Chapter4. Perodic Table
-
Upload
novah-guruloo -
Category
Documents
-
view
226 -
download
0
Transcript of Chapter4. Perodic Table
-
8/6/2019 Chapter4. Perodic Table
1/79
Chapter 4Periodic Table of Elements
-
8/6/2019 Chapter4. Perodic Table
2/79
Learning Objectives
Analyse the Periodic Table of ElementsAnalyse Group 18 elements
Analyse Group 1 elementsAnalyse Group 17 elementsAnalyse elements in a period
Understand transition elements
-
8/6/2019 Chapter4. Perodic Table
3/79
A. The Periodic Table of Elements
Look at these pictures carefully.
-
8/6/2019 Chapter4. Perodic Table
4/79
A. The Periodic Table of Elements
Why the items in the supermarket or hypermarket are arranged systematically andorderly?
-
8/6/2019 Chapter4. Perodic Table
5/79
A. The Periodic Table of Elements
Similarly, elements are arranged systematically and orderly.
-
8/6/2019 Chapter4. Perodic Table
6/79
Historical development of the Periodic Table
Antoine Lavoisier Classify substances into
metals and non-metalsUnsuccessful because
light, heat and some other compounds where notelements.
(1743-1794)
-
8/6/2019 Chapter4. Perodic Table
7/79
Historical development of the Periodic Table
Johann Dobereiner Introduced triads.Elements were classifiedinto groups of three
elements with samechemical propertiesThe atomic mass of middleelements was approximately
the average atomic mass of the other two elements
(1780-1849)
-
8/6/2019 Chapter4. Perodic Table
8/79
-
8/6/2019 Chapter4. Perodic Table
9/79
Historical development of the Periodic Table
John NewlandsArranged elements in order of increasing atomic mass.
Elements with similar propertiesrecurred at every eight element.This was known as the Law of OctavesFailed because only obeyed by first 17
elements only
(1837-1898)
-
8/6/2019 Chapter4. Perodic Table
10/79
Historical development of the Periodic Table
Lothar Meyer Plotted a graph of the atomicvolume against atomic mass.Elements with similar chemicalproperties occupied samepositions.Successful in showing theproperties of elements formed a
periodic pattern against their atomic masses.
(1830-1895)
-
8/6/2019 Chapter4. Perodic Table
11/79
-
8/6/2019 Chapter4. Perodic Table
12/79
Historical development of the Periodic Table
Dmitri MendeleevArranged the elements in order of increasing atomic mass.Successful because
--he left gaps for elements yet to bediscovered. He used the table to predictthe existence and properties of undiscovered elements
--he change the order of elements of theproperties are not similar.
(1834-1907)
-
8/6/2019 Chapter4. Perodic Table
13/79
http://en.wikipedia.org/wiki/File:Mendelejevs_periodiska_system_1871.png -
8/6/2019 Chapter4. Perodic Table
14/79
Historical development of the PeriodicTable
Henry J. G. Moseley (1887-1915)
Concluded that proton number shouldbe the bases for the periodic change of chemical propertiesArranged the elements in order of increasing proton number in thePeriodic Table.
http://www.google.com.my/imgres?imgurl=http://www.glogster.com/media/4/21/1/8/21010805.jpg&imgrefurl=http://www.vegetariansplitpeasoup.com/bg-Moseley-Periodic-Table/&usg=__IljwSG2UGtnwJXH0pqICegtAdM4=&h=393&w=255&sz=15&hl=en&start=5&zoom=1&itbs=1&tbnid=57dfAVAuYuSLfM:&tbnh=124&tbnw=80&prev=/images%3Fq%3DHenry%2BG.%2BJ.%2BMoseley%26hl%3Den%26sa%3DG%26rlz%3D1T4ADSA_enMY385%26biw%3D1276%26bih%3D582%26tbm%3Disch&ei=V_C2Td-GH8vOrQest_jFDQ -
8/6/2019 Chapter4. Perodic Table
15/79
Modern Periodic Table of Elements
Elements are arranged according their increasingproton number.Vertical columns = groups (according to their number of valence electron/sHorizontal rows = periods (number of electronshells filled by electrons)
-
8/6/2019 Chapter4. Perodic Table
16/79
Modern Periodic Table of Elements
-
8/6/2019 Chapter4. Perodic Table
17/79
Group 18/VIII Elements
Known as noble gases/ inert gases(chemically unreactive elements)Non-metals that exist as monoatomic colourless gases.
Members :Helium (He), Xenon (Xe),Neon (Ne), Radon (Ra).
Argon (Ar),Krypton (Kr),
-
8/6/2019 Chapter4. Perodic Table
18/79
Physical Properties of Group 18
Very small atomic sizes.Low melting and boiling points
Weak Van der Waals forces of attraction between atoms.
Low densitiesVery small masses but huge volumes.
Melting and boiling points of elements increasedown the Group 18.
-
8/6/2019 Chapter4. Perodic Table
19/79
-
8/6/2019 Chapter4. Perodic Table
20/79
-
8/6/2019 Chapter4. Perodic Table
21/79
Uses of Group 18Elements
HeliumTo fill airships and weather balloons.
Airships
-
8/6/2019 Chapter4. Perodic Table
22/79
Uses of Group 18Elements
Airships
-
8/6/2019 Chapter4. Perodic Table
23/79
Uses of Group 18Elements
Airships
-
8/6/2019 Chapter4. Perodic Table
24/79
Uses of Group 18 Elements
Weather balloon
-
8/6/2019 Chapter4. Perodic Table
25/79
Uses of Group 18 Elements
Weather
balloon
-
8/6/2019 Chapter4. Perodic Table
26/79
Uses of Group 18 Elements
Heliumused as artificial atmosphere in oxygen tankfor divers.
Oxygentank
-
8/6/2019 Chapter4. Perodic Table
27/79
Uses of Group 18 Elements
Oxygentank
U f 18
-
8/6/2019 Chapter4. Perodic Table
28/79
Uses of roup 18Elements
Oxygentank
-
8/6/2019 Chapter4. Perodic Table
29/79
Uses of Group 18 ElementsNeon
Advertising lights
U f G 18
-
8/6/2019 Chapter4. Perodic Table
30/79
Uses of Group 18Elements
NeonUsed in aeroplane runway lights
-
8/6/2019 Chapter4. Perodic Table
31/79
-
8/6/2019 Chapter4. Perodic Table
32/79
Uses of Group 18 Elements
KryptonUsed in lasers to repair the retina of the eye.
-
8/6/2019 Chapter4. Perodic Table
33/79
Uses of Group 18 Elements
KryptonTo fill photographic flash lamps.
-
8/6/2019 Chapter4. Perodic Table
34/79
Uses of Group 18 Elements
XenonMaking electron tubes and stroboscopic lamps
-
8/6/2019 Chapter4. Perodic Table
35/79
Uses of Group 18 Elements
XenonUsed in bubble chambers in atomic energyreactors.
U f G 18
-
8/6/2019 Chapter4. Perodic Table
36/79
Uses of Group 18Elements
RadonUsed to treat cancer
-
8/6/2019 Chapter4. Perodic Table
37/79
Group 1 ElementsConsists of lithium (Li), sodium (Na), potassium
(K), rubidium (Rb), caesium (Cs) and francium(Fr).They are known as alkali metals because theyreact with water to produce alkali ne solution.
-
8/6/2019 Chapter4. Perodic Table
38/79
Physical Properties of Group 1 Elements
SoftLow melting pointsLow densitiesShiny and silvery surfaceGood conductor of heatGood conductor of electricity
-
8/6/2019 Chapter4. Perodic Table
39/79
Physical Properties of Group 1 Elements
Hardness, melting point andboiling of the elements decrease .
-
8/6/2019 Chapter4. Perodic Table
40/79
Physical Properties of Group 1 Elements
Hardness, melting point and boiling of the elementsdecrease.
Metallic bonding in metals is caused by the attractionbetween positive ions and the sea of negative electrons
surrounding them.
-
8/6/2019 Chapter4. Perodic Table
41/79
Physical Properties of Group 1 Elements
When go down Group 1, atom becomes larger. The positive nucleus gets
further away from the negative sea of electrons.
The force of attraction between the metal ionsand the sea of electrons gets weaker down thegroup.
Less energy is needed to overcome thisweakening force of attraction.
-
8/6/2019 Chapter4. Perodic Table
42/79
Chemical Properties of Group 1Elements
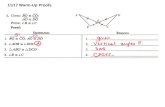
All react with water to produce alkaline metalhydroxide solution and hydrogen gas .2X(s) + 2H 2O(l) 2XOH(aq) + H 2(g)
How to test the alkaline solution?1 542 3
-
8/6/2019 Chapter4. Perodic Table
43/79
Group 1 metals + Wate
http://brainiac%20alkali%20metals.flv/http://brainiac%20alkali%20metals.flv/ -
8/6/2019 Chapter4. Perodic Table
44/79
Chemical Properties of Group 1Elements
All burn in oxygen gas to produce white solidmetal oxides .4X(s) + O 2(g) 2X 2O(s)
The oxide dissolve in water to form alkaline metalhydroxide solution .X2O(s) + H 2O(l) 2XOH(aq)
-
8/6/2019 Chapter4. Perodic Table
45/79
Chemical Properties of Group 1Elements
All burn in chlorine gas to produce white solidmetal chlorides .
2X(s) + Cl 2(g) 2XCl(s)
Alkali metal + water
-
8/6/2019 Chapter4. Perodic Table
46/79
Alkali metal + water Alkali metal Observation Equation
Lithium (Li) Lithium moves slowly on the water surface with a soft hiss sound. Acolorless solution that turns red litmuspaper blue is formed
Sodium (Na) Sodium melts to become a smallsphere , moves rapidly and randomly onthe water surface with a soft hiss sound. A colorless solution that turnsred litmus paper blue is formed
Potassium (K) Potassium melts to become a smallsphere , burns with a lilac flame , very rapidly and randomly on the water surface with a soft hiss and pop sound. A colorless solution that turnsred litmus paper blue is formed
Alkali metals + Oxygen gas
-
8/6/2019 Chapter4. Perodic Table
47/79
Alkali metals + Oxygen gasAlkali metal Observation Equation
Lithium (Li) Lithium burns slowly with a red flame andliberates white fumes which become a whitesolid on the cooling to room temperature. Thewhite solid dissolves in the water to produce acolorless solution . This solution turns redlitmus paper blue
Sodium (Na) Sodium burns rapidly and brightly with ayellow flame and liberates white fumes whichbecome a white solid on the cooling to roomtemperature. The white solid dissolves in thewater to produce a colorless solution . Thissolution turns red litmus paper blue
Potassium(K)
Potassium burns very rapidly and brightly witha lilac flame and liberates white fumes whichbecome a white solid on the cooling to roomtemperature. The white solid dissolves in thewater to produce a colorless solution . This
solution turns red litmus paper blue
-
8/6/2019 Chapter4. Perodic Table
48/79
Alkali metals + Chlorine gasAlkali metal Observation Equation
Lithium (Li) Lithium burns slowly with a red flame andliberates white fumes which become a whitesolid at the end of the reaction.
Sodium (Na) Sodium burns rapidly and brightly with ayellow flame and liberates white fumes whichbecome a white solid at the end of thereaction
Potassium(K)
Potassium burns very rapidly and brightly witha lilac flame and liberates white fumes whichbecome a white solid at the end of thereaction
-
8/6/2019 Chapter4. Perodic Table
49/79
Chemical Properties of Group 1Elements
Why all elements in Group 1 have samechemical properties?
Chemical reaction is all about the activity of electron/sAll the elements have one valence electron .Each of them reacts by donating one valenceelectron to form an ion with a charge of +1 toachieve stable electron arrangement.
-
8/6/2019 Chapter4. Perodic Table
50/79
Reactivity of Group 1 Elements
Chemical Properties of Group 1
-
8/6/2019 Chapter4. Perodic Table
51/79
Chemical Properties of Group 1Elements
Why the reactivity of elements increases down the Group 1?Atomic size of Group 1 elements increases from lithium tofrancium./ Number of shells occupied by electrons increases .Distance between the valence electron in the outermost shell andpositive nucleus increases down the Group 1.Attraction between nucleus and valence electron decreases .
It is easier for the atom to lose the valence electron to achieve stableelectron arrangement.
Group 17
-
8/6/2019 Chapter4. Perodic Table
52/79
Group 17Elements
Members are fluorine(F 2) , chlorine(Cl 2),bromine(Br 2), iodine(I 2), and astatine(At 2)
The elements are also known as halogens which exist as diatomic molecules .
Physical Properties of Group
-
8/6/2019 Chapter4. Perodic Table
53/79
Physical Properties of Group17
They have low melting and boiling pointsbecause molecules are attracted to each other by weak van der Waals forces of attraction.
The melting and boiling points of the elementsincreases down Group 17.This change the states of elements from gas to
solid and the colour of elements from lighter colour to darker colour.
-
8/6/2019 Chapter4. Perodic Table
54/79
Physical Properties of Group 17
Elements State Colour
Fluorine Gas Pale yellow
Chlorine Gas Greenish-yellowBromine Liquid Reddish-brown
Iodine Solid Purplish-black
-
8/6/2019 Chapter4. Perodic Table
55/79
Physical Properties of Group 17
Why the melting and boiling points of elementsincreases down Group 17?
Molecular size/relative molecular mass of the
elements increases down Group 17.Forces of attraction betweenmolecules/Intermolecular forces of attractionincreases .
More heat is needed to overcome the stronger forcesof attraction between the molecules.
-
8/6/2019 Chapter4. Perodic Table
56/79
Chemical Properties of Group 17
All members have similar chemical properties but differ inthe reactivity.
React with water to form two acids
X2(g) + H
2O(l) HX(aq) + HOX(aq)
Example:
Cl2(g) + H 2O(l) HCl(aq) + HOCl(aq)
hydrochloric hypochlorousacid acid
Hypochlorous acid is a bleaching agent (bleach both blue and redlitmus paper)
-
8/6/2019 Chapter4. Perodic Table
57/79
Chemical Properties of Group 17
All members have similar chemical properties but differ inthe reactivity.
Halogens in gaseous state react with hot iron to form brownsolid.2Fe(s) + 3X 2(g) 2FeX 3(s)
Example:
2Fe(s) + 3Cl 2(g) 2FeCl 3(s)
solid iron(III) chloride(brown)
Chemical Properties
-
8/6/2019 Chapter4. Perodic Table
58/79
Chemical Propertiesof Group 17
All members have similar chemical properties but differ in the reactivity.
Halogens react with sodium hydroxide solution to producesodium halide, sodium halate(I) and water X2 + 2NaOH(aq) NaX(aq) + NaOX(aq) + H 2O(l)
Example:
Cl2 + 2NaOH(aq) NaCl(aq) + NaOCl(aq) + H 2O(l)
Sodium chlorate(I)
Chemical Properties
-
8/6/2019 Chapter4. Perodic Table
59/79
Chemical Propertiesof Group 17
Why all halogens possess similar chemicalproperties?Chemical reaction = lose or accept electron/sAll halogens always gain one electron to achievestable octet electron arrangement.Therefore, they have similar chemical properties.
Chemical Properties
-
8/6/2019 Chapter4. Perodic Table
60/79
Chemical Propertiesof Group 17
Why chemical reactivity of halogens decreasesdown Group 17?Atomic size/number of electron occupied shells of halogens increases down Group 17.The outermost shell becomes further from thenucleus of the atom.
Strength to attract one electron into the outermostshell by the nucleus becomes weaker.Reactivity decreases.
-
8/6/2019 Chapter4. Perodic Table
61/79
PeriodElements across a period exhibit a periodic changein properties.Period 3Proton number increases by one unit from oneelement to the next element
-
8/6/2019 Chapter4. Perodic Table
62/79
PeriodAll the atoms of the elements have three shellsoccupied with electronsThe number of valence electrons in each atomincrease from 1 to 8
-
8/6/2019 Chapter4. Perodic Table
63/79
PeriodAll the elements exist as solid except chlorine andargon which are gasesThe atomic radius of elements decreases. This isdue to the increasing nuclei attraction on thevalence electrons.
-
8/6/2019 Chapter4. Perodic Table
64/79
PeriodThe electronegativity of elements increases. Thisis also due to the increasing nuclei attraction onthe valence electrons and the decreases in atomic
size.
-
8/6/2019 Chapter4. Perodic Table
65/79
Period
Metals Metalloid Non-metalsMetalloid semi-metal, reacts with acid only, weakconductor, brittle and not malleable and ductile.
-
8/6/2019 Chapter4. Perodic Table
66/79
PeriodUses of metalloid
Make diodes and transistorsA diode A transistor
Both are commonly used in the making of microchipsMicrochips are widely used in the manufacture of computers,mobile phones, televisions, video recorders, calculators, radio andetc.
-
8/6/2019 Chapter4. Perodic Table
67/79
PeriodOxides of elements change from basic to amphoteric andthen to acidic across the period towards the right.
Basic oxides react with acids to form salt and water Acidic oxides react with alkalis to form salt and water Amphoteric oxides react with both acids and alkalis toform salt and water.
Transition
-
8/6/2019 Chapter4. Perodic Table
68/79
TransitionElements
Elements from Group 3 to Group 12 in the PeriodicTable.Common characteristics
Solid metal with shiny surface.Good conductor of heat and electricity.High melting and boiling points.
Hard, malleable and ductile.
Transition
-
8/6/2019 Chapter4. Perodic Table
69/79
TransitionElements
Special characteristicsAll are metalsShow different oxidation numbers in their compoundsForm coloured ions or compoundsUse as catalysts
Form complex ions
Transition
-
8/6/2019 Chapter4. Perodic Table
70/79
TransitionElements
Show different oxidation numbers in their compound
Compound Formula Oxidation number
Chromium(III) chloride Cr Cl3
+3
Potassium dichromate(VII) K 2Cr 2O 7 +6
Manganese(II) sulphate MnSO 4 +2
Manganese(VI) oxide MnO 2 +4
Potassium manganate(VII) K MnO 4 +7
Iron(II) sulphate Fe SO 4 +2
Iron(III) chloride Fe Cl3 +3
Copper(I) oxide Cu 2O +1
Copper(II) sulphate Cu SO 4 +2
Transition
-
8/6/2019 Chapter4. Perodic Table
71/79
TransitionElements
Form coloured ions or compoundsElement Ion Colour
Chromium Cr 3+ Green
CrO 42- Yellow
Cr 2O 72- Orange
Manganese Mn 2+ Pale pink
MnO 4- Purple
Iron Fe 2+ Pale green
Fe 3+ Yellowish brown
Cobalt Co 2+ Pink
Nickel Ni 2+ Green
Copper Cu 2+ Blue Green
Transition
-
8/6/2019 Chapter4. Perodic Table
72/79
TransitionElements
Form coloured ions or compoundsGemstone Transition metal Colour
Emerald Ni and Fe Green
Amethyst Fe and Mn Purple
Sapphire Co and Ti Blue
Ruby Cr Red
Topaz Fe Yellow
Transition
-
8/6/2019 Chapter4. Perodic Table
73/79
TransitionElements
Emerald
Transition
-
8/6/2019 Chapter4. Perodic Table
74/79
TransitionElements
Amethyst
Transition
-
8/6/2019 Chapter4. Perodic Table
75/79
TransitionElements
Sapphire
Transition
-
8/6/2019 Chapter4. Perodic Table
76/79
TransitionElements
Ruby
Transition
-
8/6/2019 Chapter4. Perodic Table
77/79
TransitionElements
Topaz
Transition
-
8/6/2019 Chapter4. Perodic Table
78/79
TransitionElements
As catalyst
Process Catalyst Tomanufacture
Haber Process Iron fillings, Fe Ammonia
ContactProcess
Vanadium(V)oxide, V 2O 5
Sulphuric acid
OstwaldProcess
Platinum, Pt Nitric acid
Hydrogenation Nickel, Ni Margarine
Transition
-
8/6/2019 Chapter4. Perodic Table
79/79
TransitionElements
To form complex ions
Element Complex ions Formula
Iron Hexacyanoferrate(II) ion [Fe(CN)6]4-
Hexacyanoferrate(III) ion [Fe(CN) 6]3-
Chromium Hexaamina chromium(III) ion [Cr(NH 3)6]3+
Copper Tetraamina copper(II) ion [Cu(NH 3)4]2+
Tetrachlorocuprate(II) ion [CuCl 4]2-