Capa Value
-
Upload
avinashonscribd -
Category
Documents
-
view
227 -
download
0
Transcript of Capa Value
-
8/12/2019 Capa Value
1/2
Compliant, Effective, Efficien
www.pathwise.co
866.580.PAT
The Value of an Effective and Efficient CAPA System
During the establishment of a CAPA System it is extremely important to ensure this critical Quality System is
both suitable and effective for your organization. Suitable means that it is appropriate foryour organization
andcompliant to the applicable regulations. Effective means that it works, as designed, and you get resultsAlso, because you run a business, your CAPA System must be efficient. You cannot waste resources and
opportunity.
The PathWise CAPA System provides the necessary process improvement to better execute the work contentrequired of running a CAPA System and executing CAPA projects and enables this work to be conducted in an
extremely efficient manner.
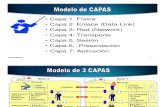
Within all work process there exist three variables.
1. Work Content--The work necessary to execute CAPA (System and Projects)2. Resources--The resources required to execute CAPA effectively3. Time-- The time it takes your resources to execute CAPA (System and Projects)
Implementing the PathWise CAPA System will enable your organization to improve your Quality ManagementSystem and establish a means to effectively repair and improve all other elements within your system. The
PathWise CAPA System establishes priorities in terms of addressing:
Issues of High Severity (Risk Management) / Recall Avoidance Issues arising from Special Cause Variation (Process Control) / Scrap, Downtime, etc. Opportunity arising from Common Cause Variation (Process Improvement) / Yield
The PathWise CAPA System will provide your organization with a Return On Investment for years to come.
-
8/12/2019 Capa Value
2/2
Compliant, Effective, Efficien
www.pathwise.co
866.580.PAT
Value of a Compliant CAPA SystemAs an organization operating within a regulated environment it is extremely important to ensure this criticaQuality System element is suitable and effective for your organization.
The FDA expects an organization to be capable of identifying relevant issues and repairing them in such a way
so they will not reoccur. The expectation is that this is done through a Corrective and Preventive Actionselement within an organizations Quality Management System.Without a compliant and effective CAPA System, it is clear the FDA quickly loses confidence in the
organization and tends to quickly escalate the regulatory response beyond issuing 483 observations. A
compliant and effective CAPA System can make the difference between assuring the FDA your organization is
in control and capable of repairing itself or causing the FDA to place your organization in a higher risk category
by issuing a Warning Letter to your Company.
The PathWise CAPA System ensures control by addressing:
Issues of High Severity (Risk Management) as a priority Issues arising from Special Cause Variation (Process Control) as a priority Effective Execution of CAPA (System and Projects) to prevent the recurrence of issues
The PathWise CAPA System will provide confidence to the FDA that you are in control.
Compliant Satisfies the
regulations or
requirements.
Effective The system
works, as designed, and
you get results.
Efficient work
processes are streamlined
and require minimal
resources necessary for
execution.