BIOB111 SN12 Lecture Lipids · ... alcohols – Lipids are ... of Fatty Acids • Water Solubility...
Transcript of BIOB111 SN12 Lecture Lipids · ... alcohols – Lipids are ... of Fatty Acids • Water Solubility...

BIOCHEMISTRY
Lipids
BIOB111
CHEMISTRY & BIOCHEMISTRY
Session 12

Session Plan
• Introduction to Lipids
• Classification of Lipids
• Structure and Function
• Types of Fatty Acids
• Triacylglycerol's
• Phospholipids
• Sphingolipids
• Cholesterol
• Cell Membranes
• Bile Acids
• Messenger LipidsAn electron micrograph of
adipocytes, the body’s
triacylglycerol-storing cells.
Note the bulging spherical
shape.

Functional groups in Lipid
Stoker 2014, p695
Carboxylicacid
Alcohol
Ester
Lipid
Triacylglycerol (TAG)

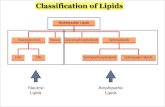
Classification of Lipids• LIPIDS
– Organic compounds found in living organisms that are:• Insoluble in water
• Soluble in non-polar organic solvents
• 5 categories of Lipids based on their function:– Energy-storage lipids:
Triacylglycerol's
– Membrane lipids:
Phospholipids, Sphingoglycolipids & Cholesterol
– Emulsification lipids:
Bile acids
– Chemical messenger lipids:Steroid hormones & Eicosanoids
– Protective-coating lipids:
Biological waxes

Polar/non-polar solubility of compounds
• Lipids (non-polar)
– Lipids are very long hydrocarbons with other attachments present
• O, N or ring structures: can be esters, amides, alcohols
– Lipids are insoluble in polar solvents e.g. H2O
– Lipids are soluble in organic solvents e.g. chloroform (CHCl3)
• Table salt: NaCl (polar)
– NaCl is an ionic compound composed on +ve and –ve ions
• NaCl is soluble in polar solvents e.g. H2O
• NaCl is insoluble in non-polar solvents e.g. chloroform (CHCl3)

Types of Fatty Acids (FAs)• Fatty acids are naturally occurring long chains of carbons that
contain a single carboxylic acid group at one end
• Fatty acids have an even number of C atoms in their chain:– Short-chain FAs: C4 – C6
– Medium-chain FAs: C8 – C10
– Long-chain FAs: C12 – C26
• FAs are rarely found free in nature, they mostly occur as a part of other lipid molecules
• SATURATED FATTY ACIDS (SFAs)– All C-C bonds are single
• UNSATURATED FATTY ACIDS (UFAs)– Monounsaturated FAs (MUFAs) – contain 1 C=C bonds
– Polyunsaturated FAs (PUFAs) – contain 2 or more (up to 6) C=C bonds

Saturated Fatty Acids
• Contain only single C-C bonds in their chain
Stoker 2014, p644

Unsaturated Fatty Acids
• Have at least 1 C=C in their chain.
– Naturally cis-isomers predominate
Stoker 2014, p644

Polyunsaturated Fatty Acids• 2 types of PUFAs:
– Omega-3 = ω-3 FAs
• Polyunsaturated fatty acid with 1st C=C bond
positioned 3 C atoms away from its –CH3 end
– Omega-6 = ω-6 FAs
• Polyunsaturated fatty acid with 1st C=C bond
positioned 6 C atoms away from its –CH3 end.
• Essential Fatty Acids (EFAs) – must be obtained from the diet,
as the body can not make them
• Linoleic acid (ω-6) – 18:2
• Linolenic acid (ω-3) – 18:3
18:3No. of carbons
No. of double bonds between
carbons

PUFAs
ω-3 (18:3)
Stoker 2014, p645-6
Linoleic acid
Linolenic acid
Monounsaturated fatty acid

Stoker 2014, Table 19-1 p646

Physical Properties of Fatty Acids
• Water Solubility
– Is a direct function of the C chain length
– Short-chain FAs have slight solubility in
water
• Due to the polarity of Carboxyl group
– Long-chain FAs are insoluble in water
http://www.jamesciesla.com/wp-content/uploads/2014/12/oilwaterinbeaker.jpg

Physical Properties of Fatty Acids• RULE: The more double carbon to
carbon bonds a fatty acid has the lower the melting point
• Saturated fatty acids are more stable than unsaturated fatty acids
– Saturated fatty acids have a highermelting points than unsaturated fatty acids
– Unsaturated fatty acids have lower melting points
• The single carbon to carbon bonds within saturated fatty acids result in straight chains, meaning the saturated fatty acids easily stack on top of each other
• The double carbon to carbon bonds within unsaturated fatty acids result in bending, meaning it is difficult for the unsaturated fatty acids to stack on top of each other
Stoker 2014, Figure 19-2 p648

Stoker 2014, Figure 19-3 p648
Space-filling models of four C18 FAs, which differ in the number of C=C present.
More double bonds = more bending

Triacylglycerol's (TAGs)
• Triacylglycerol's are energy storage lipids– Tri-esters of a Glycerol molecule & 3 long-chain FAs
• Triacylglycerol's are found in animal fats & vegetable oils
Stoker 2014, p650
Acyl group

Triacylglycerol's• Most fats & oils are mixed TAGs
– They contain 2-3 different FAs
– 18:0, 18:1 and 18:2 are all examples of different of fatty acids
• TAGs are the most common– Monoacylglycerol’s & diacylglycerol’s also common
• TAGs insoluble in water:
– Hydrophobic (water-repellent) character of fats is due to long hydrocarbon chains
– The Ester groups (–COO–) within TAG are polar
• Hidden in the non-polar environment of
the long carbon chainsStoker 2014, Figure 19-6 p650

Fats & Oils• Fats & oils are naturally occurring mixtures of TAGs,
in which many different kinds of TAG molecules are present
• FAT– Mixture of TAGs predominantly with saturated FAs
• Solid at room temperature (250C)
• Usually from animal sources
• Can easily stack on top of each other
= strong intermolecular forces
• OIL– Mixture of TAGs predominantly with unsaturated FAs
• Liquid at room temperature (250C)
• Usually from plant sources
• Hard for them to stack on top of each other due to bending
= weaker intermolecular forces than fats
Stoker 2014, Figure 19-7 p652

Explain the difference between saturated
fatty acids and unsaturated fatty acids, with
reference to carbon to carbon double and single bonds.
Why do lipids that contain mostly
saturated fatty acids form solid fats?
Why do lipids that contain mostly
unsaturated fatty acids form liquid oils?
G
Key concept: saturated and unsaturated fatty acids,physical state at room temp.

Attempt Socrative questions: 1 to 3
Google Socrative and go to the student login
Room name:
City name followed by 1 or 2 (e.g. PERTH1)
1 for 1st session of the week and 2 for 2nd session of the week

Hydrolysis of TAGs
• Hydrolysis is the reverse of Esterification– TAGs are formed by esterification (3FA + glycerol >>> TAG)
– TAGs are broken down by hydrolysis (TAG >>> 3FA glycerol)
• Complete Hydrolysis– In a laboratory setting
– Acid & steam are required (see Reactions of Esters in SN10).
• Partial Hydrolysis– In the small intestine:
• Pancreatic lipase enzymes catalyze the hydrolysis of dietary TAGs – Producing Monoacylglycerol’s & free FAs

Stoker 2010, Figure 19-10 p659

Saponification
• Saponification is hydrolysis of TAGs in an alkaline (basic) solution– Products = glycerol & fatty acid salts
• Fatty acid salt = soap
• Involves 2 steps:– 1. Hydrolysis of TAGs (esters)
Fat or Oil + 3 H2O → 3 Fatty Acids + Glycerol
– 2. Acid-Base reaction
3 Fatty Acids + 3 NaOH → 3 Fatty Acid Salts + 3 H2O
Sodium stearate

The Cleansing Action of Soaps
Stoker 2014, p661
Micelle formation
Grease isnon-polar.
Grease is kept away from the
H2O by the non-polar lipid
tails of the soap
Polar head group allows
soap to be stable in H2O

Hydrogenation of TAGs
• Addition of H atoms to an unsaturated organic compound,
which decreases the degree of unsaturation– Can become saturated (see below)
Stoker 2013, p671
Stoker 2014, Figure 19-11 p662

Trans Fatty Acids (TFAs)
• Trans fatty acids are found naturally in meat & dairy– Do not cause problems
• During the production of margarine– Excessive TFAs are the by-product of partial hydrogenation
of unsaturated fatty acids (UFAs)• Not all double bonds present in the UFAs are hydrogenated
• Some double bonds will isomerize from cis- to trans-UFA– There is sufficient energy during hydrogenation to alter the position
of the hydrogen atom near the double bond » Results in cis to trans switch
The human body is not well equipped to
process large amounts of TFAs
– Studies indicate that TFAs contribute to:
• Increased LDL cholesterol (“bad cholesterol”)
• Development of cardiovascular disease (CVD)

Stoker 2014, p663
Structural changes occurring during partial hydrogenation of UFAs

Oxidation of TAGs
• The C=C in UFAs can oxidize when exposed to oxygen from
the air, leading to C=C breakage– Produces aldehydes or carboxylic acids
• Have unpleasant odors & are responsible for the rancid smell of oxidized fats & oils
• Antioxidants are added to commercially produced foods to prevent this oxidation
Stoker 2014, p662

Stoker 2014, p665
Structure of TAGs
Summary
Lipids are implicated in a number of human diseases, including cancer &
cardiovascular disease, sometimes in a detrimental & sometimes in a
beneficial manner.

Attempt Socrative questions: 4 to 7
Google Socrative and go to the student login
Room name:
City name followed by 1 or 2 (e.g. PERTH1)
1 for 1st session of the week and 2 for 2nd session of the week

Membrane Lipids
• All body cells are surrounded by a cell membrane
– Separates the intracellular from the extracellular environment
• Up to 80% % of the cell membrane is composed of lipids & the
remaining 20% are mostly proteins
• 3 types of membrane lipids:
– Phospholipids – the most abundant
– Sphingoglycolipids
– Cholesterol

Phospholipids• Composition of Phospholipids:
– Platform molecule: glycerol or sphingosine
– 1 or more FAs (SFAs or UFAs)
– Phosphate group
– Alcohol (usually Choline)
Stoker 2014, p666

Glycerophospholipids
• Platform molecule: Glycerol
• 2 Fatty Acids
• Alcohol: 1 of 3 Aminoalcohols– Choline – Phosphatigylcholines (Lecithins)
– Ethanolamine – Phosphatidylethanolamines
– Serine – Phosphatidylserines
NOTE:4 ester bonds
present
Stoker 2014, p666

Stoker 2014, Figure 19-13 p669
Phosphatidylcholines are Amphipathic• Polar head – Hydrophilic (“water-liking”)
– Alcohol (Choline) + phosphate
• Non-polar tails – Hydrophobic (“water-hating”)– Long fatty acid hydrocarbon tail

Sphingoglycolipids
• Sphingoglycolipids contain– Fatty acid & Carbohydrate attached to
Sphingosine
• Cerebrosides = simple sphingoglycolipids, contain a single monosaccharide unit (either glucose or galactose)
– Primarily in brain (7% of dry mass)
– Myelin sheath (surround the core of nerve)
Stoker 2014, p671-2

Steroids
• Steroids are lipids based on a fused-ring structure:– Three 6-membered & one 5-
membered rings
• The steroid nucleus has a designated numbering system
• Steroids differ in the location of their substituents (branches) off the steroid nucleus
Stoker 2014, p673

Cholesterol
• Cholesterol is a C27 steroid compound– Suffix –ol indicates that
Cholesterol is an alcohol
• Cholesterol is the most abundant steroid in the body.– Important component of cell
membranes & nervous tissue
– Precursor to other Steroids
Stoker 2014, Figure 19-15 p674

Cholesterol • Cholesterol is found in cell membranes of all body
cells (25% of membrane mass),
in brain tissue (10%) & in all body fluids– 100 mL of blood plasma contains:
• 50 mg of free cholesterol
• 170mg of cholesterol esterified with various FAs
• Apart from dietary cholesterol, the body makes its own cholesterol in the liver
– 800–1,000 mg made daily
– Transported via blood to all tissues
• Cholesterol is not soluble in water & requires a protein carrier called lipoprotein– Lipoproteins = the combination of cholesterol &
protein.
Low-Density Lipoproteins (LDLs)• Transport cholesterol from the
liver to other tissues• “bad cholesterol” >>>
development of Atherosclerotic plaque
• Hardening of the arteries
High-Density Lipoproteins (HDLs)• Transport excess cholesterol back
to the liver• “good cholesterol”
• The liver uses cholesterol to make fatty acids

Cell Membranes
• The structure of the cell membrane = “Fluid mosaic”
– The cell membrane is formed as a phospholipid bilayer
– Not static, can move around easily
• The polar heads of phospholipids are positioned towards the polar environments
– the watery extracellular fluid & the cytosol
• The non-polar tails are orientated towards each other within the membrane,
– Do not come in contact with water.
Stoker 2014, Figure 19-17 p676

Stoker 2013, Figure 19-21 p686
Proteins & cholesterol are important
structural components of cell membranes.

Which part of a phospholipid is referred to as
the polar head and which is the non-polar tail?
How are the polar heads and non-polar tails within the
phospholipids arranged within the lipid bilayer?
Is it the polar heads or non-polar tails that are exposed to
the polar environment (H2O) inside & outside the cell? Why?
G
Key concept: phospholipids, polarity, cell membranes

Attempt Socrative questions: 8 to 11
Google Socrative and go to the student login
Room name:
City name followed by 1 or 2 (e.g. PERTH1)
1 for 1st session of the week and 2 for 2nd session of the week

Bile Acids
• Bile acids are cholesterol derivatives– Emulsify dietary lipids in the aqueous environment of the digestive tract
• Emulsifier = a substance that can disperse & stabilize non-polar substances in aqueous solutions
– Similar activity to soap.
• Liver converts about 1/3-½ of its daily cholesterol production to bile acids– An amino acid (Glycine or Taurine) is always part of the bile acid structure
• Bile = fluid containing emulsifying bile acids– Secreted by the liver
– Stored in the gallbladder
– Released into the small intestine during digestion

Stoker 2014,
Figure 19-24 p681
The 2 main Bile Acids

Messenger Lipids – Steroid Hormones
• Hormones serve as a means of communication between various tissues
– Some hormones are lipids
• SEX HORMONES >>> steroid hormones
– Androgens (Testosterone)
– Estrogens (Estradiol)
– Progestins (Progesterone)
• ADRENOCORTICOID HORMONES– Mineralocorticoids (Aldosterone)
– Glucocorticoids (Cortisol)
Stoker 2014, Figure 19-26 p682
Stoker 2014, Figure 19-27 p683

Structures of selected
adrenocorticoid hormones
& related synthetic compounds

Messenger Lipids – Eicosanoids• Arachidonic acid (20:4) derivatives
– Have profound physiological effects at extremely low concentrations & a very short “life”
– Act as local hormones
Physiological effects of Eicosanoids:
• PROSTAGLANDINS– Involved with contraction & relaxation of smooth muscle
– Water & electrolyte balance
– Enhancing inflammation
– Pain & body temperature
• THROMOXANES– Produced by platelets (Thrombocytes)
– Initiate platelet aggregation & formation of a blood clot (Thrombus)
• LEUKOTRIENES– Produced by white blood cells (Leukocytes)
– Promote inflammation & hypersensitivity (allergy) responses

Biological Waxes• Biological waxes are esters of a long-chain (C14-36)
saturated fatty acid & a long-chain (C16-30) alcohol– Found in plants
• Form coatings of fruits, leaves & stems– Prevent water loss & damage from pests
• Waxes also found on animals, provide a water proof coating– Skin, Fur, Feathers
– Lanolin = a mixture of waxes from sheep wool, used in creams & ointments to retain water & soften skin
– Beeswax & Carnauba wax (Brazilian palm tree)• Used in furniture, car & floor polishes & waxes

Stoker 2014, p690
Lipids
Summary

Readings & Resources• Stoker, HS 2014, General, Organic and Biological Chemistry, 7th edn,
Brooks/Cole, Cengage Learning, Belmont, CA.
• Stoker, HS 2004, General, Organic and Biological Chemistry, 3rd edn, Houghton Mifflin, Boston, MA.
• Timberlake, KC 2014, General, organic, and biological chemistry: structures of life, 4th edn, Pearson, Boston, MA.
• Alberts, B, Johnson, A, Lewis, J, Raff, M, Roberts, K & Walter P 2008, Molecular biology of the cell, 5th edn, Garland Science, New York.
• Berg, JM, Tymoczko, JL & Stryer, L 2012, Biochemistry, 7th edn, W.H. Freeman, New York.
• Dominiczak, MH 2007, Flesh and bones of metabolism, Elsevier Mosby, Edinburgh.
• Tortora, GJ & Derrickson, B 2014, Principles of Anatomy and Physiology, 14th edn, John Wiley & Sons, Hoboken, NJ.
• Tortora, GJ & Grabowski, SR 2003, Principles of Anatomy and Physiology, 10th edn, John Wiley & Sons, New York, NY.