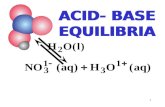Applied Geochemistry & Lab Ch.1 Phase Equilibria in a Simple System
-
Upload
eliana-moody -
Category
Documents
-
view
49 -
download
0
description
Transcript of Applied Geochemistry & Lab Ch.1 Phase Equilibria in a Simple System

JYU
Applied Geochemistry & Lab
Ch.1 Phase Equilib-ria in a Simple Sys-
tem

Studying the basic principles of Understanding equilibrium assemblage
under a certain T, P, X Estimating T, P, and X from the equilib-
rium assemblages Determining if it’s really in equilibrium
AIMS

Phase (P): A part of the system, which is physicochem-ically homogeneous and mechanically separable from the rest.
Component (C): An operator consisting of the mini-mum number of the operators being capable of repre-senting the compositions of all the phases in the system Depending on the # of C Unary, binary, ternary, quaternary
system, …… Degree of Freedom (F): The minimum # of the inde-
pendent variable required to define the state of the sys-tem. Depending on the # of F bi(di)variant field, univariant
curve, invariant pt.
1. DEFINITIONS

P + F = C + 2
(for two independent variable system)
Can you tell P, C, and T in the following ex-amples?Can you apply the phase rule?
2. Gibbs’ Phase Rule


http://serc.carleton.edu/research_education/equilibria/phaserule.html