5 immunology-csbrp
-
Upload
prasad-csbr -
Category
Health & Medicine
-
view
758 -
download
0
Transcript of 5 immunology-csbrp

AmyloidosisAmyloidosisDr.CSBR.Prasad, M.D.
APR-2015-CSBRP

Gist - AMYLOID
• It’s only a manifestation of different diseases• it’s an abnormal protein • It’s deposited Extracellularly• It’s bland and Non-reactive • Enzyme resistant• Affects all tissues• It causes organ failure by pressure atrophy
APR-2015-CSBRP

Gist - AMYLOID
Two processes are associated with such deposition
• Prolonged inflammation• Excessive immunoglobulin synthesis
APR-2015-CSBRP

Amyloid
Amyloid is a pathologic proteinaceous substance, deposited between cells in
various tissues and organs of the body in a wide variety of clinical settings.
APR-2015-CSBRP

AmyloidAmyloid
With the light microscope and standard tissue stains, amyloid appears as
• An amorphous, eosinophilic, hyaline like, extracellular substance
APR-2015-CSBRP

DD Hyaline & AmyloidFeatureFeature HyalineHyaline AmyloidAmyloidLocation Both extra & intracellular Extracellular
Nature Protein / others Protein
Inflammation Inflammatory reaction No inflammation
Etiology Variety of agents Immunological
H&E Eosinophilic Eosinophilic
Congo red Negative Positive
CR+Polarizer No change Apple green
EM variety of morphology Nonbranching fibrilsAPR-2015-CSBRP

Amyloid
• Is not a single protein• Chemically different proteins with
identical staining properties
APR-2015-CSBRP

Amyloid – Physical nature
By electron microscopy:By electron microscopy: (It’s common to all amyloid proteins(It’s common to all amyloid proteins))
• Non-branching fibrils• Diameter of 7.5-10 nm• cross-β-pleated sheet conformation
This β-pleated nature is responsible for its special staining character
APR-2015-CSBRP

Amyloid – Physical nature
APR-2015-CSBRP

The Nature of Amyloid
Ultrastructure
Structure of an amyloid fibril
APR-2015-CSBRP

Amyloid – Chemical nature
Mainly composed of:
1. Fibrillary protein - 95%2. P-component - 5%3. Glycoprotein - a fraction
APR-2015-CSBRP

gist - The Nature of Amyloid• Physical Nature:
• Electron microscopy: Amyloid material contains two components. (1) Rigid nonbranching fibrils* (7.5 to 10 nm):
Constitute the major component (about 90%) (2) Pentagonal rod shaped sub-unit (P components): Forms 10% of amyloid tissue and is a glycoprotein (variable PAS positive staining).
• X-ray diffraction analysis: Characteristic β-pleated configuration* in both major subtypes of amyloid.
• Chemical nature: • Peptide mapping analysis of the sequence of amino
acids: Of the 15 biochemically distinct forms identified, 3 are major chemical types of amyloid: (1) Amyloid
of light chain origin (AL) from plasma cells, (2) Unique nonimmunoglobulin protein synthesized in the liver (AA), (3)
Aβ amyloid in cerebral lesion of Alzheimer disease
APR-2015-CSBRP

Classification
Amyloid may be:Amyloid may be:Systemic (generalized), involving several organ systems, or
Localized, when deposits are limited to a single organ, such as the Heart, Skin et.c.
APR-2015-CSBRP

Localized Amyloid DepositsEndocrine associated:
• Amyloid deposits in the stroma of medullary thyroid carcinoma, a C-cell tumor; amyloid is derived from a polypeptide hormone calcitonin.
• Amyloid deposits in the stroma of islet cell tumors, particularly those producing insulin (amyloid is produced by proteolysis of insulin or its
prohormone).Senile amyloidosis:
• Deposits are common in the aged (seventies and eighties); found in heart (atria affected more frequently than ventricles) and brain (as
senile plaques). • The plaques found in Alzheimers disease is derived from β-amyloid
protein (Aβ), derived from a glycoprotein called amyloid precursor protein (APP).
Other localized deposits: Microscopic deposits to nodular masses in sites such as lung, larynx, skin, bladder, and tongue.
Consist of AL protein in most cases.
APR-2015-CSBRP

Clinicopathologic Category Associated Diseases Major Fibril Protein
Chemically Related Precursor Protein
Systemic (Generalized) AmyloidosisImmunocyte dyscrasias with amyloidosis (primary amyloidosis)
Multiple myeloma and other monoclonal B-cell proliferations AL Immunoglobulin light
chains, chiefly λ type
Reactive systemic amyloidosis (secondary amyloidosis) Chronic inflammatory conditions AA SAA
Hemodialysis-associated amyloidosis Chronic renal failure Aβ2m β2-microglobulin
Hereditary amyloidosis
Familial Mediterranean fever — AA SAA
Familial amyloidotic neuropathies (several types) — ATTR Transthyretin
Systemic senile amyloidosis — ATTR Transthyretin
Localized AmyloidosisSenile cerebral Alzheimer disease Aβ APP
Endocrine
Medullary carcinoma of thyroid — A Cal Calcitonin
Islet of Langerhans Type II diabetes AIAPP Islet amyloid peptide
Isolated atrial amyloidosis — AANF Atrial natriuretic factor
Prion diseases Various prion diseases of the CNS
Misfolded prion protein (PrPSC)
Normal prion protein PrP
APR-2015-CSBRP

Classification of Systemic AmyloidosisClassification of Systemic Amyloidosison clinical groundson clinical grounds
• Primary amyloidosis, when associated with some immunocyte dyscrasia, or
• Secondary amyloidosis, when it occurs as a complication of an underlying chronic inflammatory or tissue destructive process.
• Hereditary or familial amyloidosis constitutes a separate, albeit heterogeneous group, with several distinctive patterns of organ involvement.
• Hemodialysis associated Amyloidosis• Localized amyloidosis - (A). Endocrine
associated, (B). Senile, and (C). Others
APR-2015-CSBRP

Immunocyte Dyscrasias with Amyloidosis (Primary Amyloidosis)
• Most common form of amyloidosis• many of these patients have some form of
plasma cell dyscrasia / B-cell dyscrasia• Amyloid has AL composition
APR-2015-CSBRP

Reactive Systemic Amyloidosis• Secondary to an inflammatory condition • Protracted breakdown of cells• Composed of AA protein
Chronic osteomyelitis Rheumatoid arthritis
Bronchiectasis Connective tissue diseaseTuberculosis RCCLeprosy Hodgkin’s disease
APR-2015-CSBRP

Patients on long-term hemodialysis for renal failure High β2-microglobulin in the serum Amyloid deposits in the synovium, joints, and
tendon sheaths
Hemodialysis-Associated Amyloidosis
APR-2015-CSBRP

• Many familial forms of amyloidosis• Confined to limited geographic areas• The most common is familial Mediterranean fever• Others - familial amyloidotic polyneuropathies
Heredofamilial Amyloidosis
APR-2015-CSBRP

Microscopic localized deposits of amyloid in endocrine tumors,
Eg: Medullary carcinoma of the thyroid gland, Islet tumors of the pancreas Pheochromocytomas and Undifferentiated carcinomas of the stomachAmyloid derived from hormones or unique proteins
Endocrine Amyloid
APR-2015-CSBRP

Several well-documented forms of amyloid deposition occur with aging
Senile systemic amyloidosis - systemic deposition of amyloid in elderly patients called senile cardiac amyloidosis
Amyloid of Aging
APR-2015-CSBRP

Amyloid – Chemical nature
Fibrillary component: 15 bio-chemically distinct forms have been identified
Three are most common: (1) AL (amyloid light chain) immunoglobulin light
chains(2) AA (amyloid-associated) nonimmunoglobulin
protein synthesized by the liver and (3) Aβ amyloid is found in Alzheimer disease
APR-2015-CSBRP

Proposed Scheme of Pathogenesis of Two Major Forms of Amyloid Fibrils
APR-2015-CSBRP

Pathogenesis of Amyloidosis
• Amyloid fibrils (AL protein or AA protein) formed in vivo from a variety of polypeptide fragments, derived
either from immunoglobulin of plasma cell origin or from other protein (SAA) of hepatocytic origin.
• Amyloidogenic proteins (Immunoglobulin light chains or SAA) present in the blood is deposited in
various sites following limited proteolysis by the action of monocyte-derived enzymes.
• Thus, mononuclear phagocyte system may play a role by taking up immunoglobulins, Ag-Ab
complexes, or other proteins and produce necessary polypeptides for formation of amyloid fibrils.
APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

The “Beta-Sheet”, rigid folding pattern
APR-2015-CSBRP

Morphology
APR-2015-CSBRP

Specific Organ Involvement in Amyloid Disease
• Kidney • Liver• Spleen• Heart• Lymph nodes• Adrenal• Thyroid• Pituitary• Gastrointestinal tract• Respiratory tract
APR-2015-CSBRP

Cutaneous Amyloidosis
APR-2015-CSBRP

Skin
APR-2015-CSBRP

APR-2015-CSBRP

Bilateral periorbital ecchymosis (raccoon eyes) and mild proptosis.
Another example of "raccoon eyes" which is a clinical sign of a base of skull fracture.
APR-2015-CSBRP

APR-2015-CSBRP

Kidney in AmyloidosisGross: The organ is usually enlarged and pale but a small scarred kidney results in long standing cases due to secondary ischemic changes.
Micro: Amyloid is deposited primarily in the glomeruli. Mesengial deposition and the deposits along basement membrane cause capillary narrowing and distortion of glomerular tuft. The arterioles, and interstitial tissue between the tubules are also affected.
APR-2015-CSBRP

Kidney in Amyloidosis
Clinical impact: Renal involvement in amyloidosis is most common and result is nephrotic syndrome, often as the presenting feature. It is the most serious manifestation and major cause of death.
APR-2015-CSBRP

Renal biopsy in AL amyloidosisStaining with anti-K light chain Abs
APR-2015-CSBRP

Renal biopsy in secondary AA amyloidosisGlomerulus is strongly positive for AA protein
APR-2015-CSBRP

Liver in Amyloidosis
Gross: Liver is enlarged, heavy, pale, and firm.Micro: Amyloid is deposited in the space of Disse between the endothelium and the liver cells, and progressively encroaches on adjacent hepatocytes and sinusoids. The liver cells undergo atrophy. Vascular involvement and Kupffer cell depositions are frequent.Clinical impact: Liver function is preserved in the presence of marked involvement.
APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

Spleen in Amyloidosis
Gross: Spleen is enlarged and firm. Focal distribution is common. The cut surface shows numerous translucent nodules distributed
throughout the red pulp (characteristic appearance, called ‘sago spleen’). A diffuse type is also recognized.
Micro: In localized type amyloid is laid down in the walls of the arterioles in the white pulp and subsequently replaces the malpighian
bodies. In diffuse type, amyloid is laid down in the walls of the sinuses and tends to spare the follicles. Fusion of early deposits lead
to large map-like areas of amyloidosis (lardaceous spleen). APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

Cycus Revoluta (Sago palm)
APR-2015-CSBRP

Lardaceous spleen
• fat from the abdomen of a pig, especially as prepared for use in cooking
APR-2015-CSBRP

Lardaceous spleen
• fat from the abdomen of a pig, especially as prepared for use in cooking
APR-2015-CSBRP

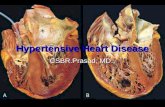
Heart in AmyloidosisCardiac involvement is the major manifestation of primary amyloidosis. It occurs in multiple myeloma and usually in elderly people.Gross: Heart is enlarged, rigid, and firm.Micro: Begin with focal subendocardial deposition. In due course widespread deposition of amyloid in the walls of blood vessels and in the interstitial tissue surrounding and replacing muscle fibers.
Clinical impact: Cardiomegaly with intractable heart failure. Heart block and arrhythmia due to involvement of conduction system are common.
Condition mimicking constrictive pericarditis due to rigidity of the muscle.
APR-2015-CSBRP

CNS
APR-2015-CSBRP

Congo Red Staining of Liver in Amyloidosis
APR-2015-CSBRP

Clinical feature
APR-2015-CSBRP

Immunocyte-derived Systemic Amyloidosis
• Found in with 5 to 15% multiple myeloma patients and in patients with B-cell dyscrasias (monoclonal B-cell proliferations).
• Amyloid is systemic indistribution (AL type) and derived from Ig light chains (chiefly λ type).
• An excess of corresponding light chain is usually present in the blood and secreted in urine as Bence-Jones protein
Organ involvement and its effect:• Kidney involvement is a major feature and usual cause of death.
• Myocardium : refractory heart failure.• Tongue: enlargement interferes with speech.
• Esophagus: dysphagia.• Intestine: lesions lead to obstruction, bleeding, and perforation.
• Skin: deposits cause purpura.• Nerve: peripheral neuropathy.
• The liver, spleen, lungs, and endocrine organs are affected less frequently.
APR-2015-CSBRP

Diagnosis
APR-2015-CSBRP

Diagnosis of Amyloid Disease in Life• Amyloid may be demonstrated in biopsy material obtained from liver,
spleen, kidney, intestine, and bone marrow. Most common site of biopsy is kidney, when renal manfestations are present.
• Tissue involved by amyloidosis has a tendency to bleed when subjected to trauma. Biopsy of gingiva and rectal mucosa has
advantage over other sites in that, and should hemorrhage occur, it can readily be controlled.
• Fine needle aspiration biopsy of the abdominal fat is also an easily followed and extemely useful procedure for detection of systemic
amyloidosis.• Appearance in routine stains: With light microscope and standard
tissue stains, amyloid appears as an amorphous, eosinophilic, hyaline, extracellular substance.
• Appearance by special stain (Congo red): Amyloid stains an orange red color under ordinary light but when the stained slide is viewed in a
polarizing microscope, a characteristic green birefringence is imparted.
• Other investigations: In immunocyte-associated amyloidosis, serum and urine protein electrophoresis, and immunoelectrophoresis. Bone
marrow aspiration for plasmacytosis.
APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

APR-2015-CSBRP

Amyloid - Apple green birefringence under polarized light
APR-2015-CSBRP

APR-2015-CSBRP

Demonstration of AmyloidStain Result TechniqueLugol’s Iodine Mahagany brown – Blue On Fresh tissue
Methyl violet Pink to Red Metachromasia
Thiflavine-T Bright red fluorescence Fluorescent technique
Thioflavine-S Bright red fluorescence Fluorescent technique
Alcian blue Green Chemical
Silver impregnantion Black Impregnation
Congo red Red Beta-pleated nature
CR + Polarization Apple green birefringens Polarization
Sirius red Green PolarizationAPR-2015-CSBRP

Amyloidosis, Lugol's Reaction
APR-2015-CSBRP

APR-2015-CSBRP

E N D
APR-2015-CSBRP