Chapt. 11, Membrane Structure - SUNY Geneseolewisj/Cell.04/Lectures/Chapt11_3.pdfChapt. 11, Membrane...
Transcript of Chapt. 11, Membrane Structure - SUNY Geneseolewisj/Cell.04/Lectures/Chapt11_3.pdfChapt. 11, Membrane...

1
1
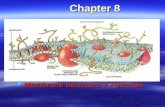
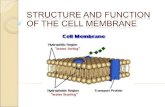
Chapt. 11, Membrane Structure
• Functions of cell membrane
2
Chapt. 11, Membrane Structure
• Functions of cell membrane– As a container/ barrier to movement of
small molecules. Figure 1-1
3
• Functions of cell membrane, cont.– Import and export of molecules.– Information transducer.– Movement. Figure 1-2.
Chapt. 11, Membrane Structure

2
4
• The lipid bilayer is composed (in animals) ofphospholipids, cholesterol and glycolipids.
• All are amphipathic.
• Structure of lipids results in self-assemblyinto a bilayer.
Chapt. 11, Membrane Structure, Lipids
5
• Phospholipid structure.
Chapt. 11, Membrane Structure, Lipids
Fig 11.6
6
• Phospholipid structure.– Phospholipids make up the bulk of the membrane
bilayer.– The four major components of a phospholipid.– Hydrophobic and hydrophilic components of a
phospholipid.– There are several types of organic headgroups.– There is usually one or more double bonds in one
of the fatty tails.
Chapt. 11, Membrane Structure, Lipids

3
7
• Phospholipids self-assemble into a 2dimensional lipid bilayer.– The importance of the hydrophobic interactions
Chapt. 11, Membrane Structure, Lipids
8
– Review of the hydrophobic interaction. Panel 2-2Chapt. 11, Membrane Structure, Lipids
9
– Review of the hydrophobic interaction. (Fig 11-9modified)
Chapt. 11, Membrane Structure, Lipids
20 high-energy watermolecules
13 high-energy watermolecules

4
10
• Phospholipids self-assemble into a 2dimensional lipid bilayer.– Review of the hydrophobic interaction.– The importance of van der Waals forces.
Chapt. 11, Membrane Structure, Lipids
11
• Phospholipids self-assemble into a 2dimensional lipid bilayer.– Review of the hydrophobic interaction. Fig 11-9– The importance of van der Waals forces.– Result: Open or closed membrane sheets (Figs.
11-12, 11-13, 11-14)
Chapt. 11, Membrane Structure, Lipids
12
Figs. 11-12, 11-13

5
13
Figs. 11-12, 11-14
14
• Lipid bilayers are said to be fluid.– What do we mean by “fluid”?– The potential types of phospholipid mobility. Fig
11-15.
Chapt. 11, Membrane Structure, Lipids
15
• Membrane phospholipid structure and degreeof fluidity.– What is “freezing” or “crystallization”?– Effect of temperature on fluidity.– Effect on length of hydrocarbon tails on fluidity.– Effect of unsaturation on fluidity.
Chapt. 11, Membrane Structure, Lipids

6
16
• Structure of Cholesterol. Fig 11-7b, 11-16.
Chapt. 11, Membrane Structure, Lipids
17
• The effect of cholesterol on membranefluidity. (Fig. 11-16)
Chapt. 11, Membrane Structure, Lipids
18
• Glycolipid structure. Fig 11-7c
Chapt. 11, Membrane Structure, Lipids

7
19
• Terminology.– “Leaflet”
Chapt. 11, Membrane Structure, Lipids
20
• Terminology.– “Leaflet”– Problems with
“inside” and “outside”– Suggested
terminology“cytosolic” and “non-cytosolic”
Chapt. 11, Membrane Structure, Lipids
Fig. 11-19
21
• The bilayer is asymmetrical.– Phospholipids - some phospholipids are mainly in
the cytosolic leaflet, others in the non-cytosolicone. Fig. 11-17
Chapt. 11, Membrane Structure, Lipids

8
22
• The bilayer is asymmetrical.– Glycolipids - found (almost) exclusively on the
non-cytosolic leaflet. Fig. 11-17
Chapt. 11, Membrane Structure, Lipids
23
• The bilayer is asymmetrical..– Cholesterol -- found on both leaflets as it easily
flips.
Chapt. 11, Membrane Structure, Lipids
24
• Amounts of proteins in membranes.• Some functions of proteins. (Table 11-1)
Chapt. 11, Membrane Structure, Proteins

9
25
• Types of membrane proteins; transmembraneFig 11-21
Chapt. 11, Membrane Structure, Proteins
26
• Types of membrane proteins; lipid linked Fig11-21.
Chapt. 11, Membrane Structure, Proteins
27
• Types of membrane proteins; peripheral(protein attached) Fig 11-21.
Chapt. 11, Membrane Structure, Proteins

10
28
• A more detailed consideration oftransmembrane proteins.– Proteins that have buried hydrophobic parts go
all the way through the bilayer; there are nohalf-transmembrane proteins.
– The portion of the protein that is in thehydrophobic interior always adopts into a alphahelix or beta sheet. Why? (Fig. 11-24 and Fig10-15 in Big Alberts.)
Chapt. 11, Membrane Structure, Proteins
29
Fig. 11-24 and Fig 10-15 in Big Alberts.
30
• Difficulties in studying membrane proteins.– Hard to physically separate protein of interest
from other proteins.– Even if you could, hydrophobic portions of the
proteins would aggregate.
• The solution: detergents. Fig 11-26, 11-27
Chapt. 11, Membrane Structure, Proteins

11
31
Fig. 11-26
32
Fig 11-26
33
• You can isolate proteins, Now what?– Sequence.– Use to raise antibodies (Panel 4-6).– SDS gel electrophoresis (Panel 4-5).
Chapt. 11, Membrane Structure, Proteins

12
34
SDS PAGE (preparing the sample)
SDS
-
--
-
-
--- -
---
---
- --
-
----
----
--
-
----
----
----
----
35
SDS PAGE (loading the gel)
--
-
-
--- -
---
--- - -
-
-
----
-----
--
----
----
----
----
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
36
SDS PAGE (after 2 hrs.)
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
-
--- -
---
--- - -
-
-
----
----
--
- --
-
----
-----
---
----

13
37
SDS PAGE (after 2 hrs.)
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
-
--- -
---
--- - -
-
-
----
----
--
- --
-
----
-----
---
----
112,000 daltons90,000 daltons
66,000 daltons
34,000 daltons
15,000 daltons
38
• Membrane proteins are polar andasymmetric.
Chapt. 11, Membrane Structure, Proteins
NH2
COOH
39
• How do we know membrane proteins are polarand asymmetric?
• Vectorial labeling– Reagent cannot penetrate membranes– Reagent can label proteins.
Chapt. 11, Membrane Structure, Proteins

14
40
Vectorial labeling
SDS gel
41
Vectorial labeling
42
Vectorial labeling
SDS gelstained for total protein
SDS gelvectoriallabel

15
43
• The complete structure is known forrelatively few proteins.– Difficulties in determining structure.– Examples of membrane proteins where the
structure is known.• Bacteriorhodopsin (Fig. 11-28).
Chapt. 11, Membrane Structure, Proteins
44
Fig. 11-28 Bacteriorhodopsin
45
• The complete structure is known forrelatively few proteins.– Difficulties in determining structure.– Examples of membrane proteins where the
structure is known.• Bacteriorhodopsin (Fig. 11-28).• Photosynthetic reaction center of Rhodopseudomonas.
(Fig. 11-29)
Chapt. 11, Membrane Structure, Proteins

16
46
Fig. 11-29
Bacterialphoto-chemicalreactioncenter
47
• The red blood cell and the plasma membrane.– Why study the R !BC?– Kinds of proteins present
• A multipass membrane protein (band 3)• A single pass membrane protein (glycophorin)• Numerous peripheral proteins including:
– Spectrin– Actin
• A summary Fig 11-31.
Chapt. 11, Membrane Structure, Proteins
48
Fig. 11-32

17
49
50
• How common are RBC proteins?
• RBC proteins and related proteins in humanhealth.– Spherocytosis– Muscular Dystrophy
Chapt. 11, Membrane Structure, Proteins
http://www.diseasedir.org.uk/genetic/genex01.htm
Dystrophin
51
• Glycoproteins as well as glycolipids havesugars on the non-cytosolic side.– Especially true for plasma membranes (where
they are present on the outside of the cell), butalso on luminal side (=inside = non-cytosolic side)of internal compartments.
– Fig 11-32
Chapt. 11, Membrane Structure, Proteins

18
52
Fig. 11-32
53
• Possible functions of sugars on the plasmamembrane.– Membrane protection and lubrication– Electrical insulation– Cell recognition
• An example: snagging neutrophils to infection sites.Fig. 11-33
Chapt. 11, Membrane Structure, Proteins
54

19
55Fazlebas & Kim, 2003, Science 299:355-356
• Another example: the embryo makes selectins that snag carbohydrates on the uterine wall.
56
• Many proteins can diffuse within the lipidbilayer.– Why does this make sense?– What kind of mobility is possible?
• No flip-flop.• Rapid spinning• Lateral diffusion
Chapt. 11, Protein mobility
57
• How can one show lateral mobility? Fig 11-34
Chapt. 11, Protein mobility

20
58
• Fluorescence Recovery after Photobleaching(FRAP): a way to measure rates of lateraldiffusion.
Chapt. 11, Protein mobility
59
• Restrictions of lateral mobility. (Fig. 11-35)
Chapt. 11, Protein mobility
60
• An example where this is very important:tight junctions in the intestinal epithelium.(Fig. 11-36)
Chapt. 11, Protein mobility