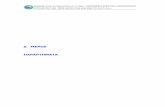
Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC...
-
date post
21-Dec-2015 -
Category
Documents
-
view
228 -
download
0
Transcript of Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC...
![Page 1: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/1.jpg)
electrode surface
stirredδ: const
unstirredδ: ↑w time
unstirredδ: ↑w time
i = nFADA (əCA / əx)
= nFADA [(CA - CAº ) /δ]
(əCA / əx) = conc. gradient
= slope of conc. profile
α diffusion rate
as CAº → 0
il = nFADACA / δ = kACA
For A + ne- = P
Eapp = E1/2 – (0.0592 / n) log (CPº / CAº)
CPº : surface conc. of product P
CAº : surface conc. of analyte A
![Page 2: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/2.jpg)
1 ms
5 ms
i α(əCA / əx) conc. gradient
α diffusion rate
10 ms
Eappl
Solution: stirred vs. unstirred
Eappl: linear scan vs. stepped
diffusion layer ↑ w time
![Page 3: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/3.jpg)
FIGURE 25-9 Current response to a stepped potential for a planar electrode in an unstirred solution. (a) Excitation potential. (b) Current response.
![Page 4: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/4.jpg)
FIGURE 25-10 Concentration distance profiles during the diffusion-controlled reduction of A to give P at a planar electrode. (a) Eappl = 0 V. (b) Eappl = point Z in Figure 25-6; elapsed time: 1, 5, and 10 ms.
![Page 5: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/5.jpg)
FIGURE 25-16 Vol’tammogram for the reduction of
oxygen in an air-saturated 0.1-M KCl solution. The lower curve is for a 0.1-M KCl solution in which the oxygen is removed by
bubbling nitrogen through the solution.
deaerated (by N2)
air-saturated
DME
0.1 M KCl
![Page 6: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/6.jpg)
FIGURE 25-17(a)
A schematic of a voltammtric system for detecting electroactive species as they elute from a column. The cell volume is determined by the
thickness of the gasket.
墊片
Amperometric Detector in Liquid Chromatography
Amperometry: to measure i at the fixed applied potential
i
Eapplied
E
thin-layer cell
amperometric
![Page 7: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/7.jpg)
FIGURE 25-17(b)
Detail of a commercial
flow cell assembly.
![Page 8: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/8.jpg)
FIGURE 25-17(c)
Configurations of working electrode blocks. Arrows shown the direction of flow in the cell.
![Page 9: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/9.jpg)
Professor Kissinger is a part time facult
y member and entrepreneur who found
ed the drug development company
Bioanalytical Systems, Inc. He regularl
y counsels students on career opportun
ities in the pharmaceutical industry and
is active in the
Indiana Instrumentation Institute (III) an
d development of the
Purdue Research Park.
Professor— Analytical Chemistry
Department of Chemistry, Purdue University
Peter T. Kissinger
B.S., 1966, Union College; Ph.D., 1970, University of North Carolina; Postdoctoral Associate, 1970-1972.
![Page 10: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/10.jpg)
Parkinson’s Disease 巴 ( 帕 ) 金森症
• 原因不明的退化性腦疾病
• 缺少 Dopamine ( 多巴胺 ) – a neurotransmitter ( 腦神經傳導物質 )
• 症狀: 顫抖 , 肌肉僵硬 , 動作緩慢
• 研究方法: mouse ( 小白鼠 ), rat ( 大白鼠 )
LC – UV
LC – Fluorescence
LC – EC
![Page 11: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/11.jpg)
pump
LC-EC
micro-dialysis
微透析
![Page 12: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/12.jpg)
From column
To waste
UV/Vis LC Detector
![Page 13: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/13.jpg)
![Page 14: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/14.jpg)
FIGURE 25-18
The Clark voltammetric
oxygen sensor.
Cathodic reaction:
O2 + 4H+ + 4e- → 2H2O.
Anodic reaction:
Ag + Cl- → AgCl(s) + e-
1956
Clark oxygen sensor
To determine the dissolved oxygen in a variety of aqueous environment, e.g., sea water, blood, sewage, etc.
Pt
Ag
Teflon or
Polyethylene (PE)
![Page 15: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/15.jpg)
(NH2)2CO + 2H2O + H+ 2NH4+ + HCO3
- (23-23)urease
pH 7.5urea
2NH3 + 2H+
Clinical test
尿素氮 ( 腎功能 )
BUN (Blood Urine Nitrogen)
FIGURE 23-13 Enzyme electrodes for measuring urea.
![Page 16: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/16.jpg)
P.10Ch1 Introduction
ISE
Immunosensors 免疫感測器
Antigen = target analyte
![Page 17: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/17.jpg)
FIGURE 21-14 Surface plasmon resonance. Laser radiation is coupled into the glass substrate coated with a thin metal film by a half-cylindrical prism. If total internal reflection occurs, an evanescent wave is generated in the medium of lower refractive index. This wave can excite surface plasmon waves. When the angle is suitable for surface plasmon resonance, a sharp decrease in the reflected intensity is observed at the detector.
Au
![Page 18: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/18.jpg)
P.734Ch25 Voltammetry
antibody
Au film
antigen= target analyte
Surface plasmon resonanace: a label-free immunosensor
![Page 19: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/19.jpg)
FIGURE 25-19(a) A: electrode containing immobilized antibody (Y); B: binding of target analyte (▼) to electrode-bound antibody; C: binding of alkaline phosphatase-labeled antibody to electrode-bound analyte; D: application of 320 mV to the electrode and addition of hydroquinone diphosphate (HQDP). Electrochemical oxidation of AP-generated hydroquinone (HQ) generates a current at the electrode that is proportional to the amount of analyte bound to the electrode.
Label 標示基
Fluorescent molecule
enzyme
(alkaline phosphatase)
Sandwich assay
antibody antigen= target analyte
W.E.
hydroquinone diphosphate
hydroquinone
quinone
i α [analyte]
![Page 20: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/20.jpg)
FIGURE 25-19(b) Photograph of the biosensor showing the arrangement of lrOx 1-mm-diameter working electrodes, 4-mm-diameter counter electrode, 7-mm-outside-diameter Ag-AgCl reference electrode, and electrical contacts on the substrate (28 × 35 × 1 mm). For clarity, the sample well is not shown.
![Page 21: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/21.jpg)
at a fixed applied potential
end point
A R A + R
A + R → P
Analyte Reagent i
E
Eapplied
titration reaction:
Stirred
P: electro-inactive
X20
![Page 22: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/22.jpg)
FIGURE 25-21 (a) Side view of an RDE showing solution flow
pattern. (b) Bottom view of a disk electrode.
rotate the electrode
rotating disk electrode, RDE
![Page 23: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/23.jpg)
FIGURE 25-21
(c) Photo of a commercial RDE.
(d) Bottom view of a ring-disk electrode.
X
![Page 24: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/24.jpg)
Rotating Disk Electrode, RDE
● The most common method for obtaining a rigorous description of the
hydrodynamic flow of stirred solution is based on a rotating disk electrode.
● Numerous studies of the kinetics and the mechanisms of electrochemical
reaction have been performed with rotating disk electrode.
il = 0.620 n F A D2/3 CA ν-1/6 ω 1/2 (25-15)
ν : kinematic viscosity (cm2/s)
ω : angular velocity
= 2 π f
f : rotation speed ( r p m )
limiting i
修訂錯誤
Levich equation:
![Page 25: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/25.jpg)
FIGURE 25-22 Disk (a) and ring (b) current for reduction of oxygen at the rotating-ring-disk electrode.
hydrodynamic
X
![Page 26: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/26.jpg)
FIGURE 25-16 Vol’tammogram for the reduction of
oxygen in an air-saturated 0.1-M KCl solution. The lower curve is for a 0.1-M KCl solution in which the oxygen is removed by
bubbling nitrogen through the solution.
deaerated (by N2)
air-saturated
DME
0.1 M KCl
![Page 27: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/27.jpg)
Cyclic Voltammetry (CV)
potential waveform
Ei (initial potential)
forward scan
reverse scan
switching potential
in unstirred soln
scan rate: 50 mV/s
װ
循環伏安法
23
![Page 28: Electrode surface stirred δ: const unstirred δ: ↑w time unstirred δ: ↑w time i = nFAD A (əC A / ə x) = nFAD A [(C A - C A º ) /δ] (əC A / əx) = conc. gradient.](https://reader036.fdocuments.net/reader036/viewer/2022062313/56649d6d5503460f94a4d34d/html5/thumbnails/28.jpg)
FIGURE 25-24 (a) Potential versus time waveform (b) cyclic voltammogram for a solution th
at is 6.0 mM in K3Fe(CN)6 and 1.0 M in KNO3.
Stationary soln
Pt-disk electrode
no reax
at Ei
Fe(CN)63-
+ e- → Fe(CN)6
4-
Fe(CN)64-
→ Fe(CN)63-
+ e-
for reversible reaction :
Epa – Epc = 0.0592 / n
ipc ≈ ipa
Eº’ = (Epc + Epa) / 2
for diffusion-controlled reax :
ip α ν1/2
a: anodic 氧化的c: cathodic 還原的