Discussion Amines
17
Discussion Amines
description
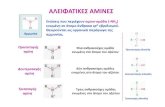
Discussion Amines. diisopropylamine. pyrrole. pyrrolidine. histamine. nicotine. Which is the strongest base?. Which is the weakest base?. lysine. Which is the most basic amino group in lysine? Why?. Which structure best represents lysine in living cells (physiological conditions). - PowerPoint PPT Presentation
Transcript of Discussion Amines

Discussion Amines

diisopropylamine
nicotine histamine pyrrolidine
pyrrole

Which is the strongest base?
Which is the weakest base?

lysine
Which is the most basic amino group in lysine? Why?
Which structure best represents lysine in living cells (physiological conditions)


Acetamides


Regiochemistry: Anti-Markovnikov
Stereochemistry: syn addition to double bond


How would you synthesis these starting materials?

Diazonium chemistry
& more




or
Retrosynthetic analysis

Synthesis plan

Formation of amide