Chemical Sedimentary Rocks: CARBONATES · 2018-04-26 · • Carbonate rocks are chemical...
Transcript of Chemical Sedimentary Rocks: CARBONATES · 2018-04-26 · • Carbonate rocks are chemical...
WhatareCarbonates?
• Carbonaterocksarechemicalsedimentaryrockscomposedmainlyoronlybycarbonateminerals
• CarbonatemineralsincludetheionCO32-intheir
composition
• Thereareseveralcarbonatemineralsbutonlytwoarereallycommon:– Calcite– Dolomite
CarbonateRocks
• Carbonaterocksincludeallrocksformedbycarbonateminerals
• Byfar,themostcommoncarbonaterocksare:– LimestoneCaCO3
• Mostlycalcite• Sometimesaragonite
– Dolostone(CaMg)[CO3]2• Dolomite
Calcitevs.Aragonite• Magnesiumanditsroleintheformationofcalcite
• Calcitevs.aragonite– Polymorphs!– Whydosomeorganismspreferaragonitetocalcite?
– Whatistheroleofplatetectonicsintheestablishmentof“calciteseas”vs.aragoniteseas”?
SchematiccrystalstructureoftheCaCO3polymorphsaragoniteandcalcite
From:https://www.researchgate.net/figure/Schematic-crystal-structure-of-the-CaCO-3-polymorphs-aragonite-and-calcite-Aragonite-a_fig1_305654328
Calcitevs.Dolomite
• Calcitevs.dolomite– Wheredoesmagnesiumgo?
• Relativeabundanceoflimestonesanddolostones– Today:dolostonesarerare– Geologicalrecord:dolostonesalmostascommonaslimestonesinshallow-waterdeposits
3-DstructureofcalciteanddolomiteBoth crystals consistof alternating sheetsof carbonate ionsbut incalcite carbonate sheets can only alternate with calcium ions. Incalcite, magnesium can disturb the calcium sheets, originatingimpurities. In dolomite instead, magnesium forms its ownseparated,alternatingsheets.
Modifiedfromhttp://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-50532013000200018
CarbonatechangesthroughGeologicTime
• Mostdolomiteisnotaprimaryprecipitatebutformsasaslowalterationoforiginalcalcite
• Oceanconditionsthatfavordolomitereplacementforcalciteare;– Highsalinity– HighpH– LowCa2+/Mg2+ratio– Hightemperature
Relativevariationofdolomite(andotherparameters)overgeologictime
TheDolomitesofItaly
TreCime(ThreePeaks)diLavaredoDolomitiregionofBellunoandBolzano,Italy
Dolomites as rocks were first investigated and described in northern Italy by theFrenchgeologistDeodatdeDolomieu.Themineral(dolomite),therock(dolostone),andthiswholegeologicalregion(Dolomiti)arenamedafterhim.
©AlessandroGrippo
Calcite• Calciteisthemostcommoncarbonatemineralinnature• Calcitecanformeitherinorganicallyorbecauseofthebiological
actionoforganisms
• Equilibriumreactionofcalciteinwater: CaCO3+H2O+CO2!"Ca2++2HCO3
-
• AddingCO2tosolutiondissolvescalciteintoitscomponentions• RemovingCO2fromsolutionwillcauseprecipitationofcalcite
• HighpressureandlowtemperaturekeepCO2insolution• LowpressureandhightemperatureremoveCO2fromsolution
TypesofLimestones
• Inorganic– formdirectlyinwaterbecauseofchangingchemicalequilibriuminsolution• Travertine,Tufa,OoliticLimestone
• Biochemical/Bioclastic– formbecauseofthebiologicalactivityofanorganism(biochemical)andsuccessivemechanicalweatheringofremains(bioclastic)• CoralReefsandStromatolites,FossiliferousLimestone,Coquina,Chalk,Micrite
Travertine
• TravertineiscalciumcarbonatethatformsdirectlyinthephysicalenvironmentuponlossofCO2fromwater
• Terrestrialenvironments:waterfall,springs,hotsprings,cavesandcaverns
Above:fossilleafinItaliantravertine,GettyCenter,LosAngeles,CABelow:bandedtravertine
SMCrockcollection,SantaMonica,CaBothimages©AlessandroGrippo
Travertineactivelyforming(andimmediatelycoveredbymoss)ataspring,creatingitsownwaterfall.Carbondioxideleavesthesolutionuponwaterleavingacloseenvironment(groundwater)andsprayingatthewaterfall
AnevaRiver,LabantediCastelD’Aiano,Bologna,Italy
Uponcomingtothesurface,hotwatercanquicklylosecarbondioxide,favoringtheprecipitationofcalciteTravertineterraces,YellowstoneNationalPark,Wyoming,USA
©AlessandroGrippo
Waterdrippingfromtheceilingofacavequicklylosescarbondioxidetotheatmosphere,favoringtheformationoftravertine.Inthiscase“dripstones”areexpressedasstalactiteshangingfromabove
MitchellCaverns,MojaveNationalPreserve,Kelso,California,USA
©AlessandroGrippo
Tufa
• TufaisavarietyoftravertinethatformsinafewplayalakesoftheAmericanWest,wheredifferentwaterchemistriesmix
• Apresentday-exampleisMonoLake
• AnextinctsystemisatSearlesLake(Trona)
Above:MonoLake,LeeViningCaliforniaBelow:Tronapinnacles,atdrySearlesLake,Tron,California
©AlessandroGrippo
Oolites(ooliticlimestones)
• Inorganiclimestoneformedbythecementationofsand-sizedooids
• Ooidsaresmallspheresofcalciteprecipitatedinwarm,shallowseawater
• Tidalcurrentsandsea-wavesmovegrains,particles,smallfossils,thatcoatthemselvesincalciumcarbonateupongrowingonatropicalcarbonateshelf
Whiteoolite
SMCrockcollection
©AlessandroGrippo
Blackoolite
SMCrockcollection
©AlessandroGrippo
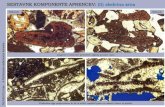
Thinsectionofanoolite,
showingbothconcentricandradialgrowth
BiochemicalandBioclasticLimestones
• Biochemicallimestonesarethoseproducedbytheactivityoforganisms– Mostlymarineorganisms– Alsofreshwaterorganisms
• Bioclasticlimestonesarethoseformedbythecementationofbrokenshells,piecesofcoralreef,etc.