50
Rawal’s diene
Kozmin, S. A.; Rawal, V. H. J. Org. Chem. 1997, 62, 5252.Kozmin, S. A.; Rawal, V. H. J. Am. Chem. Soc. 1997, 119, 7165.
Regioselectivity

51
LUMO
HOMO
Z
Z
Coefficients of Frontier Orbitals
large-largesmall-small
large-smallsmall-large
Fleming, I. Molecular Orbitals and Organic Chemical Reactions, John Wiley & Sons: West Sussex, 2009.

52
Z X C
Z X C
Z X C

53
endo/exo-selectivity
secondary orbital overlap
OHC
H HO
H
H
OHC H
O
H
HO
H

54
endo/exo-selectivity
electrostatic repulsion
Fleming, I. Molecular Orbitals and Organic Chemical Reactions, John Wiley & Sons: West Sussex, 2009.
H
O
H
(+)
(+)
HH
H
O
H
H
H (+)
(+)
Ohfune, Y.; Tomita, M. J. Am. Chem. Soc. 1982, 104, 3511.
N
O
Boc
TBSO
+
OTMStoluene135 ºC
N
O
Boc
TBSO H
H
OTMS
HN
HO2C H
H
CO2H
HO2C
(–)-domoic acid
regio-, endo/exo-, and facial selectivities

55
Lewis (Brønsted) acid-accelerated Diels–Alder reaction
Yates, P.; Eaton, P. J. Am. Chem. Soc. 1960, 82, 4436.
Pfeiffer, M. W. B.; Phillips, A. J. J. Am. Chem. Soc. 2005, 127, 5334.
MeO
+
Cl CNCu(BF4)2
(cat.)
0 ºC>90%
MeO
CN
Cl
KOH
H2O/DMSO80%
MeO
O
O
AcO
O
OMe
Corey lactone
CO2H
OHHO
HO
H
H
(±)-PGF2
Corey, E. J. et al. J. Am. Chem. Soc. 1969, 91, 5675.

56
O
H
O
H
H
X
X
Hashimoto, S.-I.; Komeshima, N.; Koga, K. J. Chem. Soc. Chem. Commun. 1979, 437.
Catalytic Asymmetric Diels–Alder Reaction

57
練習問題6下に示した反応の主生成物として化合物1が得られる理由を考察せよ(立体化学についても考察せよ)。
Myers, A. G. et al. J. Am. Chem. Soc. 2005, 127, 8292.
練習問題7下に示した(−)-fusarisetin A全合成中の分子内Diels−Alder反応の直前に、I2・sunlampで処理している理由は何か、考察せよ。
Theodorakis, E. A. et al. J. Am. Chem. Soc. 2012, 134, 5072.
OO
Ph3P
61% (E:Z = 3:2)
OI2 (cat.), sunlamp
then Et2AlCl, –78 ºC
82% (d.r. > 10:1)
CHO
H
H
H
H
H
H
O
NO O
H
OH
Me
OH
(–)-fusarisetin A

58
Chapter 6
Sigmatropic Rearrangement
[3,3]-Sigmatropic Rearrangement
Cope rearrangement
Claisen rearrangement

59
Claisen Rearrangement
O
OO
OMeO
HO
63%
Nicolaou, K. C.; Li, J. Angew. Chem. Int. Ed. 2001, 40, 4264.
Stereochemistry
H
O
H
H
HH
H
HO
H
H
HH
H
H
H
RR
O O
Me Me
H
O
H
H
HH
Me
HO
H
H
HH
Me
H
H
MeMe
O O
R R
Me Me
O O
Me Me
H
O
H
H
MeH
Me
HO
H
H
MeH
Me
H
H
MeMeMe Me
Me Me

60
Ziegler, F. E.; Piwinski, J. J. J. Am. Chem. Soc. 1982, 104, 7181.
(Anionic) Oxy-Cope Rearrangement
Schreiber, S. L.; Santini, C. Tetrahedron Lett. 1981, 22, 4651.
O
O
O
OO
O
18-crown-6

61
Aza-Cope Rearrangement
Knight, S. D.; Overman, L. E.; Pairaudeau, G. J. Am. Chem. Soc. 1995, 117, 5776.
aza-Cope–Mannich
Johnson–Claisen Rearrangement
OH
OEt
OEt
OEt
H+
OEt
OEt
OHO
OEtEtO
O
OEt
O
OEt
O
OEt
H

62
Johnson–Claisen Rearrangement
Takahashi, T. et al. J. Org. Chem. 1986, 51, 3393.
Ireland–Claisen Rearrangement
O
O
LDATHF
–78 ºC
LDATHF/HMPA
–78 ºC
O
LiO
O
LiO
Me3SiCl
Me3SiCl
O
TMSO
O
TMSO
O
TMSO
O
TMSO
HMPA: hexamethylphosphoramide
HMPT: hexamethylphosphorous triamide
Ireland, R. E.; Mueller, R. H.; Willard, A. K. J. Am. Chem. Soc. 1976, 98, 2868.

63
Stork, G. et al. J. Am. Chem. Soc. 1998, 120, 1337.
Carroll–Claisen Rearrangement
Eschenmoser–Claisen Rearrangement
OH
NMe2
OMe
OMe
NMe2
OMe
OHO
NMe2MeO
O
NMe2
O
NMe2
O
NMe2
H

64
[2,3]-Sigmatropic Rearrangement
Mislow rearrangement
S O S O
[2,3]-Wittig rearrangement
[3,3]-Sigmatropic Rearrangement
Cope rearrangement

65
total number of electrons = (4n + 2) ....... allowed
suprafacial-suprafacial
1
m
n
1'
[m,n]
[1,n]-Shift (Suprafacial Shift)....(4n + 2) electrons
Fleming, I. Molecular Orbitals and Organic Chemical Reactions, John Wiley & Sons: West Sussex, 2009.

66
cf. BeckmannCurtiusBaeyer–Villiger
2 electrons
Wagner–Meerwein rearrangement
[1,n]-Shift
Suprafacial Shift....(4n + 2) electrons
Antarafacial Shift....(4n) electrons
1
n
H
[1,n]
1
n
H

67
Hoeger, C. A.; Okamura, W. H. J. Am. Chem. Soc. 1985, 107, 268.Fleming, I. Molecular Orbitals and Organic Chemical Reactions, John Wiley & Sons: West Sussex, 2009.
[1,7]-Shift
4 electrons

68
Woodward–Hoffmann rule
A ground-state pericyclic change is
symmetry-allowed when the total number of
(4q+2)s and (4r)a components is odd.
Woodward, R. B.; Hoffmann, R. Angew. Chem. Int. Ed. 1969, 8, 781.
練習問題8Johnson-Claisen転位およびEschenmoser-Claisen転位を利用した以下の2つの合成スキームに関して、A~Fの化合物として妥当なものは何か? 構造式で記せ。