CHAPTER 3B Stoichiometry. Counting Atoms Amu – atomic mass unit 1 amu = 1.66 x 10 -24 g Ex. 1...
-
Upload
eustace-stafford -
Category
Documents
-
view
243 -
download
0
Transcript of CHAPTER 3B Stoichiometry. Counting Atoms Amu – atomic mass unit 1 amu = 1.66 x 10 -24 g Ex. 1...
Counting Atoms
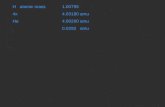
Amu – atomic mass unit1 amu = 1.66 x 10-24 g
Ex. 1 carbon atom = 12 amu 1 Fe atom = 55.85 amu1 Na atom = ??? Amu
Counting Atoms
Calculate the mass, in amu, of a sample of Fe that contains 15 atoms.
Calculate the mass, in amu, of a sample of carbon that contains 62 atoms.
Counting Atoms
Calculate the number of copper atoms present in a sample that has a mass of 1779.4 amu.
1 Cu atom = ??? Amu
Calculate the number of argon atoms present in a sample that has a mass of 3755.3 amu.
Moles
An amount of matter that contains as many objects as the number of atoms in exactly 12 grams of pure carbon 12. This number is 6.02 x 1023.
Avogadro's number = 6.02 x 1023
Moles
1 dozen eggs = 12 eggs
1 mole eggs = 6.02 x 1023 eggs
1 mole of elephants = 6.02 x 1023
elephants
1 mole of Al = 6.02 x 1023 Al atoms
Moles
But they all have different masses.
1 mol of C and 1 mol of Mg have the same number of particles
but C has a mass of 12 amu and Mg a mass of 24 amu
If C has a mass of 12 g by definition then Mg = 24 grams.
So….. 1 mole of the element = 6.02 x 1023 atoms of that
element and…1 amu of an element = grams of 1 mole of
element
Element # of atoms Mass (g) MolesAl 6.02 x 1023 26.98 1 moleAu 6.02 x 1023 196.97 1 moleS 6.02 x 1023 32.07 1 moleFe 6.02 x 1023 55.85 1 mole
Mass/Mole
1 mol of an atom= average atomic mass of the element
Ex. 1mol H = 1.008 g H
1mol C = 12.01 g C
1mol Na = 22.99 g Na
Can then use this as a conversion factor
Mass/Mole
Calculate the number of moles of atoms in 57.7 g sample of sulfur.
Calculate the number of moles in 43.15 g of carbon.
Calculate the mass in grams of 0.251 moles of sodium.
Calculate the mass in grams of 5.58 moles of copper.
Molar Mass
The total mass of a compound.
What is the mass of H20?
1 atom H2O = 1 atom O and 2 atoms H
So…1 mol H20 = 1 mol O and 2 mols H
Mass of 1 mole of O = 1 x 16.00g = 16.00gMass of 2 moles of H = 2 x 1.008g = 2.016gMass of 1 mole of H2O = 18.12g
Molar Mass
What is the mass in grams of 1.48 moles of potassium oxide?
Calculate the mass of 4.85 mol of acetic acid, HC2H3O2.
How many moles of formaldehyde (H2CO) does a 7.55 g sample represent?
How many moles of tetraphosphorus dioxide does a 250.0 gram sample represent?
Percent Mass Composition
Determining the percentage of how much of the total weight each element weighs.
Ex. Determine the mass percent of each element in sulfuric acid (H2SO4).
% H =% S =% O =
Formulas
Empirical formula – a formula of a compound that has the smallest whole-number ration of atoms present.
Molecular formula – the actual formula of a compound.
Ex. Determine the empirical formula for C4H10
H2O2
CCl4
Calculating Empirical Formulas1. Find mass of each element2. Number of moles of each element3. Divide each elements moles by the
smallest number of moles.4. If the moles are not whole numbers,
multiply each element by the number that converts the fraction to whole numbers
Calculating Empirical FormulasEx. A 1.500 gram sample of a compound
containing only carbon and hydrogen is found to contain 1.198 gram of carbon. Determine the empirical formula for this compound.
Calculating Empirical FormulasEx. A sample of phosphoric acid contains
0.3086 grams of hydrogen, 3.161 grams of phosphorus, and 6.531 grams of oxygen. Determine the empirical formula for phosphoric acid.
Calculating empirical Formulas
Ex. The simplest amino acid, glycine, has the following mass percents: 32.00% carbon, 6.714% hydrogen, 42.63% oxygen, and 18.66% nitrogen. Determine the empirical formula for glycine.
Mole Ratios
A balanced equation of a reaction gives a ratio of moles of each compound involved in the reaction.
Remember a mole has no dimension until you add one.
Ex. NO + H2 → N2 + H2O
Ex. SiH4 + NH3 → Si3N4 + H2
Mole Ratios
Methane burns in oxygen to form carbon dioxide and water.
CH4 + 2O2 → CO2 + 2H2O
What number of moles of oxygen gas is required to react with 7.4 moles of methane?
How many moles of carbon dioxide will be produced by reacting 2.6 moles of oxygen with excess methane?
Mole Ratios
Hydrogen sulfide gas reacts with oxygen to produce sulfur dioxide gas and water.
How many moles of oxygen are required to react with 5.6 moles of hydrogen sulfide?
How many moles of sulfur dioxide gas will be produced by reacting 7.3 moles of hydrogen sulfide with excess oxygen?
How many moles of sulfur dioxide gas will be produced by reacting 7.3 moles of oxygen with excess hydrogen sulfide?
Mole-Mass Conversion
Solutions of sodium hydroxide absorb carbon dioxide from the air, forming sodium carbonate and water.
Calculate the mass of carbon dioxide gas that is required to react with a solution containing 10.0 grams of sodium hydroxide.
Calculate the mass of sodium carbonate that is produced when 10.0 grams of sodium hydroxide reacts with an excess of carbon dioxide.
Mass/Mole
Steps for calculating
Balance equation
Convert mass of known compound to moles.
Use mole ratios from balanced equation as a
conversion to moles of desired compound.
Convert moles of desired compound to mass
Limiting Reactants
A reactant that stops the reaction because it is consumed.
Steps to calculating limiting reactants Balance the equation. Convert to moles. Use the mole ratios to determine limiting reagent. Using limiting reactant compute the number
moles of desired product. Convert to moles.
Limiting reactants
Iron(III) oxide reacts with carbon monoxide to form iron metal and carbon dioxide gas. In a certain experiment 5.0 grams of Iron (III) oxide is reacted with 5.0 grams of carbon dioxide.
What mass of iron will be produced, assuming a complete reaction?
What mass of carbon dioxide will be produced, assuming a complete reaction?
Limiting reactants
Iodine reacts with chlorine gas to form iodine chloride. In a certain experiment, 10.0 grams of iodine is reacted with 10.0 grams of chlorine gas.
What mass of aluminum chloride will be produced, assuming a complete reaction?
Percent yield
In a certain experiment, the expected yield is 1.325 grams. The actual yield was 1.279 grams. Determine the percent yield.
2NH3 + CO2 → CN2H4O + H2O
If 100.0 grams of ammonia is reacted with 100.0 grams of carbon dioxide and 120.0 grams of urea are produced. Determine the percent yield.
yeild100%
Theoretical yeild
actualx