Chapter 8 Amines Chemistry 20. Amines: Are derivatives of ammonia NH 3. Contain N attached to one or...
-
Upload
marsha-perry -
Category
Documents
-
view
256 -
download
0
Transcript of Chapter 8 Amines Chemistry 20. Amines: Are derivatives of ammonia NH 3. Contain N attached to one or...

Chapter 8
Amines
Chemistry 20

Amines:
• Are derivatives of ammonia NH3.
• Contain N attached to one or more alkyl (Aliphatic amine) or aromatic groups (Aromatic amine).
CH3-NH2 CH3-NH-CH3
Amines
-NH2 amino group
CH2CH3 CH3 CH=CH2
TolueneEthylbenzene Styrene
NH2

Amines are classified into three groups:
depending on the number of carbon groups bonded to nitrogen.
CH3 CH3
CH3—NH2 CH3—NH CH3—N—CH3
Amines
Primary 1° Secondary 2° Tertiary 3°

Naming Amines
The same method as we did for alcohols.
- Drop the final “-e” of the parent alkane and replace it by “-amine”.
- Use a number to locate the amino group (-NH2) on the parent chain.
IUPAC name – 1° amines
CH3CHCH3
NH2
NH2
H2N NH2
1,6-HexanediamineCyclohexanamine2-Propanamine
CH3-CH-CH3
NH2
2-propanamine
CH3-CH-CH-CH3
NH2
Cl
3-chloro-2-butanamine 1,6-hexanediamine
123 1 2
3
4
1234
56

Common name
Names of alkyl groups (In alphabetical order) + “amine”
CH3—CH2—NH2 ethylamine
CH3—NH —CH3 dimethylamine
CH3
|
CH3—N—CH2—CH3 ethyldimethylamine
Naming Amines

CH2CH3 CH3 CH=CH2
TolueneEthylbenzene Styrene
CH2CH3 CH3 CH=CH2
TolueneEthylbenzene Styrene
CH2CH3 CH3 CH=CH2
TolueneEthylbenzene Styrene
Aniline (common name)
NO2
CH3 CH3
Aniline
4-Nitroaniline 3-Methylaniline 4-Chloro-3-methylaniline
NH2
NH2NH2 NH2
Cl
1 2
3
4
1 2
3
1 2
34
CH2CH3 CH3 CH=CH2
TolueneEthylbenzene Styrene

Naming Amines
– Take the largest group bonded to nitrogen as the parent amine.
– Name the smaller group(s) bonded to nitrogen, and show their locations on nitrogen by using the prefix “N”.
IUPAC name – 2° and 3° amines
NCH3
CH3
NHCH3
N,N-Dimethyl- cyclopentanamine
N-Methylaniline
NCH3
CH3
NHCH3
N,N-Dimethyl- cyclopentanamine
N-Methylanilineaniline
CH3-N-CH2-CH3
CH3
N,N-Dimethylethanamine

Heterocyclic amines
When N is one of the atoms of a ring.
N
HHCH3
Pyrrolidine Pyridine Nicotine

Physical properties of Amines
1. They have unpleasant odors (rotting fish like ammonia).
2. They are polar compounds.
Difference in electronegativity between N - H (3.0 – 2.1 = 0.9)
3. 1° and 2° amines have hydrogen bonds (N-H).
Weaker than alcohols (O-H).
3° amines do not form hydrogen bonds (no H atom).
4. Boiling points: Hydrocarbons< Amines < Alcohols
5. Almost soluble in water (hydrogen bonding).

Chemical properties of Amines
They are weak bases (like ammonia): react with acids.
N
H
H
CH3 .. + H – O – H....N
H
H
CH3 H+
O – H......
-
Some amines present in our blood and make it approximately basic (pH = 7.4).
(to form water-soluble salts)

Chemical properties of Amines
C6H5NH2
CH3CH2NH2Aliphatic
Ammonia
Aromatic
3.0 - 4.0
4.74
8.5 - 9.5
Class pKb Example Name
Ethanamine
Aniline
Stronger base
Weaker base
Aliphatic amines are weak bases by comparison with inorganic bases
such as NaOH, they are strong bases among organic compounds.
Aliphatic amines are stronger bases than aromatic amines.
(slightly stronger than NH3)

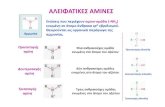
Complete each acid-base reaction and name the salt formed.
(CH3CH2)2NH HCl
NCH3COOH
+(a)
(b) +
Examples

Complete each acid-base reaction and name the salt formed.
Examples
Solutions:
(CH3CH2)2NH HCl
NCH3COOH
NH
(CH3CH2)2NH2+Cl-
CH3COO-
Diethylammoniumchloride
+Pyridinium acetate
+(a)
(b) +




![ALIPHATIC & AROMATIC AMINES - jeemain.guru · (a) From Ammonolysis of alkyl halides [Hofmann's ammonolysis] : When an aqueous solution of ammonia is heated with alkyl halide all the](https://static.fdocuments.net/doc/165x107/5e0d42079bbb2e69d86e5db0/aliphatic-aromatic-amines-a-from-ammonolysis-of-alkyl-halides-hofmanns.jpg)